Advances in Clinical and Experimental Medicine
2022, vol. 31, nr 6, June, p. 597–606
doi: 10.17219/acem/146776
Publication type: meta-analysis
Language: English
License: Creative Commons Attribution 3.0 Unported (CC BY 3.0)
Download citation:
Cite as:
Nie W, Zhu L, Yan P, Sun J. Thyroid nodule ultrasound accuracy in predicting thyroid malignancy based on TIRADS system. Adv Clin Exp Med. 2022;31(6):597–606. doi:10.17219/acem/146776
Thyroid nodule ultrasound accuracy in predicting thyroid malignancy based on TIRADS system
1 Department of Ultrasound, Penglai People’s Hospital, Yantai, China
2 Deptartment of Endocrinology, Penglai People’s Hospital, Yantai, China
Abstract
Background. A frequent prevalence of thyroid nodules in patients prioritizes the need for an accurate method that characterizes them as benign or malignant. Fine-needle aspiration biopsy (FNAB) and thyroid ultrasonography (USG) are currently used for this purpose. However, since FNAB is complicated, time-consuming and expensive, thyroid USG, a fast and highly sensitive method, is preferably used. Although USG is reported as a suitable method for characterization of thyroid nodules, there are some contrasting studies available which report its limited use in the differentiation of benign and malignant thyroid nodules.
Objectives. This meta-analysis aims to assess the accuracy of ultrasound in predicting thyroid cancer in terms of sensitivity, specificity and diagnostic odds ratios (ORs) for positive and negative results.
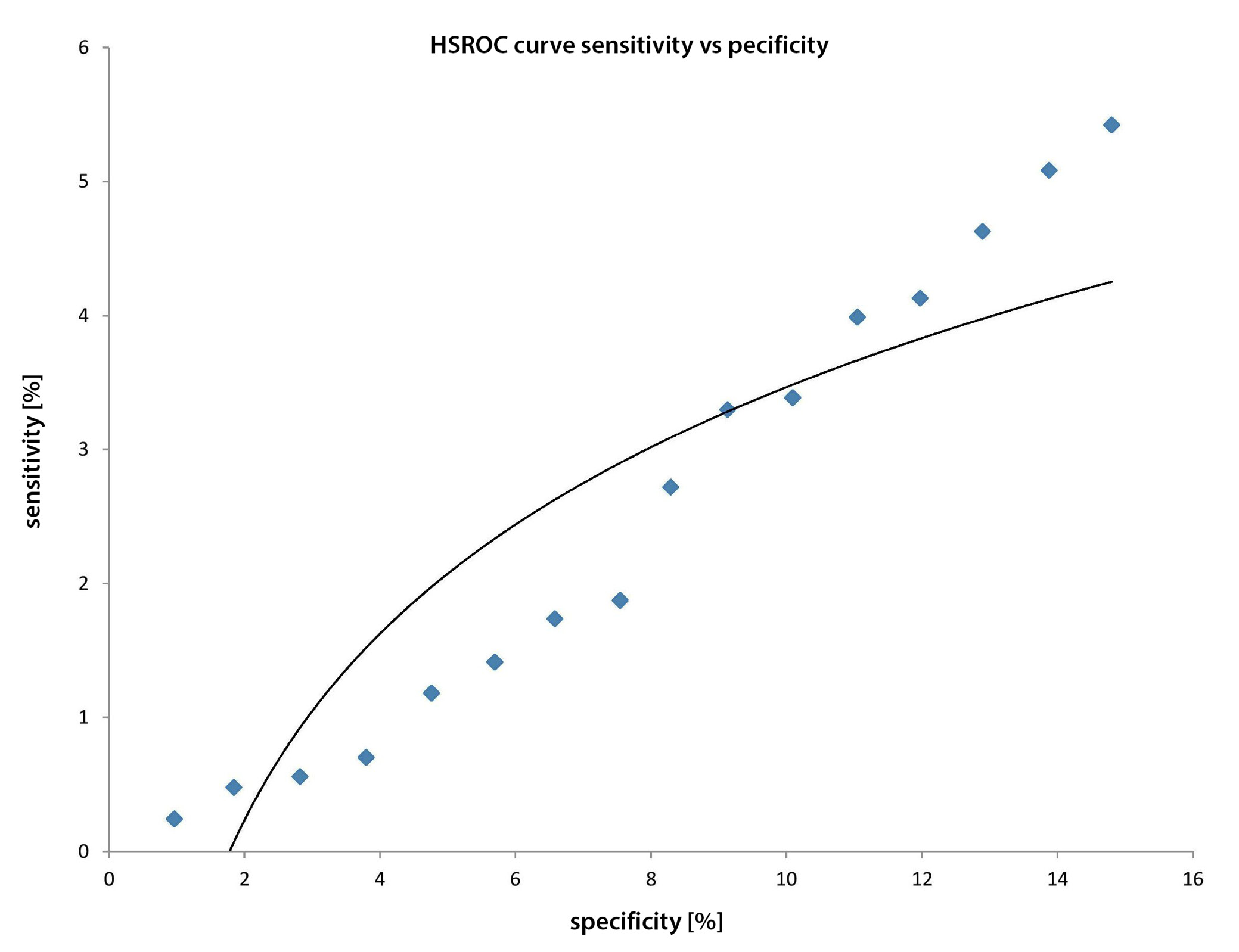
Material and Methods. Systematic and extensive literature search on the use of ultrasound (US) to predict thyroid cancer was conducted in the databases of Scopus, CINAHL (via EBSCO), MEDLINE (via PubMed), and Web of Science, covering the period from 2010 till 2021. The morphological features of thyroid nodules observed during the USG were analyzed based on Thyroid Imaging Reporting And Data System (TIRADS) guidelines. The accuracy of thyroid US was determined using parameters such as sensitivity, specificity, positive likelihood ratio (PLR), negative likelihood ratio (NLR), and diagnostic ORs. Moreover, the respective forest plot and hierarchical summary receiver operating characteristics (HSROC) curve were plotted.
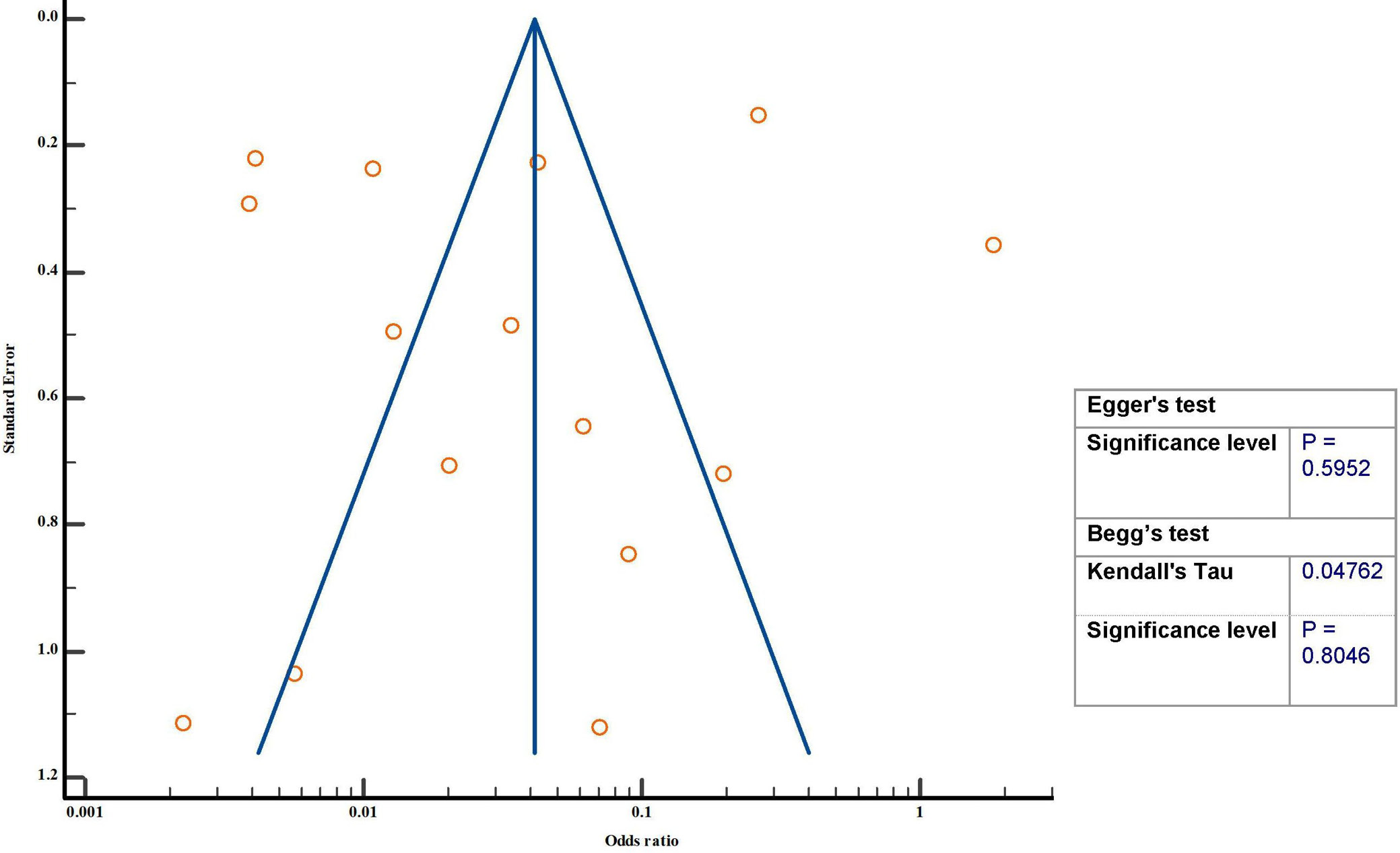
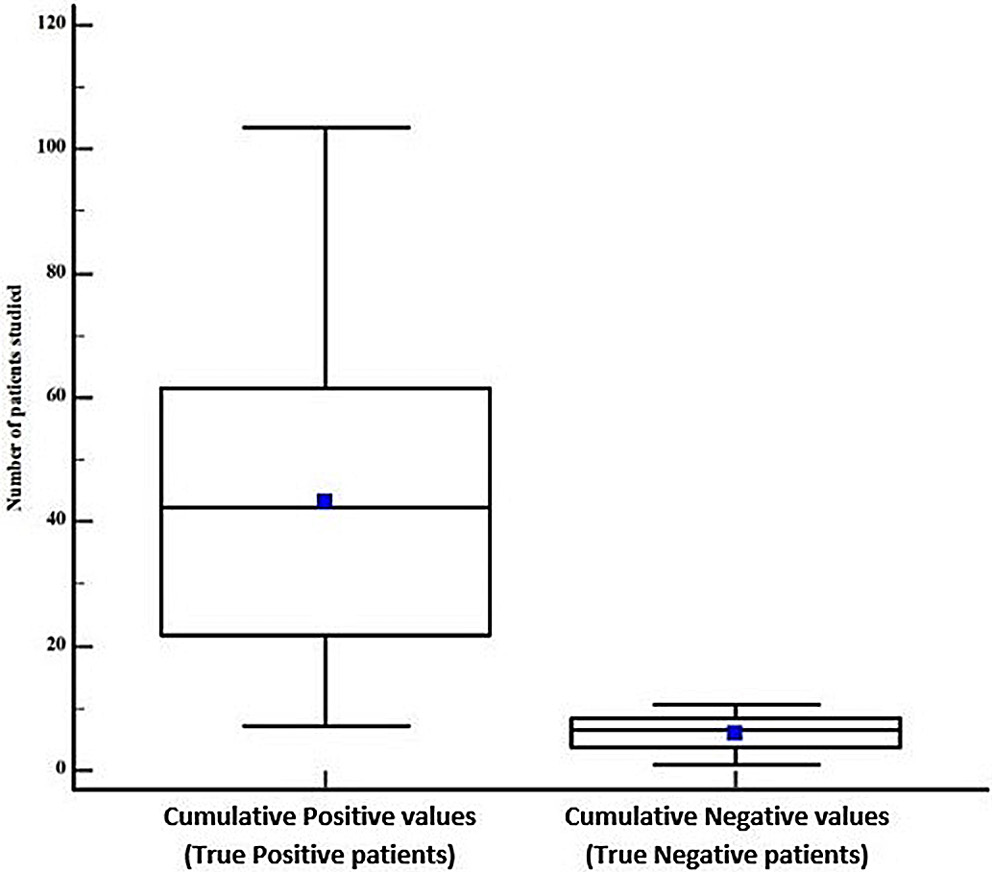
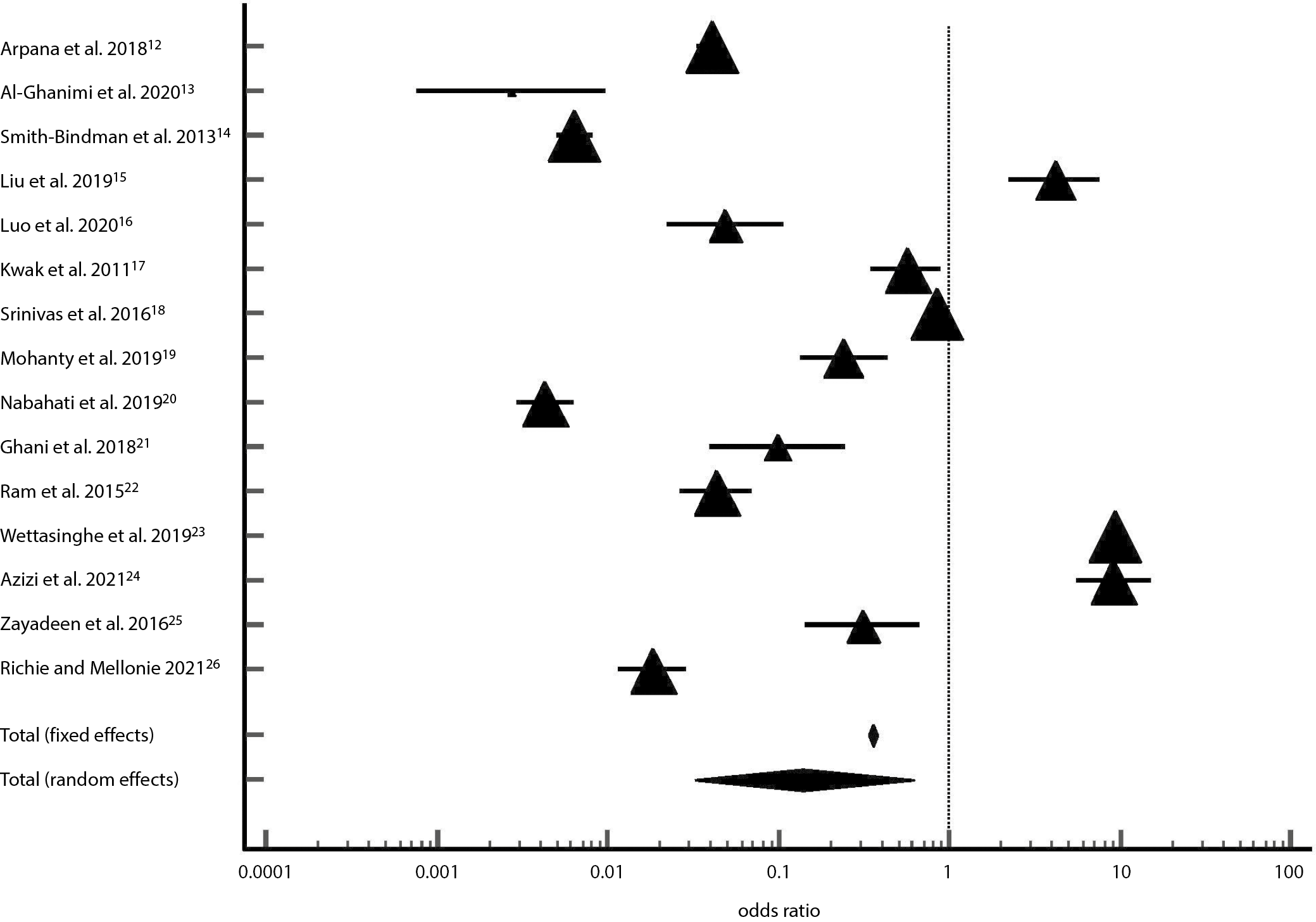
Results. A total of 2765 reference studies were examined, and among them, 15 relevant references were selected. The selected studies were heterogeneous and included retrospective and prospective studies. The risk of publication bias is low as the p-value for both Egger’s and Begg’s tests is >0.05. The overall sensitivity of 92.53% (95% confidence interval (95% CI): [84.55%; 96.33%]), specificity of 33.88% (95% CI: [23.16%; 45.53%]) and diagnostic OR of 12.36 (95% CI: [3.90%; 54.11%]) are achieved. These results were statistically significant with a p-value < 0.001 and are predictive of US accuracy in detecting cancer.
Conclusion. The present meta-analysis, on the basis of statistically significant results, demonstrated the high accuracy of thyroid ultrasound in detection of malignant nature of nodules in patients suspected with a worrisome thyroid nodule.
Key words
ultrasound, thyroid nodule, fine-needle aspiration biopsy (FNAB), thyroid imaging – reporting and data system (TIRADS), benign and malignant nodule
Tables
|
Study ID and year |
Study type |
Study duration |
Total sample size |
Age [years] |
Gender M/F |
Type of US probe |
|---|---|---|---|---|---|---|
|
Arpana et al. 201812 |
cross-sectional |
1 year |
85 |
14–70 |
15/70 |
NR |
|
Al-Ghanimi et al. 202013 |
retrospective |
2 years |
68 |
8–82 |
20/48 |
Esaote US machine (MyLab™ ClassC, Esaote, Genoa, Italy) and electronically focused near-field probes with a bandwidth of 7–12 MHz |
|
Smith-Bindman |
retrospective |
5 years |
11618 |
30–70 |
2277/9341 |
NR |
|
Liu et al. 201915 |
retrospective |
5 years |
1568 |
18–80 |
412/1156 |
IU22 device (Philips Medical Systems, Bothell, USA; 5–12 MHz linear probe) or the S3000 device (Siemens Medical Solutions, Mountain View, USA; 5–14 MHz linear probe) |
|
Luo et al. 202016 |
retrospective |
2 years |
296 |
30–50 |
54/168 |
The Mylab™ 90 (Esaote SpA, Genoa, Italy) ultrasound image system was used for US examination, the L522 probe (4–9 MHz; Esaote SpA) for CEUS and the L523 probe (7.5–13.0 MHz, Esaote SpA) for conventional gray-scale US, CDUS and ES. |
|
Kwak et al. 201117 |
retrospective |
8 months |
1638 |
11–81 |
265/1373 |
5–12 MHz linear-array transducer (iU22; Philips Medical Systems. |
|
Srinivas et al. 201618 |
prospective |
4 years |
365 |
18–68 |
22/334 |
GE VOLUSON 730 PRO machine (GE Healthcare, Milwaukee, USA) equipped with a 7.5–12 MHz high-frequency linear array transducer with color and power Doppler capability. |
|
Mohanty et al. 201919 |
prospective |
1 year |
50 |
40–50 |
10/40 |
GE Logic F8 ultrasound machine with a 6–12 MHz linear array transducer and Samsung HS70A ultrasound machine with 4–18 MHz linear array transducer (Samsung Neurologica Corp., Danvers, USA) |
|
Nabahati et al. 201920 |
cross-sectional |
2 years |
718 |
14–83 |
NR |
Samsung H60 ultrasound machine, with a 3–14 MHz linear array transducer (Samsung Neurologica Corp.) |
|
Ghani et al. 201821 |
retrospective |
2 years |
91 |
27–80 |
21/83 |
linear array transducer (5–12 MHz) on ultrasound scanners HD11/HD11 XE/iU22 (Phillips Medical Systems) or Toshiba Xario200 (Toshiba Corp., Tokyo, Japan) |
|
Ram et al. 201522 |
cross-sectional |
2 years |
101 |
15–73 |
20/81 |
High frequency linear probe with 7.5 MHz bandwidth (models Zario and Nemio; Toshiba Corp.) |
|
Wettasinghe et al. 201923 |
prospective |
1.5 years |
263 |
16–74 |
16/247 |
NR |
|
Azizi et al. 202124 |
prospective |
1 year |
355 |
40–50 |
45/310 |
virtual organ computer-aided analysis; (VOCAL; GE Healthcare) and a 3-D multi-planar display with rendering in HDLive and HDLive Silhouette (GE Healthcare). |
|
Zayadeen et al. 201625 |
retrospective |
3 years |
1466 |
11–96 |
265/1201 |
5–12 MHz linear probe (iU22, Philips Healthcare) or a 6–15-MHz linear probe (Logiq E9, GE Healthcare) |
|
Richie and Mellonie 202126 |
retrospective |
2 years |
226 |
18–62 |
39/187 |
NR |
|
Subgroup |
p-value |
|---|---|
|
Full texts compared to abstracts |
NA |
|
High compared to low risk of bias |
NA |
|
Prospective compared to retrospective studies |
0.024* |
|
Adults compared to mixed population |
0.924 |
|
Proportion of female participants |
0.05* |
|
Proportion of obese participants |
NA |
|
Type of ultrasound probe |
0.034* |
|
Ultrasonographer experience |
0.001* |
|
Clinical probability of TC |
0.001* |
|
Study ID and year |
Specificity [%] |
95% CI upper limit |
95% CI lower limit |
Sensitivity [%] |
95% CI upper limit |
95% CI lower limit |
|---|---|---|---|---|---|---|
|
Kwak et al. 201117 |
24.06 |
21.51 |
26.75 |
96.66 |
94.88 |
97.95 |
|
Smith-Bindman et al. 201314 |
23.61 |
18.83 |
28.95 |
87.94 |
83.56 |
91.50 |
|
Ram et al. 201522 |
8.06 |
2.67 |
17.83 |
97.50 |
86.84 |
99.94 |
|
Zayadeen et al. 201625 |
14.25 |
11.98 |
16.78 |
97.57 |
96.46 |
98.41 |
|
Srinivas et al. 201618 |
48.15 |
28.67 |
68.05 |
96.45 |
93.88 |
98.15 |
|
Ghani et al. 201821 |
23.08 |
11.13 |
39.33 |
93.62 |
82.46 |
98.66 |
|
Arpana et al. 201812 |
32.35 |
17.39 |
50.53 |
88.57 |
73.26 |
96.80 |
|
Wettasinghe et al. 201923 |
13.68 |
9.55 |
18.75 |
96.55 |
82.24 |
99.91 |
|
Luo et al. 202016 |
84.52 |
77.84 |
89.82 |
74.63 |
62.51 |
84.47 |
|
Liu et al. 201915 |
57.93 |
55.12 |
60.71 |
84.00 |
79.89 |
87.56 |
|
Nabahati et al. 201920 |
8.98 |
6.37 |
12.21 |
96.18 |
94.09 |
97.68 |
|
Mohanty et al. 201919 |
60.00 |
36.05 |
80.88 |
95.45 |
77.16 |
99.88 |
|
Azizi et al. 202124 |
14.08 |
10.21 |
18.74 |
92.65 |
83.67 |
97.57 |
|
Al-Ghanimi et al. 202013 |
50.00 |
15.70 |
84.30 |
91.67 |
81.61 |
97.24 |
|
Richie and Mellonie 202126 |
45.45 |
24.39 |
67.79 |
98.53 |
95.76 |
99.70 |
|
Study ID and year |
Benign nodule (simple cyst) |
Benign nodule (solid cyst) |
Malignant nodule (solid cyst) |
Malignant nodule (simple/mixed cyst) |
Diagnostic odds ratio |
95% CI upper limit |
95% CI lower limit |
|
|---|---|---|---|---|---|---|---|---|
|
Kwak et al. 201117 |
578.00 |
805.00 |
255.00 |
20.00 |
9.15 |
5.74 |
14.61 |
|
|
Bindmann et al. 201314 |
248.00 |
220.00 |
68.00 |
34.00 |
2.25 |
1.44 |
3.54 |
|
|
Ram et al. 201522 |
39.00 |
57.00 |
5.00 |
1.00 |
3.42 |
0.38 |
30.43 |
|
|
Zayadeen et al. 201625 |
1043.00 |
734.00 |
122.00 |
26.00 |
6.67 |
4.32 |
10.29 |
|
|
Srinivas et al. 201618 |
326.00 |
14.00 |
13.00 |
12.00 |
25.23 |
9.76 |
65.20 |
|
|
Ghani et al. 201721 |
44.00 |
30.00 |
9.00 |
3.00 |
4.40 |
1.09 |
17.60 |
|
|
Arpana et al. 201812 |
31.00 |
23.00 |
11.00 |
4.00 |
3.71 |
1.05 |
13.13 |
|
|
Wettasinghe et al. 201923 |
28.00 |
202.00 |
32.00 |
1.00 |
4.44 |
0.58 |
33.75 |
|
|
Luo et al. 202016 |
50.00 |
24.00 |
131.00 |
17.00 |
16.05 |
7.50 |
32.37 |
|
|
Liu et al. 201915 |
315.00 |
517.00 |
712.00 |
60.00 |
7.23 |
5.36 |
9.74 |
|
|
Nabahati et al. 201920 |
478.00 |
365.00 |
36.00 |
19.00 |
2.48 |
1.40 |
4.39 |
|
|
Mohanty et al. 201919 |
21.00 |
8.00 |
12.00 |
1.00 |
31.50 |
3.50 |
283.30 |
|
|
Azizi et al. 202124 |
63.00 |
238.00 |
39.00 |
5.00 |
2.06 |
0.78 |
5.45 |
|
|
Ghanimi et al. 202113 |
55.00 |
4.00 |
4.00 |
5.00 |
11.00 |
2.08 |
57.91 |
|
|
Richi et al. 202126 |
201.00 |
3.00 |
10.00 |
12.00 |
55.80 |
13.50 |
229.90 |
|
Figures

References (28)
- Chaturvedi R, Kumar A, Balasubramanian B, Sreehari S. A retrospective study correlating ultrasound based Thyroid Imaging Reporting and Data System (TIRADS) with Bethesda system for thyroid cytopathology in thyroid nodule risk stratification. NEMJ. 2021;2(2):121–128. doi:10.2174/0250688203666210111152307
- Chen H, Ye J, Song J, You Y, Chen W, Liu Y. Comparison of different ultrasound classification systems of thyroid nodules for identifying malignant potential: A cross-sectional study. Clinics (Sao Paulo). 2021;76:e2126. doi:10.6061/clinics/2021/e2126
- Hahn SY, Shin JH, Oh YL, Park KW. Ultrasound-guided core needle biopsy techniques for intermediate or low suspicion thyroid nodules: Which method is effective for diagnosis? Korean J Radiol. 2019;20(10): 1454–1461. doi:10.3348/kjr.2018.0841
- Al-Salam S, Sharma C, Abu Sa’a MT, et al. Ultrasound-guided fine needle aspiration cytology and ultrasound examination of thyroid nodules in the UAE: A comparison. PLoS One. 2021;16(4):e0247807. doi:10.1371/journal.pone.0247807
- Kim SC, Kim JH, Choi SH, et al. Off-site evaluation of three-dimensional ultrasound for the diagnosis of thyroid nodules: Comparison with two-dimensional ultrasound. Eur Radiol. 2016;26(10):3353–3360. doi:10.1007/s00330-015-4193-2
- Xie C, Cox P, Taylor N, LaPorte S. Ultrasonography of thyroid nodules: A pictorial review. Insights Imaging. 2016;7(1):77–86. doi:10.1007/s13244-015-0446-5
- Tessler FN, Middleton WD, Grant EG, et al. ACR Thyroid Imaging, Reporting and Data System (TI-RADS): White paper of the ACR TI-RADS committee. J Am Coll Radiol. 2017;14(5):587–595. doi:10.1016/j. jacr.2017.01.046
- Nam SJ, Kwak JY, Moon HJ, Yoon JH, Kim EK, Koo JS. Large (≥3cm) thyroid nodules with benign cytology: Can Thyroid Imaging Reporting and Data System (TIRADS) help predict false-negative cytology? PLoS One. 2017;12(10):e0186242. doi:10.1371/journal.pone.0186242
- Russ G, Bonnema SJ, Erdogan MF, Durante C, Ngu R, Leenhardt L. European Thyroid Association guidelines for ultrasound malignancy risk stratification of thyroid nodules in adults: The EU-TIRADS. Eur Thyroid J. 2017;6(5):225–237. doi:10.1159/000478927
- Trimboli P, Durante C. Ultrasound risk stratification systems for thyroid nodule: Between lights and shadows, we are moving towards a new era. Endocrine. 2020;69(1):1–4. doi:10.1007/s12020-020-02196-6
- Colakoglu B, Yildirim D, Alis D, et al. Elastography in distinguishing benign from malignant thyroid nodules. J Clin Imaging Sci. 2016;6:51. doi:10.4103/2156-7514.197074
- Arpana, Panta OB, Gurung G, Pradhan S. Ultrasound findings in thyroid nodules: A radio-cytopathologic correlation. J Med Ultrasound. 2018;26(2):90–93. doi:10.4103/JMU.JMU_7_17
- Al-Ghanimi IA, Al-Sharydah AM, Al-Mulhim S, et al. Diagnostic accuracy of ultrasonography in classifying thyroid nodules compared with fine-needle aspiration. Saudi J Med Med Sci. 2020;8(1):25–31. doi:10.4103/sjmms.sjmms_126_18
- Smith-Bindman R, Lebda P, Feldstein VA, et al. Risk of thyroid cancer based on thyroid ultrasound imaging characteristics: Results of a population-based study. JAMA Intern Med. 2013;173(19):1788–1796. doi:10.1001/jamainternmed.2013.9245
- Liu T, Guo Q, Lian C, et al. Automated detection and classification of thyroid nodules in ultrasound images using clinical-knowledge-guided convolutional neural networks. Med Image Anal. 2019;58:101555. doi:10.1016/j.media.2019.101555
- Luo W, Zhang Y, Yuan J, et al. Differential diagnosis of thyroid nodules through a combination of multiple ultrasonography techniques: A decision-tree model. Exp Ther Med. 2020;19(6):3675–3683. doi:10. 3892/etm.2020.8621
- Kwak JY, Han KH, Yoon JH, et al. Thyroid imaging reporting and data system for US features of nodules: A step in establishing better stratification of cancer risk. Radiology. 2011;260(3):892–899. doi:10.1148/radiol.11110206
- Srinivas MN, Amogh VN, Gautam MS, et al. A prospective study to evaluate the reliability of thyroid imaging reporting and data system in differentiation between benign and malignant thyroid lesions. J Clin Imaging Sci. 2016;6:5. doi:10.4103/2156-7514.177551
- Mohanty J, Sanket, Mishra P. Role of ACR-TIRADS in risk stratification of thyroid nodules. Int J Res Med Sci. 2019;7(4):1039–1043. doi:10.18203/2320-6012.ijrms20191076
- Nabahati M, Moazezi Z, Fartookzadeh S, Mehraeen R, Ghaemian N, Sharbatdaran M. The comparison of accuracy of ultrasonographic features versus ultrasound-guided fine-needle aspiration cytology in diagnosis of malignant thyroid nodules. J Ultrasound. 2019;22(3):315–321. doi:10.1007/s40477-019-00377-2
- Ghani FA, Nurismah MI, Husyairi H, Shahrun Niza AS, Radhika S. Reliability of the ultrasound classification system of thyroid nodules in predicting malignancy. Med J Malaysia. 2018;73(5):263–271. PMID:30350802.
- Ram N, Hafeez S, Qamar S, et al. Diagnostic validity of ultrasonography in thyroid nodules. J Pak Med Assoc. 2015;65(8):875–878. PMID:26228335.
- Wettasinghe MC, Rosairo S, Ratnatunga N, Wickramasinghe ND. Diagnostic accuracy of ultrasound characteristics in the identification of malignant thyroid nodules. BMC Res Notes. 2019;12(1):193. doi:10.1186/s13104-019-4235-y
- Azizi G, Faust K, Ogden L, et al. 3-D ultrasound and thyroid cancer diagnosis: A prospective study. Ultrasound Med Biol. 2021;47(5):1299–1309. doi:10.1016/j.ultrasmedbio.2021.01.010
- Zayadeen AR, Abu-Yousef M, Berbaum K. Retrospective evaluation of ultrasound features of thyroid nodules to assess malignancy risk: A step toward TIRADS. AJR Am J Roentgenol. 2016;207(3):460–469. doi:10.2214/AJR.15.15121
- Richie AJ, Mellonie P. Accuracy of thyroid imaging and reporting data systems in risk stratification of thyroid nodules: A retrospective observational study. Int J Anat Radio Surg. 2021;10(1):58–61. doi:10.7860/IJARS/2021/47306:2627
- Latif MA, El Rakhawy MM, Saleh MF. Diagnostic accuracy of B-mode ultrasound, ultrasound elastography and diffusion weighted MRI in differentiation of thyroid nodules (prospective study). Egypt J Radiol Nucl Med. 2021;52:256. doi:10.1186/s43055-021-00640-9
- Jiang D, Zang Y, Jiang D, Zhang X, Zhao C. Value of rapid on-site evaluation for ultrasound-guided thyroid fine needle aspiration. J Int Med Res. 2019;47(2):626–634. doi:10.1177/0300060518807060
















