Abstract
Background. Many experimental data indicate interactions between peptides involved in the control of food intake, energy homeostasis and adrenocortical hormone release. Glucocorticoids stimulate or inhibit the secretion of orexigenic and anorexigenic peptides, which in turn are involved in the regulation of adrenal growth, structure and function. Galanin-like peptide (Galp) and alarin (Ala) are involved in the regulation of food intake. Galp and Ala mRNAs have already been shown to be present in the arcuate nucleus (ARC) of the hypothalamus in both rats and mice.
Objectives. To investigate the expression of Ala, Galp and their receptors in the hypothalamus and pituitary and adrenal glands of the rat hypothalamic–pituitary–adrenal (HPA) axis after intraperitoneal administration of peptides in vivo.
Materials and methods. Experimental in vivo models were used: acute and long-term exposure to peptides.
Results. The expression of Galp, Ala, their receptors, and steroidogenesis enzymes was analyzed using quantitative real-time polymerase chain reaction (qRT-PCR). Statistically significant expression changes were found in the hypothalamus and pituitary after 1-hour exposure to the peptides, such as a decrease in corticotropin-releasing hormone (CRH) expression after Ala, Galp and adrenocorticotropic hormone (ACTH) administration, and a decrease in the expression of receptors for galanin (Gal) (Galr1 and Galr2). In the pituitary, there was a statistically significant increase in the expression of Ala, Galr1, Galr2, and Galr3 receptors 1 h after Galp administration. In the adrenal glands, only a statistically significant decrease in Galr2 expression was observed after 1 h of Ala 0.5 administration. The mRNA expression of steroidogenesis enzymes also changed: for example, the expression of cholesterol desmolase increased 24 h after Ala peptide administration.
Conclusions. The results indicate that the peptides tested under in vivo conditions can alter the expression of the peptides tested, as well as of Galp, Ala and Gal receptors and steroidogenesis enzymes – Cyp11a1 (cholesterol desmolase), Cyp11b1 (11β-hydroxylase) and Cyp11b2 (aldosterone synthase).
Key words: adrenal gland, HPA axis, Galp, alarin (Ala), in vivo experiments
Background
Adrenal glands are known to be involved in the maintenance of energy homeostasis in the body. There are 2 principal mechanisms of energy homeostasis regulation, central and peripheral, which involve many different hypothalamic peptides, such as neuropeptide Y (NPY), orexins (Ox), proopiomelanocortin (POMC), ghrelin (Ghrel), and cerebelin (Cer), that are directly or indirectly involved in food intake. Numerous experimental data have provided evidence of interactions between peptides involved in the control of food intake, energy homeostasis and steroidogenic hormones released by the adrenal glands.1, 2, 3 It is well known that glucocorticoids stimulate NPY and inhibit corticotropin-releasing hormone (CRH) or POMC secretion.4, 5, 6 On the other hand, numerous orexigenic and anorexigenic peptides are involved in the regulation of growth, structure and function of the adrenal gland, such as galanin (Gal), which is the “parental” peptide of a family that also includes galanin-like peptide (Galp) and alarin (Ala). As it is known, Gal mRNA and Gal receptors are expressed in rat adrenocortical cells.7, 8, 9, 10 Gal is known to be involved in the control of energy homeostasis and plays an important role in the regulation of adrenocortical hormone secretion.7 Moreover, Gal stimulates cortisol secretion from human adrenocortical cells, acting through galanin receptor type 1 (Galr1), and stimulates the release of corticosterone and cyclic-AMP from dispersed inner rat adrenocortical cells.7, 9, 10, 11, 12
More than 20 years ago, Galp was first isolated from the porcine hypothalamus as an endogenous ligand of galanin receptor type 2 (Galr2).13 Later study provided evidence that Galp interacts also with other galanin receptor isoforms (Galr1 and Galr3).14 Galp mRNA was identified in different species, including rat, mouse and human.13, 15, 16 Immunohistochemical studies demonstrated that around 85% of arcuate nucleus Galp-positive neurons (ARC-Galp) also express leptin receptors. There are already reports in the literature regarding the involvement of the Galp peptide in the regulation of body homeostasis.17, 18, 19, 20
Another member of the Gal family – Ala, a 25-amino acids peptide – arises as a splice variant of the Galp gene.21, 22 This variant results from the exclusion of exon 3. Therefore, the N-terminal end of both Galp and Ala precursors is the same – it comprises the same signal sequence and proteolytic cleavage site. Alarin peptide was isolated from mouse brain and thymus.22 Alarin-like immunoreactivity was observed in the locus coeruleus (LC) and the ARC of rats and mice. Both regions are involved in feeding behavior and homeostatic control. Alarin was identified as an orexigenic compound involved in the regulation of reproductive hormones secretion, an effect mediated through hypothalamic gonadotropin releasing hormone 1 (Gnrh1), but also in the regulation of feeding behavior in male rats and stimulation of NPY release from hypothalamic explants.23, 24
Based on previous reports on the involvement of Galp and Ala peptides in homeostatic maintenance processes, and considering the role of Gal in adrenal cell function, the hypothalamic–pituitary–adrenal (HPA) axis and Gal involvement in the regulation of energy homeostasis, there is still a lack of data on the involvement of both Galp and Ala in the regulation of adrenal hormone secretion, and the interaction of the HPA axis.25 Previous quantitative polymerase chain reaction (qPCR) studies have demonstrated Gal receptor mRNA expression in rat adrenal glands (mainly Galr2 and Galr3).8 Since the HPA axis and adrenal function are closely linked to stress regulation and maintenance of energy homeostasis, it would be interesting to determine whether Galp and/or Ala, like Gal, affect HPA axis gene expression or adrenal function.
Objectives
The aim of the study was to investigate the expression of Ala, Galp and their receptors in hypothalamus, pituitary gland and adrenal gland of the HPA axis of the rat after in vivo intraperitoneal (ip.) peptides administration.
Materials and methods
Animals, reagents
and experimental design
Adult male Wistar rats (final body weight 100–150 g) from the Laboratory Animal Breeding Center, Poznan Science and Technology Park of Adam Mickiewicz University Foundation, Poznań, Poland (the specific-pathogen-free (SPF) category), were used. The total number of animals used was 144 (104 in the acute exposition group and 40 in the prolonged exposition group). This study was carried out in accordance with the recommendations of the Directive 2010/63/EU of the European Parliament and of the Council of 22 September, 2010 on the protection of animals used for scientific purposes, as stated in the Polish law (Act of January 15, 2015 on the Protection of Animals Used for Scientific or Educational Purposes). The study protocol was approved by the Ethics Committee for Animal Studies of the Department of Animal Physiology, Biochemistry and Biostructure, Poznań University of Life Sciences (resolution No. 11/2015). Animals of both experimental groups were maintained in constant, strictly defined conditions, i.e., at a temperature of 22°C ±2°C and air humidity 55–60%, in a daily cycle of 12 h of light (12-hour continuum)/12 h of dark (12-hour continuum), in a room where the air exchange was at the level of 15 exchanges/h, with free access to standard pellets and tap water. Galp and Ala peptides were obtained from Phoenix Pharmaceuticals, Inc. (cat. No. 026-52 and 026-33, respectively; Phoenix, USA).
In order to determine the effect of both peptides (Galp and Ala) on HPA axis genes expression, 2 experimental in vivo models were used: acute and prolonged exposition to peptides. During the surgical operation, both control and experimental groups of animals were under standard ketamine and xylazine anesthesia (100 mg/kg of ketamine ip. and 10 mg/kg of xylazine ip.). All experiments were performed between 9 AM and 12 AM. All possible efforts have been undertaken to minimize the number of animals and their suffering.
One-hour and 24-hour exposure to the peptides
The experimental model consisted of ip. peptides administration. Animals were initially injected ip. daily for 2 weeks with 0.2 mL of 0.9% saline to prepare for the experiment. Next, they were given an ip. injection of tested substances and peptides. After the exposition time (1 h – the acute exposition group, or 24 h – the prolonged exposition group), the animals were decapitated. The trunk blood was collected in the presence of ethylenediaminetetraacetic acid (EDTA; 1 mg/mL), and plasma was separated and stored at −20°C until biochemical assays were performed. Hypothalami, pituitary glands and adrenal glands were collected to an RNAlater (Sigma-Aldrich, St. Louis, USA) and stored in −80°C for further qRT-PCR analysis.
Each experimental group consisted of 8 animals and there were 13 groups (7 groups in the 1 h experiment and 6 groups in the 24 h experiment, 104 animals in total), as follows: 0,9% NaCl × 2 (negative control), 60% acetonitrile in water with dimethyl sulfoxide (DMSO) × 1 (Galp negative control), Galp (1.5 nmol) × 2, Galp (0.5 nmol) × 2, Ala (1.5 nmol) × 2, Ala (0.5 nmol) × 2, and ACTH (10−7) × 2 (adrenocorticotropic hormone, positive control). Since the Galp peptide was dissolved in 30 μL of 60% acetonitrile in water with DMSO, and it is known that DMSO affects the expression of genes, additional negative control by means of Galp solvent was used.27 Alarin peptide was dissolved in 0.9% saline. Peptide concentrations were established based on earlier experimental data and the team members’ experience.26, 27
Two-day exposure to peptides
The experimental model consisted of subcutaneous (sc.) mini osmotic pumps (ALZET 2001; Durect Corp., Cupertino, USA). Animals were first administered daily for 2 weeks with a sc. injection of 0.2 mL of 0.9% saline to prepare them for the experiment. Next, they underwent a surgical operation during which mini osmotic pumps were inset under the skin of ridge. Pumps were loaded with test substances (Galp and Ala) and control substances (NaCl, DMSO and ACTH) at 0.5 nmol/100 g body weight/0.5 μL. The secretion of examined substances was 2.2 nmol/day. After the exposition time (2 days) animals were decapitated. The trunk blood was collected in the presence of EDTA (1 mg/mL), and plasma was separated and stored at −20°C until biochemical assays were performed (cf. the ELISA experiments). Hypothalami, pituitary glands and adrenal glands were collected to RNAlater and stored in −80°C for further qRT-PCR analysis. There were 5 groups with 8 animals in each group. As noted above, since Galp peptide was dissolved in 30 μL of 60% acetonitrile in water with DMSO, additional negative control by means of Galp solvent was used. Alarin peptide was dissolved in 0.9% saline.
RNA isolation
Total RNA was extracted from the obtained tissue (hypothalami, pituitary glands and adrenal glands) using GeneMATRIX Universal RNA Purification Kit (cat. No. E3598-02; EURx Ltd., Gdańsk, Poland). First, tissues were mechanically shredded with a homogenizer. Then, samples were centrifuged for 3 min at maximum speed (13,000 × g). The supernatant was collected and 350 μL of 70% of ethanol were added to every sample. The mixture was transferred to pure mini-columns and centrifuged for 1 min at 11,000 × g. Subsequently, 3 consecutive steps consisted in adding the appropriate rinsing buffer concentration to all samples. Each time, samples were centrifuged for 1 min at 11,000 × g. In the final stage, the mini-columns were placed in clean tubes and 30 μL of water were added. The amount of total RNA was determined using optical density (OD) at 260 nm, and its purity was estimated by 260/280 nm absorption ratio (higher than 1.8) (NanoDrop Spectrophotometer; Thermo Fisher Scientific, Waltham, USA). Samples were stored at −80°C for further qRT-PCR analysis.
Reverse transcription PCR (RT-PCR)
Reverse transcription was performed using Transcriptor High Fidelity cDNA Synthesis Kit (cat. No. 05081963001; Roche, Basel, Switzerland) at 42°C for 60 min (Thermocycler UNO II; Biometra; Analytik Jena GmbH, Jena, Germany). The primers were designed using Primer 3 software (Whitehead Institute for Biomedical Research, Cambridge, USA; Table 1). The primers were purchased from the Laboratory of DNA Sequencing and Oligonucleotide Synthesis, Institute of Biochemistry and Biophysics, Polish Academy of Sciences, Warszawa, Poland.
qRT-PCR analysis
Expression levels of selected genes (Table 1) were performed by means of qRT-PCR (7900HT Fast Real-Time PCR System; Applied Biosystems, Waltham, USA). Using the primers presented in Table 1, the SYBR-Green detection system was applied according to the specific protocol. Every 25 μL reaction mixture contained: 2 μL of template cDNA, 1 μL of every gene-specific primer (0.3 μM), 12.5 μL of 2× Maxima SYBR Green/ROX qPCRMaster mix (Thermo Fisher Scientific), and 8.5 μL of RNaze-free water. The qRT-PCR program included: 10-minute initial denaturation step to activate the Taq DNA Polymerase (95°C), followed by a 3-step amplification program: 1) denaturation at 95°C for 15 s; 2) annealing at 60°C for 30 s; and 3) extension at 72°C for 30 s. The specificity of reaction products was checked by determination of melting points (0.1°C/s transition rate).
ELISA – hormones level detection
Plasma was separated using centrifuge and the blood serum was stored at −20°C for enzyme-linked immunosorbent assay (ELISA) analysis. Galarin, Ala, ACTH, corticosterone, and aldosterone concentration were determined with the ELISA method performed in adherence to specific protocols. Galarin ELISA kit was obtained from MyBioSource, Inc. (Rat Galanin-like peptide (GALP) ELISA Kit, cat. No. MBS7216341; San Diego, USA). Alarin and ACTH ELISA kits were obtained from Phoenix Pharmaceuticals, Inc. (cat. No. EK-026-33 and cat. No. EK-001-21, respectively). Corticosterone and aldosterone ELISA kits were obtained from Demeditec Diagnostics GmbH (cat. No. DEV9922 and cat. No. DE5298, respectively; Kiel, Germany).
Statistical analyses
Statistical analyses of the data were performed using the Kruskal–Wallis test and Dunn’s post hoc test. Data are presented as medians, 1st and 3rd quartiles (boxes) and ranges (whiskers – minimum and maximum without outliers).
Results
qRT-PCR analysis
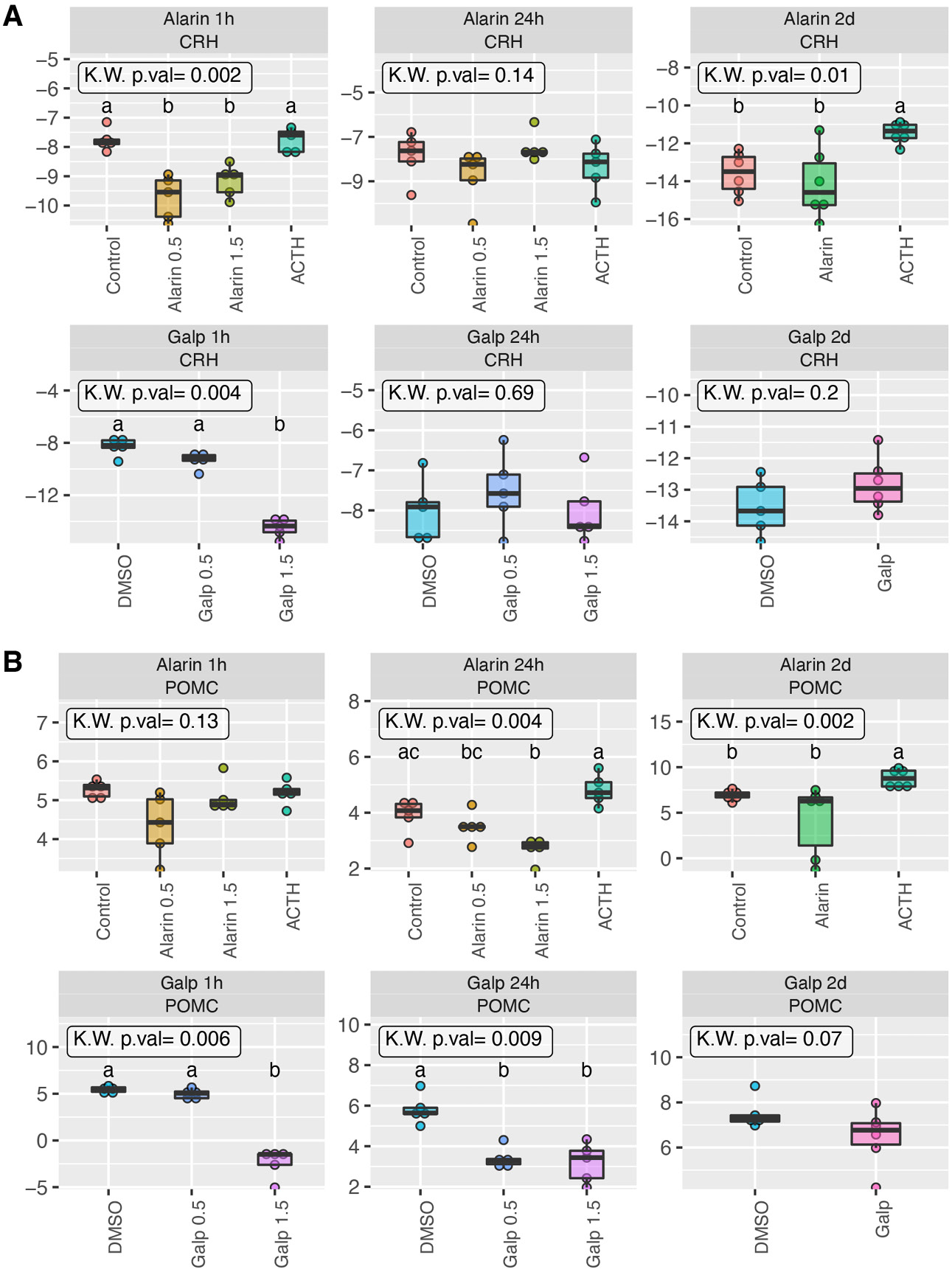
The expression analysis of Galp, Ala, Galr1, Galr2, and Galr3, CRH, and proopiomelanocortin genes was performed by means of qRT-PCR. In addition, the expression of Cyp11a1 (cholesterol desmolase), Cyp11b1 (11β-hydroxylase) and Cyp11b2 (aldosterone synthase) as steroidogenic enzymes in the adrenal glands was determined. The qRT-PCR graphs of 3 experiments were presented in Figure 1, Figure 2, Figure 3, Figure 4, Figure 5.
One-hour, 24-hour and 2-day exposure to the peptides in hypothalamus
Statistically significant changes of genes expression were observed mostly in hypothalamus and pituitary gland after 1 h of exposition to peptides (Figure 1, Figure 3).
In hypothalamus, after both doses of Ala peptide administration, the decrease of expression of mRNA Galp, Ala and their receptors was noted, compared with control and/or ACTH. Expression changes were visible especially after 1 h and 2 days of the peptide administration. However, after 24 h of Ala administration, the decrease of expression of Galr1 mRNA and the increase of expression of Galr3 mRNA was also noted (Figure 1).
On the other hand, there was a significant decrease in mRNA expression of Galp, Ala, Galr1, and Galr2 1 h after Galp 1.5 nmol peptide administration (Figure 1). In contrast, a decrease in mRNA expression of only Galp, Ala and Galr2 was observed after 24 h of Galp administration (both doses), whereas Galr1 and Galr3 mRNAs showed an increase in their expression.
After 2 days of experiment, there was a statistically significant increase in mRNA expression of Ala only (Figure 1). After ACTH administration, there was an increase in the expression of most of the mRNAs tested, especially 1 h and 2 days after peptide administration compared to control and test peptides groups (Figure 1). In the hypothalamus, under the influence of both Ala and Galp, there was also a decrease in CRH expression 1 h after the peptide administration (Figure 2A).
One-hour, 24-hour and 2-day exposure to the peptides in the pituitary gland
In the pituitary gland, a decrease in the expression of Galr2 and Galr3 mRNAs was observed 1 h after the administration of Ala peptide at a dose of 1.5 nmol, while at the same time, after the administration of Galp at a dose of 1.5 nmol, there was an increase in the expression of all tested mRNAs (Figure 3).
In contrast, after 24 h of the experiment, a decrease in Galp and Ala mRNA expression was noted after the administration of Ala peptide (both doses), and after the administration of Galp peptide (both doses), a decrease in expression of all mRNAs tested was observed.
Two days after the administration of the Ala peptide, there was an increase in the expression of Ala mRNA only, while 2 days after administration of Galp peptide, there was a statistically significant decrease in Galp mRNA only (Figure 3).
For POMC mRNA expression in the pituitary gland, a statistically significant decrease in its expression was noted 1 h after the Galp administration at a 1.5 nmol dose and 24 h after both Ala and Galp peptide administration (both doses) (Figure 2B).
One-hour, 24-hour and 2-day exposure
to the peptides in the adrenal gland
Changes in single mRNA expression were also noted in the adrenal glands, mainly under the influence of the Ala peptide (Figure 4). One hour after the administration of the Ala peptide (both doses), a statistically significant decrease in the expression of receptor 2 mRNA was observed, whereas after 24 h, an increase in the expression was noted for Galp and Galr3 mRNA (only under the influence of 0.5 nmol Ala).
A statistically significant decrease in the expression was also noted for Galr2 and Galr3 mRNA under the influence of Ala peptide 2 days after its administration.
Slight changes in expression were also noted for Cyp11a1, Cyp11b1 and Cyp11b2 mRNAs (Figure 5). Under the influence of Ala peptide, statistically significant expression changes were observed for Cyp11a1 and Cyp11b1 mRNAs, visible especially after 24 h and 2 days of the experiment, compared to control and ACTH.
When exposed to Galp peptide (at a dose of 1.5 nmol), there was an increase in the expression of aldosterone synthase mRNA, only 24 h after the peptide administration. No other changes in the expression of the tested steroidogenesis enzymes under the influence of Galp were noted.
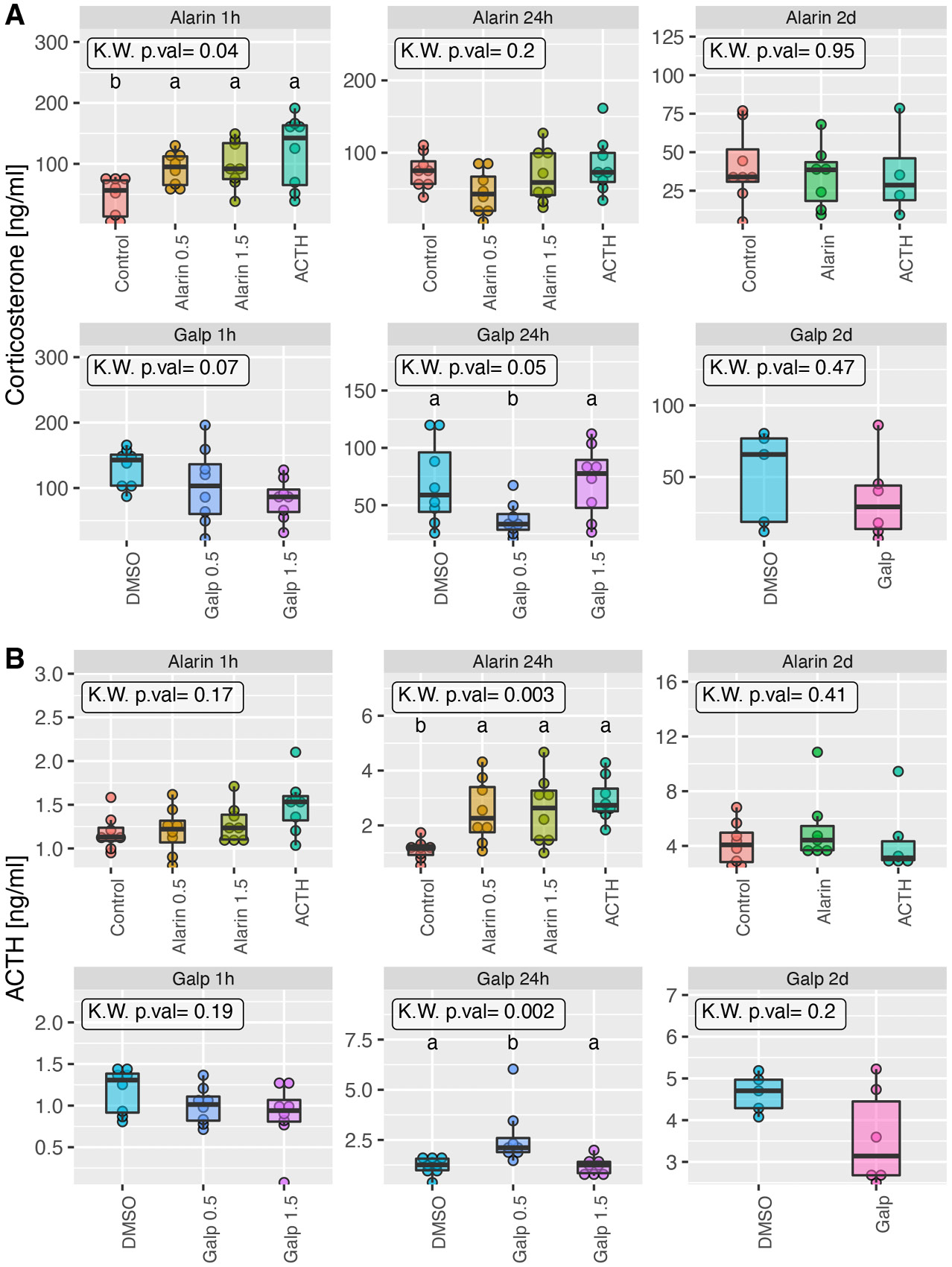
ELISA
After in vivo experiments, blood levels of several hormones were determined: ACTH, corticosterone, aldosterone, and the Galp and Ala peptides themselves. It was found that ip. administration of the Ala peptide at both doses (0.5 nmol and 1.5 nmol) led to a significant increase in blood ACTH concentration 24 h after peptide administration. A significant increase in blood ACTH concentration was also observed 24 h after the administration of Galp peptide at a dose of 0.5 nmol (Figure 6). There was also a significant increase in blood corticosterone concentration after the administration of Ala peptide, but only 1 h after administration. In contrast, the administration of Galp peptide at 1.5 nmol and 0.5 nmol led to a decrease in corticosterone levels in the blood of rats at 1 h and 24 h after the administration, respectively (Figure 6).
No effect of peptides on blood levels of either aldosterone, Galp or Ala has been demonstrated (data not shown).
Discussion
According to the available literature, little is known about the involvement of Galp and Ala in the regulation of adrenocortical hormone secretion and the interaction of HPA axis organs. In rat brain, Galp mRNA and protein have been detected in cell bodies located exclusively in the ARC of the hypothalamus and the medial ganglion.16, 28, 29, 30 It is well known that galanin, the main peptide of the Gal peptide family, is involved in the control of energy homeostasis. It plays an important role in the regulation of adrenal hormone secretion.11, 12, 31 As previously reported, Galp infusion has been shown to stimulate luteinizing hormone (LH) secretion in adult ovariectomized mice given estradiol. This effect was abolished by pretreatment with a gonadotropin-releasing hormone 1 (Gnrh1) antagonist.32 Furthermore, central administration of Galp increased serum LH levels in male diabetic rats.33 On the other hand, Boughton et al. found that acute intracerebroventricular (icv.) administration of Ala stimulates food intake and increases circulating LH levels in male rats.23
Our study, as well as several others, demonstrated the effects of Galp and Ala peptides on the expression of enzymes involved in steroidogenesis. Both Galp and Ala were shown to affect the expression of HPA axis mRNAs, including CRH, POMC, cholesterol desmolase, 11b-hydroxylase, and aldosterone synthase. As shown above, CRH expression was strongly downregulated in the hypothalamus after both doses of Ala (0.5 nmol and 1.5 nmol) and Galp (1.5 nmol), especially after 1 h of ip. exposure to the peptides. These results appear to confirm those previously obtained by Wang et al., according to whom icv. administration of 1.0 nmol Ala to the mouse brain resulted in a reduction of CRH mRNA levels in the hypothalamus.34 In this context, the decrease in POMC mRNA expression that we noted 24 h after administration of the tested peptides is also significant. The decrease in POMC expression coincides with a decrease in mRNA expression of steroidogenesis enzymes. Changes in mRNA expression of steroidogenesis enzymes in adrenal gland took place mainly under the influence of the Ala and ACTH peptides, especially after 24 h and prolonged exposure to the peptides. It can be observed that both Ala and Galp peptides affect the expression of major HPA mRNAs such as CRH and POMC, whereas for enzymes, changes in their mRNA expression are primarily seen under the influence of the Ala peptide, suggesting that it may have a more important function in regulating HPA axis function.
In 2005, Onaka et al. presented evidence that icv. injection of Galp significantly increased plasma ACTH concentrations.35 Similarly, our results showed an increase in blood ACTH levels after 24-hour ip. administration of Ala (both doses) and Galp (0.5 nmol dose only). In contrast, Wang et al. showed that an icv. injection of 1.0 nmol Ala resulted in a decrease in blood levels of CRH, ACTH and corticosterone.34 In our experiment, there was an increase in blood corticosterone levels 1 h after exposure to Ala (both doses). However, 24-hour ip. administration of both Ala and Galp reduced plasma corticosterone levels, but the result was significant only with Galp peptide administration. The effect of both Galp and Ala peptide was short-lived, as no changes in blood hormone concentrations were observed after longer (2-day) experiments. Equally important, neither Galp nor Ala caused statistically significant changes in blood aldosterone concentrations.
It would seem interesting to establish whether the peptides studied affect their own expression in HPA organs, and also whether and to what extent they affect the expression of their own receptors, the expression of which is known to have been recorded in all 3 organs of the HPA.7, 8 It has been clearly shown that both peptides influence the mRNA expression of Galp, Ala peptides and their receptors. However, it is primarily the Galp peptide that modulates their expression, especially in the hypothalamus and pituitary gland, where it decreases the expression of the mRNAs studied in a statistically significant manner. In the adrenal glands, where the mRNA expression of these peptides was already at a very low level, the mRNA expression of the Galp–Ala system was mainly influenced by the Ala peptide.
Limitations of the study
A limitation of the present study is the small size of the experimental groups, but these are standard group sizes in our in vivo animal experiments. Furthermore, the animal analyses were carried out according to the 3R principle (replacement, reduction, refinement) of animal studies to make them more humane. Thus, the present experiments were designed in such a way as to not involve too many animals. The experimental protocol could be strengthened by additional doses of administered peptides, e.g., a dose of 1.0 nmol or 2.0 nmol could be added, or the number of time intervals could be increased to test the effect of the tested peptides additionally after a longer time after injection (2 h or 6 h). However, such changes would also involve increasing the number of experimental animals, which could raise the objections from the local ethics committee.
Conclusions
There is evidence in the scientific literature for a link between both Galp and Ala peptides in the regulation of food intake, hormone release and control of metabolic processes.14, 19, 23, 24, 25, 31, 36, 37, 38 However, there is little data on the effects of the studied peptides on the blood levels of adrenal hormones and on the mRNA expression of steroidogenesis enzymes within the organs of the HPA axis, which makes it very difficult to discuss the results of the studies of interest. So far, our results coincide, at least in part, with literature data. Unfortunately, it is hard to find pure relationships between the action of the 2 peptides. However, it seems that the Galp peptide is the one that has a greater effect on mRNA expression of the Galp–Ala system in the hypothalamus and pituitary gland, while the Ala peptide affects mRNA expression of steroidogenesis enzymes in the adrenal glands and hormone levels in the blood. A number of further studies, e.g., adrenal cell culture and an analysis of signaling pathways in adrenocortical cells, are required to determine exactly how the Ala peptide is involved in adrenal gland function.
Additional data
Additional material showing the exact statistical analysis data has been deposited at https://zenodo.org/ (https://doi.org/10.5281/zenodo.6145227). The calculations are presented in separate files – as appropriate to the figures presented in the manuscript.
We generated the graphs in R, where it turned out that the ggplot2 library inserts minimum and maximum values into the boxplots as wishers, ignoring the outliers (https://r-coder.com/boxplot-r/). A data point considered as outlier is greater than Q3 + 1.5*IQR*IQR (right outlier), or is less than Q1 – 1.5*IQR*IQR (left outlier), but we still left such points on the graphs.