Abstract
Background. Epidemiological studies of tetracycline (TE) resistance genes and integron gene cassettes, particularly in urine samples, are limited in Turkey.
Objectives. To investigate antibiotic susceptibility profiles, extended-spectrum beta-lactamase (ESBL) positivity, tet gene types, class-I/-II integron gene cassettes, and clonal relationships among tet-resistant isolates of Escherichia coli from urine cultures of outpatients.
Materials and methods. Isolates were identified using conventional methods and the automated Vitek® 2 Compact system. Antimicrobial susceptibility was performed for 19 antibiotics. The ESBL production was performed using the Kirby–Bauer disk diffusion test. The double disk synergy test was used for confirmatory testing. Polymerase chain reaction (PCR) was used to determine the presence of class-I/-II integron gene cassettes and tetA, tetB and tetD resistance genes. The pulsed-field gel electrophoresis typing was performed to identify clonal relations.
Results. A total of 121 isolates were obtained and found to be resistant or sensitive to ampicillin and amikacin/imipenem. Resistance to ceftazidime, cefotaxime and ceftriaxone was determined to be 31.3%, 77.6% and 83.1%, respectively. Tetracycline resistance was detected in 82 isolates, mostly caused by the tetB gene. No tet gene was detected in the remaining 39 isolates. Although 64 out of 82 isolates carried a class-I integron, only 4 had a class-II integron (with sizes of 800–2900 base pairs). Furthermore, tet genes were identified with different size class-I integron gene cassettes. However, tet genes were not detected in any isolate identified with integron gene cassette II. Clonally, the isolates were found to be related in subgroups because they were community-acquired.
Conclusions. This study showed that the tetB gene is most commonly found in E. coli isolates grown in urine samples from the Turkish population.
Key words: E. coli, PFGE, tetracycline resistance, ESBL, integron gene cassettes
Background
Urinary tract infections (UTIs) are usually caused by Gram-negative Enterobacterales, with the most common pathogens being Escherichia coli and Klebsiella pneumonia spp.1, 2 Antimicrobial resistance is increasing among uropathogens and the production of β-lactamases is a major resistance mechanism.3
Tetracyclines inhibit protein synthesis by binding to the bacterial ribosome. At present, the prevalence of tetracycline (TE) resistance is increasing in bacteria isolated from both, human patients4, 5 and animals.6 Resistance is widely disseminated in E. coli through TE resistance genes, such as tetA.7 A previous study found that the prevalence of TE resistance in E. coli isolates from humans increased by 0.45% per year from 1950 to 2001.5 Because of the spread of TE resistance, the use of tetracyclines in humans has gradually decreased. However, tetracyclines remain among the most used antibiotics in livestock production worldwide.8
Different TE resistance determinants have been demonstrated. They have been classified as A–E classes, being the most frequently detected genes among the Enterobacterales family.9 Tetracycline resistance genes (tet genes) are divided into 11 classes that comprise more than 40 genes.10, 11, 12 Studies from the USA have shown that tetA is the 2nd most frequent TE resistance efflux pump in both, human and animal isolates.13 In Denmark, tetA is also the most common TE efflux pump type found in clinical isolates of E. coli.14
Integrons are genetic elements that encode antibiotic resistance determinants and have the ability to integrate or transport certain genes. They can also be transferred from one bacterium to another, as they are carried by plasmids or transposons. This causes a strong antibiotic selective pressure resulting from the transfer and spread of antibiotic resistance elements.15, 16
The rapid spread of TE resistance among bacteria is due to the localization of tet genes on movable genetic structures such as plasmids, transposons and integrons. However, there are scarcely any studies showing the relationships between the main tet genes and integrons.
Objectives
The present study aimed to screen the tetA, B and D genes together with class 1 and 2 integron gene cassettes from 121 extended-spectrum β-lactamase (ESBL)-positive E. coli isolates, obtained from the urine cultures of adults. Additional aims were to determine the molecular sizes and classes of the detected integrons and the clonal relationship with pulsed-field gel electrophoresis (PFGE)among the TE resistance isolates. Studying integron gene cassettes and related resistance phenotypes will provide important information about the mechanisms of the acquisition of multiple antibiotic resistance genes in clinical isolates, which then can be used to effectively guide clinical treatment.
Materials and methods
Ethical approval
The study protocol was approved by the Faculty of Medicine Non-invasive Studies Ethics Committee, Recep Tayyip Erdoğan University, Turkey (approval No. 40465587-050.01.04-14 and No. 2021/13), and the Rize Provincial Health Directorate (Ref. No. 64247179-799).
Sample collection and bacterial
isolation/identification
Escherichia coli isolates were obtained from urine samples collected at 7 different clinics (infectious diseases, urology, internal medicine, emergency service, nephrology, neurology, and gynecology) between June 2017 and June 2018. All urine samples were obtained from outpatient adults aged 18–83 years. Since TE is not used in pediatric patients, the pediatric age group was not included in this study. The clinical data of the patients were retrospectively obtained from medical records and medical tables.
Samples were sent to a microbiology laboratory (Rize State Hospital, Rize, Turkey) for analysis. Clinical specimens were plated on eosin methylene blue (EMB)/MacConkey agar for isolation. The isolates were identified using conventional methods and the automated Vitek® 2 Compact system (BioMérieux, Marcy-l’Étoile, France).
Antimicrobial susceptibility testing
Antimicrobial susceptibility experiments were performed in accordance with the Clinical and Laboratory Standards Institute (CLSI) criteria17 using both the Kirby–Bauer disk diffusion method and the automated Vitek® 2 Compact system (BioMérieux). Sensitivity to the following antibiotics was tested: ampicillin (AMP), ampicillin/sulbactam (AMP/SAM), amoxicillin/clavulanic acid (AMC), cefazolin (KZ), cefotaxime (CTX), cefuroxime (CXM), cefixime (CFM), ceftriaxone (CRO), ceftazidime (CAZ), amikacin (AK), gentamicin (CN), tobramycin (TOB), ciprofloxacin (CIP), norfloxacin (NOR), imipenem (IPM), aztreonam (ATM), nitrofurantoin (F), trimethoprim/sulfamethoxazole (TMP/SXT), and tetracycline (TE). The E. coli ATCC 25922 (American Type Cell Culture (ATCC), Manassas, USA) was used as the quality control strain.18 Categorical variables are expressed in terms of frequency (n) and percentage (%).
Screening of extended-spectrum β-lactamase production
All isolates were screened for extended-spectrum β-lactamase (ESBL) production with the Kirby–Bauer disk diffusion test using 3 indicator cephalosporins, namely ceftazidime (30 µg), cefotaxime (30 µg), and ceftriaxone (30 µg). The test was performed by inoculation on a Mueller–Hinton agar plate (Merck, Darmstadt, Germany) with a sample of the tested strain. The isolates were considered to be resistant if the diameter of the inhibition zone for ceftazidime, cefotaxime or ceftriaxone was ≤22 mm, ≤27 mm or ≤25 mm, respectively, after 18-h incubation at 37°C.18, 19
Confirmation of ESBL–producing isolates
All E. coli isolates showing resistance to any of the 3 cephalosporins were then subjected to confirmatory testing using the double disk synergy test (DDST), according to the 2019 CLSI guidelines.18 Ceftazidime (30 µg), cefotaxime (30 µg), ceftriaxone (30 µg), and amoxicillin-clavulanic acid (20/10 µg) were used for ESBL detection. Amoxicillin/clavulanic acid and cephalosporin disks were placed at a distance of 20 mm from the center on lawn cultures on Muller–Hinton agar plates. The plates were incubated at 37°C overnight for 24 h. The zone of inhibition of cephalosporins towards the amoxicillin/clavulanic acid disk was considered to be a positive result and the isolates were interpreted as ESBL producers. Escherichia coli ATCC 25922 was used as the control.18, 19, 20
Detection of integron gene cassettes
and tet genes
The presence of integron gene cassettes and tet genes was investigated with polymerase chain reaction (PCR) method (Table 1). To obtain template DNA, single colony bacterial strains were inoculated in 3 mL of Luria–Bertani (LB) broth medium (1% tryptone, 0.5% yeast extract, 0.5% NaCl, pH 7.4) at 37°C for 16 h at 200 rpm. Next, 1.5 mL of the grown culture was used to obtain total genomic DNA using the boiling method. For PCR testing, 3 µL of the supernatant was used as the template DNA. The PCR was performed using the compositions and cycling parameters detailed in Table 1. The amplification products were then run in 1.2% agarose gel with a 100 base pair (bp) molecular marker (Thermo Fisher Scientific, Waltham, USA) and visualized with UV light. The oligonucleotides used in PCR testing are listed in Table 1.
Molecular typing of tetA-, tetB-,
and tetD-positive isolates
The PFGE typing was performed to identify the clonal relations among 46 isolates coding tetA, tetB and tetD. Isolation and deproteinization of genomic DNA were performed following a previously reported protocol with minor modifications.25 Briefly, E. coli colonies on Mueller–Hinton agar were suspended in 1 mL of cell suspension solution (CSS) (100 mL of Tris-HCl, 100 mL of ethylenediaminetetraacetic acid (EDTA) pH 8.0) and optical density (OD) was adjusted to 0.8 (590 nm). The cells were embedded in low (2%) melting agarose. Molds were prepared with the help of agarose and refrigerated at 4°C for 10 min. The prepared strain molds were mixed with cell lysis solution-1 (CLS-1) (50 mm Tris-HCl, 50 mm EDTA, pH 8.0, 2.5 mg/mL lysozyme, 1.5 mg/mL proteinase K) and incubated at 37°C for 1 h in a water bath. At the end of the period, CLS-1 was removed. The cell lysis solution-2 (CLS-2; 0.5 M EDTA, 1% sarcosyl, 400 µg/mL proteinase K) was placed on molds and incubated in a water bath at 55°C for 2 h. After the lysis step, the molds were washed 3 times, each with dH2O and 1 × TE (Tris-EDTA). After digestion of the cells and washing of the plugs, genomic DNA in the agarose plugs was restricted with 10 U of Xbal (Thermo Fisher Scientific) for 3 h at 37°C in an incubator. The DNA fragments were separated on 1% pulse-field certified agarose (Bio-Rad Laboratories, Nazareth, Belgium) gels run in 0.5 × Tris-borate-EDTA buffer (44.5 mM/L Tris, 44.5 mM/L boric acid, 1 mM/L EDTA (pH 8.0)) using a CHEF-DR II system (Bio-Rad Laboratories). The electrophoresis conditions were 14°C at 6 V/cm2 for 18 h. The initial and final switch times were 5 s and 20 s, respectively. The gel was stained with ethidium bromide (5 mg/mL) for 30 min and photographed under ultraviolet (UV) light. The DNA band profiles were analyzed using GelCompar software v. 3.0 (Applied Maths, Sint-Martens-Latem, Belgium). According to the interpretative criteria of Tenover et al., the clinical isolates were aligned according to the PFGE profiles, with profiles sharing 100% similarity grouped in clusters.26
Results
A total of 121 ESBL-positive E. coli isolates were obtained from the outpatients’ urine samples. These strains were biochemically characterized using an automated system, and subjected to antibiotic susceptibility testing, ESBL screening and phenotype confirmation tests. All isolates were tested for susceptibility to 19 antimicrobials. The frequency of resistance to each of the tested antibiotics is shown in Table 2.
Antibiograms showed that 121 (100%) isolates were resistant and sensitive to ampicillin and amikacin/imipenem, respectively. Although there was 100% resistance to ampicillin, ampicillin/sulbactam resistance was found to be 33.8%. This showed the rate of resistance against inhibitors in the isolates. Similarly, for the amoxicillin/clavulanic acid combination, the rate of resistance to the inhibitor was 12.2%. In the ESBL screening test, resistance to ceftazidime, cefotaxime and ceftriaxone was determined to be 31.3%, 77.6% and 83.1%, respectively. All ESBL-producing strains were determined to be susceptible to amikacin and imipenem. This result is important in terms of showing that there is no resistance to these antibiotics in outpatients. After amikacin and imipenem, the highest sensitivity was observed for nitrofurantoin. Tetracycline resistance was determined in 82 isolates (67.7%).
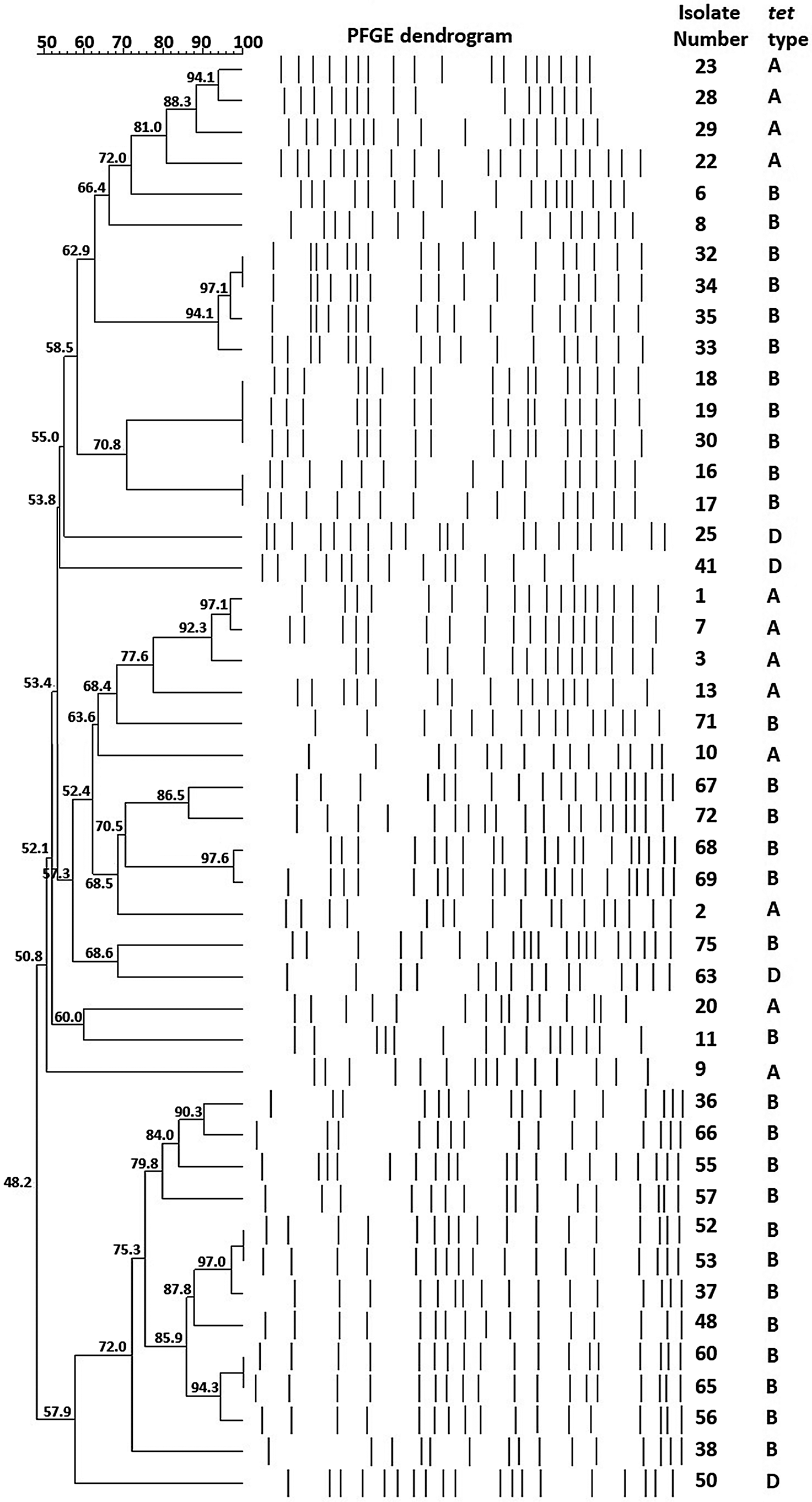
Among ESBL-producing E. coli isolates, those with TE resistance were selected and the presence of tet genes responsible for resistance and class-I/-II gene cassettes responsible for transferable resistance was investigated (Table 3). Among these samples (n = 46), tetA, tetD and tetB, which are responsible for TE resistance, were detected in 12, 4 and 30 isolates, respectively. This result showed that TE resistance in clinical E. coli samples was mostly caused by the tetB resistance gene. No tet genes were detected in the remaining 36 isolates. The integron gene cassettes were isolated in a total of 68 samples (class-I integron: n = 64; class-II integron: n = 4). The largest integron gene cassette was determined as approx. 2900 bp, while the smallest integron gene cassette was determined as 800 bp. The average integron gene cassette size was around 1600 bp. The tet genes were identified together with different size class-I gene cassettes. However, the same was not true for the integron class-II gene cassettes: they were detected in 4 samples, but none of the tetA, tetB and tetD genes were detected. This result showed that TE resistance developed in isolates carrying class-II gene cassettes by a mechanism different from these 3 genes. The dendrogram was basically divided into 2 different clades. One clade consisted of 1 tetD-positive and 12 tetB-positive isolates. In the other clade, tetA, tetB and tetD were located together in different arms (Figure 1).
Discussion
Escherichia coli isolates are present in normal human fecal flora, and some strains are the most common cause of UTIs. Antimicrobial therapy is very important in the treatment of UTIs. However, the drug resistance generated specifically by ESBL-producing microorganisms results in failure in the treatment of UTIs. In the present study, ESBL production was observed to be high in E. coli isolates obtained from outpatients with symptomatic UTI and 121 of them exhibited resistance to at least 1 of the 3 indicator cephalosporins and identified as ESBL producers. Furthermore, 82 of the ESBL-positive isolates were TE-resistant and tet genes (A, B and D types) were determined in 46 of these isolates. These 3 tet genes are associated with TE efflux in Gram-negative bacilli and have been previously detected in TE-resistant E. coli isolates.27 In addition, in UTIs, the tetD gene was present at the lowest rate while the tetB gene was present at the highest rate; notably, they did not occur together in any strain. We failed to detect the tet gene in the remaining 39 TE-resistant isolates. However, these isolates may have harbored another resistance mechanism, i.e., different tet genes besides tetA, tetB and tetD, or enzymatic alteration or ribosomal protection.28 Ribosomal protection has been demonstrated as another mechanism of resistance to TE in the Enterobacterales family that is mediated by different tet genes.29 Both tetA and tetB genes encode efflux mechanisms to confer resistance to tetracyclines, and have been previously identified as the most common TE resistance genes in E. coli of both, human and animal origin.30 It has also been reported that tetB provides additional resistance against doxycycline, in contrast to tetA which confers resistance against TE, oxytetracycline and chlortetracycline.9
The majority of ESBL positivity and tet resistance is associated with conjugative or mobilizable elements, which partially explains their wide distribution in bacterial species. Gram-negative efflux determinants are normally found in transposons inserted into a diverse group of plasmids from a variety of incompatibility groups. These plasmids may carry multiple antibiotic resistance genes, which confer resistance to various antibiotic families.27 Integrons cause the spread of antimicrobial drug resistance. Although the transport of ESBL and TE resistance on integrons was not investigated in this study, the high rate of integron gene cassette carriers has made us consider the possibility of transferring genes responsible for ESBL resistance. Tetracycline resistance genes may also be carried together with other antibiotic resistance genes by plasmids harbored integron gene cassettes. A selective advantage for resistance genes carried on the plasmid may enhance the persistence of plasmid-bearing strains in an environment contaminated with antimicrobial compounds. Effective expression of tet genes in the Enterobacteriaceae family could result in high levels of resistance to TE. We are of the opinion that this may be an important risk factor in the treatment of human infectious diseases caused by these microorganisms.
In our study, since E. coli isolates are community-acquired samples, they were not expected to be highly similar clonally. The PFGE was found to be effective for distinguishing isolates from each other only in subgroups according to tet type. Furthermore, it has been observed that PFGE grouped the isolates according to other genomic factors, rather than by the antibiotic resistance profile of isolates. For example, tetD-positive strains were co-resistant to CRO but they grouped in the different clades. Besides this, isolates No. 50 and 63 have exactly the same antibiotic profile but they were located on different branches, which also supports the notion that PFGE is independent of the resistance profile.
Limitations
Our study has 2 main limitations. Firstly, it is not a multi-institutional study. Secondly, base sequence analysis of integrons could not be performed due to limited funding.
Conclusions
In summary, this is one of limited studies investigating the coexistence of tet genes and integron gene cassettes of E. coli isolates, obtained from urine samples in Turkey. The results provide data on the coexistence of integrons and tet genes in E. coli strains isolated from the outpatients. Studying integron gene cassettes and related resistance phenotypes can provide important information about the mechanisms of acquisition of multiple antibiotic resistance genes in clinical isolates. It can be argued that uncontrolled broad-spectrum antibiotic use may be the main reason for the development and increase of resistance. Furthermore, high rates of resistance may lead to a trend towards new alternatives or antibiotics that were used extensively in the past, but their use has decreased in recent years. Comprehensive studies are needed to better examine the relationship between antibiotic resistance and integron gene cassettes together with epidemiological data.