Abstract
Anticoagulant-related nephropathy (ARN) is a novel and not well-studied cause of acute kidney injury (AKI). The prevalence of ARN varies significantly between studies and is estimated at 20% in patients treated with warfarin. Patients with ARN have a significantly higher mortality risk and an increased risk of chronic kidney disease (CKD). Unexplained AKI with hematuria are clinical manifestations of ARN. In most cases, ARN is diagnosed within the first 2 months of anticoagulant therapy, but later ARN occurrence is possible. Among the studied anticoagulants, most data concern warfarin toxicity, whereas cases of ARN caused by direct oral anticoagulants (DOACs) have also been presented. Tubular obstruction by red blood cell casts or hemoglobin and iron tubular toxicity are the postulated mechanisms of ARN. On the molecular level, the inhibition of thrombin and protease-activated receptor-1 (PAR-1), leading to endothelial susceptibility to damage or abnormal protein C endothelial signaling, is suggested to contribute to ARN. Older age, impaired kidney function, hypertension, and diabetes mellitus are the main risk factors for ARN, but their significance may differ between anticoagulants. From therapeutic options, the withdrawal of the anticoagulant and the administration of its antidote, as well as corticosteroids or N-acetylcysteine, are proposed. Since the number of patients with kidney diseases on anticoagulants increases, and DOACs are starting to be more useful in this group of patients, we aim to summarize the pathogenesis, clinical picture and possible ways of treatment of DOAC-induced ARN.
Key words: acute kidney injury, warfarin, anticoagulants, dabigatran, rivaroxaban
Introduction
Anticoagulants are one of the most commonly prescribed drugs worldwide and their administration may increase in the following years due to their suboptimal use.1 Direct oral anticoagulants (DOACs), previously called novel oral anticoagulants (NOACs), have been repeatedly shown to be not inferior in comparison with vitamin K antagonists (VKAs), in the prevention and treatment of venous thromboembolic disease, as well as in the prevention of ischemic stroke in patients with non-valvular atrial fibrillation, without increasing the risk of life-threatening bleeding.2 People with chronic kidney disease (CKD) are a special group of patients, with many comorbidities on the one hand, and with different pharmacokinetics of most drugs on the other.3 In an animal model of CKD, a lower transcription and a lower concentration of cytochrome P-450 enzymes were demonstrated, which can be responsible for a lower drug metabolism and an increased risk of overdose.4 The inhibitory effect of uremic toxins on cytochrome P-450 activity was also postulated.5 In a recently published meta-analysis by Randhawa et al. regarding patients with end-stage renal disease (ESRD), warfarin was reported not to increase the risk of ischemic stroke, major bleeding and overall mortality, contrary to a higher risk of hemorrhagic stroke, showing an increased risk of complications after the use of VKAs in the CKD population.6 Unfortunately, the lack of results of head-to-head trials comparing anticoagulants efficacy and safety, as well as the lack of data from randomized controlled trials (RCTs) on patients with estimated glomerular filtration rate (eGFR) lower than 30 mL/min taking DOACs, limit our knowledge about the appropriate anticoagulation in CKD patients. To the well-known limitations of anticoagulants, like drug and food interactions, changes in their pharmacokinetic profile according to the kidney function and the risk of vascular calcification, should be added another, recently described complication, called anticoagulant related nephropathy (ARN), primarily observed after the administration of VKAs. Since ARN can affect patients with both normal and impaired kidney function, it can possibly be caused by every anticoagulant and may lead to ESRD, this novel nephropathy will gain increasing significance.
Objectives
In this narrative review, we aimed to summarize our knowledge about DOAC-related ARN – its prevalence, risk factors, clinical manifestation, the methods of treatment, and its impact on renal outcome and survival.
Definition
Primarily, ARN was defined as an unexplained kidney injury observed within 1 week of international normalized ratio (INR) greater than 3.0 in patients on anticoagulants.7 After longer observation, the definition of ARN was modified as acute kidney injury (AKI) of unknown origin (a rise in serum creatinine level by more than 0.3 mg/dL) in a patient receiving anticoagulants, even without any signs of coagulopathy and active bleeding.8 What is interesting, except from oral anticoagulants, some researchers add antiplatelet drugs (dipyridamole or drugs used in dual antiplatelet therapy) and other anticoagulants, like low-molecular-weight heparin or unfractionated heparin, to the list of drugs potentially involved in the pathogenesis of ARN.9, 10, 11
Anticoagulant-related nephropathy was first described by Brodsky et al. in 9 patients with AKI, hematuria and warfarin overdose, so the primary name of this phenomenon was warfarin-related nephropathy.12 Each analyzed patient had an elevated INR on admission, with a mean value of 4.4 ±0.7, and a high serum creatinine level (4.3 ±0.8 mg/dL). Kidney biopsy results revealed acute tubular injury and glomerular hemorrhage, with red blood cells in Bowman’s space and plenty of red blood cell casts occluding the tubules.12 Unfortunately, only 3 out of 9 patients recovered from AKI, giving the first evidence of irreversibility of ARN in some patients.12 Similarly, Mikič et al. showed that dabigatran-related ARN could be characterized by occlusive intratubular red blood cell casts, diffuse acute tubular injury and diffuse interstitial edema.10 Interestingly, the red blood casts obstructing the renal tubules did not contain Tamm–Horsfall protein and were present more often in the distal tubules, and therefore in the renal medulla.10 It should be noted that kidney biopsy results in the suspicion of ARN are scarce, since coagulation abnormalities significantly limit the number of performed biopsies. However, kidney biopsy may reveal crucial information about the severity of kidney damage, as well as other pathologies related to abnormal kidney function, like immunoglobulin A (IgA) nephropathy or focal segmental glomerulosclerosis. Based on the provided results, a decision can also be made to withdraw particular anticoagulant or switch to another drug, potentially less related with ARN occurrence. The role of kidney biopsy in the diagnosis and prognosis of ARN is still underestimated and needs to be evaluated in future studies.
Epidemiology
Although the exact frequency of ARN is not known due to the novelty of the disease and the lack of kidney biopsy results, Brodsky et al. in a retrospective analysis of 103 patients estimated the prevalence of ARN after the administration of warfarin at 37%.13 In a further retrospective study in 15,258 patients taking warfarin, it was reported that ARN occurred in 20.5% of the entire cohort, 33% of the CKD cohort and 16.5% of the non-CKD cohort.14 Data about DOAC-related ARN prevalence are often controversial, indicating both nephroprotective and toxic effects of DOACs. However, in a large study by Marcelino et al. analyzing data from 134 national registries, dabigatran was associated with 4.6% of renal side effects, whereas other DOACs – rivaroxaban, apixaban and edoxaban – were shown to be related with 3.5%, 2.0% and 1.7% of renal side effects, respectively.15 What is interesting, the Apixaban for Reduction in Stroke and Other Thromboembolic Events in Atrial Fibrillation RCT (ARISTOTLE; https://clinicaltrials.gov/, NCT00412984) showed much higher incidence of kidney damage after apixaban, reaching 13.6% of the analyzed population, despite lower incidence of major bleeding and lower mortality.16 Due to the lack of results from other clinical trials (Renal Hemodialysis Patients Allocated Apixaban versus Warfarin in Atrial Fibrillation trial (RENAL-AF; https://clinicaltrials.gov/, NCT02942407) was prematurely terminated because of no funding and inconclusive results)6 and possibly much higher incidence of ARN than expected, new ongoing trials regarding the safety of dabigatran (Dabigatran vs Warfarin in AF Patients with T2DM and CKD (RE-ELECT; https://clinicaltrials.gov/, NCT03789695)) and apixaban (Strategies for the Management of Atrial Fibrillation in Patients Receiving Dialysis (SAFE-D; https://clinicaltrials.gov/, NCT03987711), Compare Apixaban and Vitamin-K Antagonists in Patients with Atrial Fibrillation (AF) and End-Stage Kidney Disease (ESKD) (AXADIA; https://clinicaltrials.gov/, NCT02933697)) in CKD patients hopefully will provide reliable data about the incidence of ARN after the administration of DOACs.
Pathogenesis
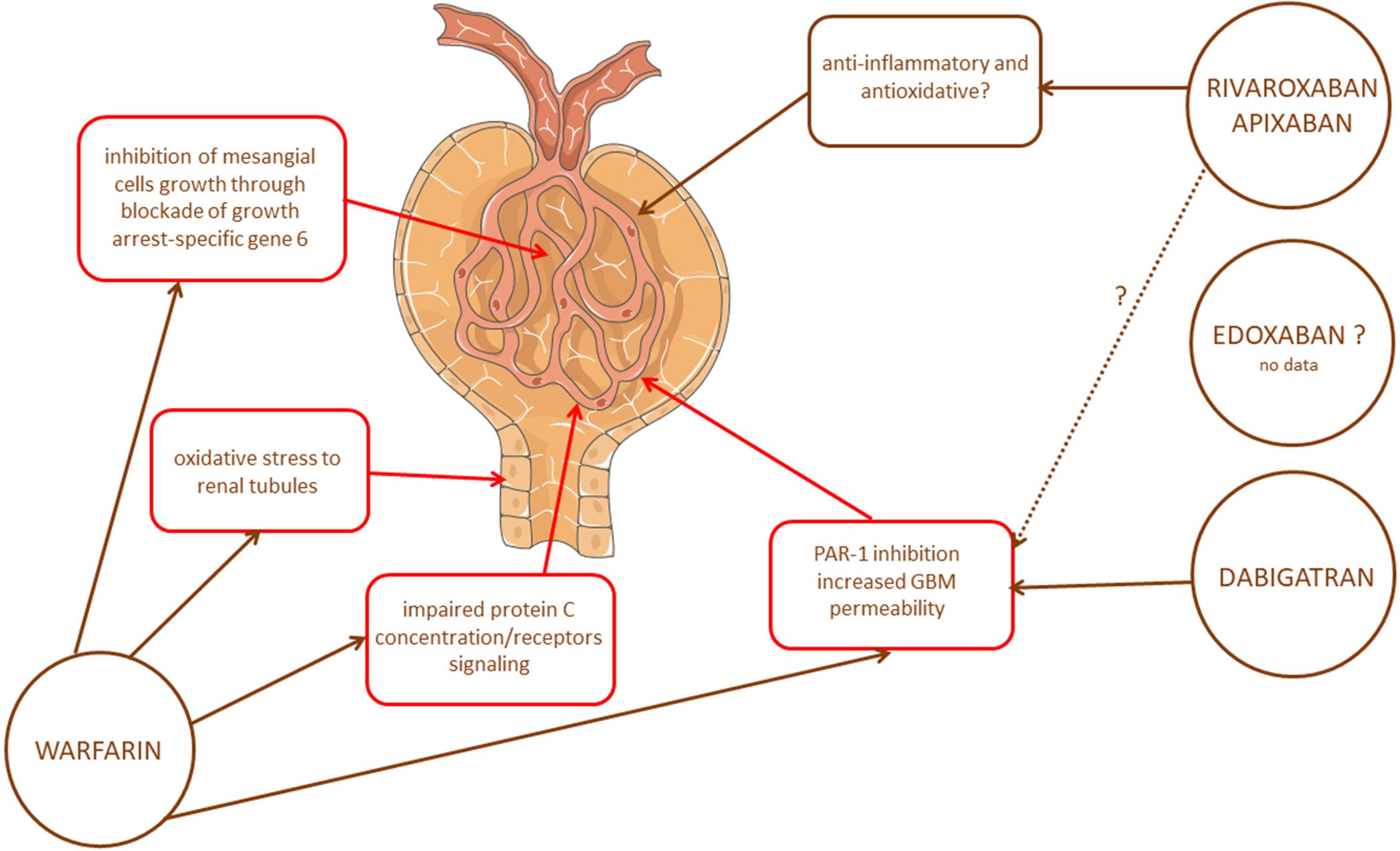
The mechanism of ARN is not well understood (Figure 1), since renal dysfunction cannot be explicitly treated as a result of the blockade of the renal tubules by red blood cells.17 Brodsky raised concerns about predicting kidney damage in ARN patients, in whom the risk of AKI cannot be directly related to INR and warfarin overdose is not sufficient to cause ARN.9 Ware et al. reported in 5/6 nephrectomy rats receiving warfarin increased oxidative stress in the kidney as compared to control, with the lack of iron in tubular epithelial cells and no beneficial effect of an iron chelator, deferoxamine, on the kidney function.18 Free hemoglobin was suggested to affect tubular epithelial cells through the production of reactive oxygen species (ROS) and lipid peroxidation,19 whereas after intracellular incorporation – to activate caspases and apoptosis.20 Later, it was postulated that heme released from hemoglobin caused further cellular damage by activating pro-inflammatory pathways.21 The activation of endothelial cells by heme to express adhesion molecules and complement the receptors, promoting the activation of the alternative complement pathway, was also suggested.22 Mitochondrial dysfunction after the intracellular incorporation of heme was postulated as well.21 Of note, patients with CKD are known to have lower activity of antioxidant enzymes, like glutathione peroxidase and catalase, which makes them especially vulnerable to warfarin toxicity.8 Other potential mechanisms are suspected in the pathogenesis of ARN, like atheroembolism, allergic interstitial nephritis, the apoptosis of glomerular endothelial cells, and the direct toxic effect of warfarin on the glomeruli.8, 23 It was suggested that warfarin might affect glomerular hemodynamics and the proliferation of mesangial cells through interfering with the process of activation of growth arrest-specific gene 6, responsible for the production of vitamin K-dependent autocrine growth factor for mesangial cells.24 Interestingly, the results of animal studies shed light on the pathogenesis of ARN, postulating that ARN is due to a diminished thrombin activity. Protease-activated receptor-1 (PAR-1) is known to take part in controlling endothelial cell functions, vascular permeability and leukocyte migration and adhesion.25, 26 The activation of PAR-1 has been shown to control endothelial monolayer integrity.27 In an animal model of CKD, Brodsky observed that the selective inhibition of PAR-1 by SCH79797 resulted in the elevation of serum creatinine, hematuria and the formation of red blood cell casts, effects that were more pronounced in 5/6 nephrectomy rats.9 This observation confirmed the theory of kidney damage caused by the direct inhibition of thrombin. A reduction in protein C concentration and impaired endothelial protein C receptor signaling are other possible mechanisms related to the pathogenesis of ARN.28, 29
Since its introduction onto the market in 2010, dabigatran has been the most extensively studied DOAC in relation to ARN.30 However, the available data remain inconclusive and do not clearly state whether dabigatran leads to kidney function impairment. As dabigatran in its unchanged form in over 80% is excreted through the kidneys, patients with impaired kidney function are already in danger of hemorrhagic complications and further kidney injury.30 Similar to animal studies with VKAs, Medipally et al. observed acute tubular epithelial cell injury, red blood cell casts in the tubules and an increase in blood pressure in 5/6 nephrectomy rats treated with dabigatran.31 Additionally, the modification of the eGFR by angiotensin-converting enzyme inhibitor, enalapril, seemed not to have any effect on dabigatran-induced ARN in that study.31 Importantly, Ryan et al. observed ARN not only in 5/6 nephrectomy rats, but also in control animals receiving dabigatran in a dose-dependent manner,32 differently than in animal studies with VKAs. Despite histopathological changes comparable to those observed in 5/6 nephrectomy rats after the administration of VKAs, the effect of a PAR-1 antagonist, SCH79797, was lesser than that of dabigatran, indicating that the inhibition of PAR-1 only partially explains the mechanism of ARN observed after this DOAC (Figure 1).
Adding to previous reports, more studies are needed to analyze the benefits and risks related with the inhibition of PAR-1 by dabigatran. In an anti-glomerular basement membrane (GBM) glomerulonephritis animal model, PAR-1−/− mice presented less kidney damage and fewer crescents, fibrin deposits and macrophage infiltrates.33 In a recently published study, dabigatran, through the inhibition of PAR-1, was shown to ameliorate tubulointerstitial fibrosis in unilateral ureteral obstruction-induced renal injury.34 The antiplatelet effect of dabigatran has also been postulated based on the effect on PAR-1 receptors,35 which in turn may increase the risk of bleeding and ARN. Interestingly, in an RCT performed by Correa et al., a PAR-1 inhibitor, vorapaxar, was shown to be safe for CKD patients.36
What is worth mentioning, the anti-inflammatory effect of rivaroxaban postulated by Terry et al.37 can be partly responsible for a lower incidence of ARN after this DOAC. It was demonstrated that rivaroxaban reduced cell proliferation, plasma monocyte chemotactic protein-1 and matrix metalloproteinase-9 (MMP-9) levels, which led to the improvement in the patency of central venous catheters in mice.37 Similarly, in preliminary studies, it was recognized that another factor X inhibitor, apixaban, reduced oxidative stress in mesangial cells through the reduction of expression and transcription of monocyte chemotactic protein-1 and intercellular adhesion molecule-1,38 which in turn might be related to a lower kidney injury level. Further studies in humans are needed to confirm this observation.
Additionally, Ware et al. reported an increase in blood pressure in control and 5/6 nephrectomy rats after the administration of warfarin and dabigatran.39 Both anticoagulants raised systolic blood pressure in the animals in a dose-dependent manner. Vitamin K prevented a blood pressure increase in warfarin-treated animals, whereas N-acetylcysteine only delayed it. A PAR-1 inhibitor, SHC79797, had similar effects to those of anticoagulants. What should be emphasized, the rise in blood pressure was independent of the kidney function, suggesting a direct drug effect.39
Risk factors
Based on clinical observations, it was shown that ARN occurred more often in patients with preexisting GBM abnormalities, like thin GBM disease40 or thick GBM in a patient with inactive systemic lupus erythematosus.41 In a cohort of Slovenian patients, Mikič et al. showed that most patients with ARN had underlying IgA nephropathy; however, the degree of histopathological disarrangement was not proportional to the level of kidney damage.10 Other diseases, like diabetic nephropathy, focal segmental glomerulosclerosis, postinfectious nephritis, chronic interstitial nephritis, nephrosclerosis, and vasculitis were also related to a higher risk of ARN.10 Brodsky et al., after analyzing the data of 4006 patients, reported that the strongest ARN risk factors were CKD, diabetes mellitus, heart failure, and hypertension.42 The presence of an arteriovenous fistula in kidney allograft recipients was also suggested to increase the susceptibility to ARN, due to local hemodynamic changes.43 Among the available medications, acetylsalicylic acid and drugs resulting in higher intraglomerular pressure, like directly acting smooth muscle relaxants and dihydropyridine calcium channel blockers, were recognized to increase the risk of ARN, indicating that glomerular hemorrhage and tubular obstruction are the 2 crucial mechanisms involved in kidney damage.14 Mitsuboshi et al. suggested that risk factors for ARN may differ between anticoagulants, with body weight higher than 80 kg and the use of dabigatran as the strongest predictors of kidney damage after DOACs, but they should be clarified in future studies.44
Clinical course
In most cases, ARN is presented as AKI in patients taking anticoagulants, after excluding other possible causes of kidney function impairment. It has been recognized that the highest risk of ARN is within the first 2 months after starting anticoagulant therapy45; however, it may vary between 1 week to 1 year after introducing DOAC (Table 1). In some cases of ARN, kidney replacement therapy may be needed due to severe kidney damage, whereas restoring the kidney function is not always observed.
Besides kidney damage, a hypertensive effect of anticoagulants should be mentioned. Human studies are often inconclusive, linking an increase in blood pressure with vascular calcification after the administration of warfarin,60 but this direct hypertensive effect should be taken into account while administering anticoagulants due to the possible increase of the cardiovascular risk.61
Therapeutic approach
The management of ARN depends on the anticoagulant responsible for kidney damage. The treatment of VKA-related ARN includes careful drug dosing, the regular monitoring of coagulation parameters, avoiding drug interactions, and the discontinuation of the use of VKAs in severe cases. The beneficial effect of N-acetylcysteine, prednisolone62 and vitamin K63 on kidney function improvement was observed, but it needs to be confirmed in future studies. Lowering the anticoagulation level or the temporal discontinuation of DOACs seem to be reasonable in patients with mild clinical manifestations of ARN, but may be insufficient in severe cases. It was shown that both hemodialysis procedures and the administration of methylprednisolone can be useful in the treatment of dabigatran-induced ARN.10 Interestingly, Alsamarrai et al. reported a case of ARN treatment with idarucizumab, a humanized monoclonal antibody, reversing the anticoagulant effect of dabigatran.53 Before the introduction of idarucizumab in 2015, hemodialysis was the only available specific therapy in dabigatran overload due to a low molecular weight of the drug and weak protein binding.64 In a recently published study, Galassi et al. proposed an algorithm of dabigatran overdose management in patients with eGFR lower than 30 mL/min.52 According to this study, idarucizumab should be considered in the case of major bleeding, surgical urgencies or in patients with a high risk of complications during hemodialysis catheter implantation, whereas hemodialysis can be performed in the presence of severe AKI and related complications.52 However, patients need to be carefully monitored due to the possible rebound increase in dabigatran level in AKI.65 Data about the specific treatment of ARN caused by rivaroxaban, apixaban and edoxaban are not available.
Effect on renal outcome and survival
Contrary to studies by Brodsky et al., confirming the impact of VKAs on irreversible kidney damage,12, 13, 14 the data concerning the effect of DOACs on patients’ outcome is scarce, since people receiving DOACs require less monitoring, fewer kidney biopsies are performed in patients with abnormal coagulation, and impaired kidney function limits the use of this group of anticoagulants. Additionally, the results of the available studies are often ambiguous, limiting our knowledge about DOAC-related ARN.
In a retrospective study, patients receiving dabigatran were shown to have ARN less frequently when compared to patients on VKAs.66 A similar effect was observed in patients with normal kidney function (hazard ratio (HR) 0.62; 95% confidence interval (95% CI): [0.49; 0.77]; p < 0.001), as well as in patients with CKD (HR 0.56; 95% CI: [0.46; 0.69]; p < 0.001), independently of the dose of dabigatran.66 In a nationwide retrospective cohort study by Chan et al., dabigatran again was associated with a lower risk of ARN in comparison with warfarin (HR 0.68; 95% CI: [0.64; 0.74] in the normal kidney function group, and HR 0.54, 95% CI: [0.49; 0.59] in the CKD group).66, 67 Unfortunately, since dabigatran is a thrombin inhibitor, it also blocks PAR-126 and may cause ARN in selected patients according to the available case reports (Table 1). Mikič et al. presented a case of an 82-year-old woman with a history of arterial hypertension, diabetes mellitus type 2, hypothyroidism, cognitive decline, and CKD, who developed AKI after the introduction of dabigatran.10 Due to severe kidney function impairment, hemodialysis procedures were started, and ARN together with IgA nephropathy were confirmed through kidney biopsy. Interestingly, the authors reported that the kidney function improved and hemodialysis treatment was stopped after the administration of methylprednisolone.10
Based on the data obtained by Mikič et al., it was surprisingly suggested that the renal outcome was slightly better in patients treated with VKAs than in those on DOACs (dabigatran or rivaroxaban).10 Similar observations were made by Marcelino et al., who analyzed data from the World Health Organization (WHO) pharmacovigilance program in the VigiAccess™ database.15 The authors showed that the annual risk of renal side effects was about 2 times higher for DOAC-treated patients as compared to patients receiving VKAs, and AKI was the most commonly reported clinical finding.15 In the available studies, most patients with ARN did not have a CKD history, had variable renal outcomes, and quite often required hemodialysis procedures; however, in most cases, kidney function improvement was observed (Table 1).
Rivaroxaban, a factor X inhibitor, is a relatively novel anticoagulant, so data about the effect of rivaroxaban on the kidney function are very limited and inconclusive. In a study by Chan et al., rivaroxaban compared to warfarin was reported to decrease the incidence of AKI in patients with normal kidney function (HR 0.73; 95% CI: [0.68; 0.79]), as well as in CKD patients (HR 0.53; 95% CI: [0.49; 0.58]).67 Adding to that, Zhang et al., in a pooled analysis of RCTs and observational studies, showed that rivaroxaban was associated with a low risk of kidney injury (HR 0.66; 95% CI: [0.55; 0.77]).68 The same conclusions were presented by Mitsuboshi et al. after the analysis of the Japanese adverse drug event report database of kidney injury cases.44
On the other hand, Marcelino et al. showed a 3.5% annual risk of renal side effects in patients receiving rivaroxaban.15 Since ARN after rivaroxaban was presented in few case reports with variable kidney outcomes (Table 1), more clinical studies should be performed to evaluate the exact role of rivaroxaban in the pathogenesis of ARN.
The same conflicting results about apixaban in relation to ARN came from other available studies. In a retrospective cohort study, apixaban was shown to decrease the risk of AKI when compared to warfarin in patients with preserved (HR 0.65; 95% CI: [0.60; 0.72]) and impaired kidney function (HR 0.50; 95% CI: [0.45; 0.56]).67
However, data from the ARISTOTLE RCT showed the worsening of GFR by more than 20% in 13.6% of patients receiving apixaban, which was associated with older age and cardiovascular comorbidities.16 As more ARN cases after the administration of apixaban were reported (Table 1), a possible impact of apixaban on the kidney function should be taken into account.
Edoxaban as the newest DOAC on the market was not well studied with regard to ARN. In recently published studies, Mitsuboshi et al.,44 as well as Zhang et al.68 did not show any harmful effect of edoxaban on the renal function, but more data are needed to draw any conclusions.
Conclusions
Anticoagulant-related nephropathy is a novel and dangerous complication in the prevention and treatment of thromboembolic episodes. Due to a large number of patients receiving anticoagulants and the lack of appropriate monitoring and histopathological testing, the prevalence of ARN is still underestimated. Scarce clinical trials on patients with impaired kidney function taking anticoagulants significantly limits our knowledge about ARN. Special awareness is needed in reference to patients taking anticoagulants, with kidney injury of unknown origin. Based on the available data, VKAs are responsible for even 37% of ARN cases, whereas DOACs for about 5–14% of them. The inhibition of thrombin and PAR-1 are the main mechanisms related to ARN. The anti-inflammatory and antioxidative effects of novel DOACs, rivaroxaban and apixaban, are interesting findings, explaining to some extent a lower incidence of ARN after these DOACs. Despite the fact that CKD is one of the strongest risk factors for ARN, anticoagulants may cause ARN even in previously healthy patients. Since in patients with ARN the kidney function may not be restored, this specific kind of kidney injury should be taken into consideration as a cause of CKD in a large group of patients.