Abstract
Background. Effects of osteoarthritis (OA) are observed in experimental animal models using different gait analysis systems.
Objectives. The aim of this study was to determine whether the Noldus CatWalk XT v. 10.9 gait analysis system (CatWalk) device can be used effectively in a chemically induced rat OA model and to reveal the strengths and weaknesses of the system compared to manual gait analysis.
Materials and methods. Ten Wistar rats were run on a manual walking platform as well as on the CatWalk and the basal values were recorded. For OA induction, monosodium iodoacetate (MIA) was injected into the left knee of all rats under anesthesia. After a period of 4 weeks for OA development, the rats were again run on both the manual and CatWalk gait platforms. For manual gait analysis, the stride length, paw print width and paw print length were measured on both knees. In addition to these parameters, the average run speed, run duration, maximum contact intensity, paw print area, mean stance, and swing speed were measured on the left knee (affected knee) using the CatWalk device.
Results. Significant differences were observed in the stride width (p = 0.0272), left stride length (p = 0.0344), and left paw print length (p = 0.0233) recorded before and after OA via the manual walking platform. For CatWalk, a significant difference was detected in the left knee’s average run speed (p = 0.0010), maximum contact intensity (p = 0.0155), paw print length (p = 0.0058), paw print width (p = 0.0324), and swing speed (p = 0.0066) based on data obtained before and after OA.
Conclusions. The CatWalk gait analysis system is suitable for the evaluation of OA rat models and related interventions. It also provides additional parameters compared to the manual system and minimizes human-related variation.
Key words: CatWalk, osteoarthritis, rat model, gait analysis, monosodium iodoacetate
Background
Animal models, especially those involving rodents, play an important role in drug development studies. For instance, molecules are used in preclinical studies involving animals to assess their effects on a specific disease to minimize the associated risks for humans before clinical studies.1 In this context, gait analysis in animal models that respond to a specific treatment and behavioral models are highly important.2
Osteoarthritis (OA) is the most frequently seen form of arthritis and its prevalence increases with age.3 Old age and obesity are particularly significant risk factors for OA.4, 5 Although OA most frequently affects the knee joints, it can be seen in any joint in the body, including hip, waist and finger joints.6 The main symptoms include joint pain, joint stiffness and swelling.7 Currently, clinicians consider OA as a failure of the entire joint structure, such that OA not only affects articular cartilage but also affects the subchondral bone, ligaments, joint capsule, synovial membrane, and periarticular muscles. By another definition, OA starts as a result of the mechanical failure of the joint, during which the joint makes an effort to repair the defect area and fix the abnormal joint biomechanics.8 For OA treatment, pharmacological, non-pharmacological and surgical options are available. However, none of the available treatments provide an absolute solution for OA. Thus, there have been continuous efforts to reveal the complete pathobiology of OA and develop better treatment options. The response of animal models to OA treatments is measured using various kinds of in vivo techniques and through postmortem evaluations.
In previous studies, various chemicals have been used for the induction of OA in animals.9 In the present study, monosodium iodoacetate (MIA) is used. Intraarticular injection of MIA is one of the animal models of chemically induced OA. As a metabolic inhibitor, MIA causes disruption of glycolysis in cells by inhibiting the glyceraldehyde-3-phosphate dehydrogenase enzyme.10 Subsequently, this leads to an increase in oxidative stress in the environment that induces loss of chondrocytes, resulting in reduction of cartilage thickness and osteolysis.11, 12, 13 This mechanism of action governs the basis of MIA-induced OA. These changes bring about histological and morphological modifications in the joint cartilage similar to the prognosis of OA patients.14, 15 Many studies have been conducted using MIA-induced OA rat models.16, 17, 18, 19
There are several methods of rodent spatiotemporal gait analysis, and these systems have significant advantages compared to the main inkpad methods used earlier. Unfortunately, the system array and reporting of multiple parameters frequently make gait analysis more difficult. The CatWalk XT v. 10.9 gait analysis system (CatWalk; Noldus Company, Wageningen, the Netherlands) is a gait analysis device suitable for rodents, especially rats and mice. The data obtained by walking a rat on a platform are automatically saved.
Objectives
The purpose of this study is to evaluate the gait analysis patterns of rats before and after onset of OA induced by MIA. Using the values before induction of OA, the rat model was evaluated in terms of how the gait parameters changed after OA induction using the CatWalk device, which is one of the most powerful gait analysis systems available for rats and mice. In addition, data collected using the CatWalk software were compared to those obtained manually on graph paper. Evaluation of this rat model of OA will provide a basis for future OA studies involving gait analysis.
Materials and methods
Animals
Our research was approved by the Research Ethics Committee for Animal Experiments of Kırıkkale University, Turkey. The study was carried out with 10 male conventional Wistar rats weighing approx. 300 ±31.3 g and aged 12–24 weeks. The rats were kept in individual cages and fed ad libitum. A pellet diet was used for food and refined tap water was provided in an autoclavable Makrolon bottle. Sawdust was used as the bedding material and cleaned 4 times per week. The rat holding room was maintained at 23°C with 60% humidity and a 12 h/12 h light/dark cycle. The air was completely replaced 15 times per hour. Prior to the gait procedures and injections, food was restricted for 24 h and water was restricted for 6 h. Rats were regularly run on the CatWalk platform prior to the study and were thus adapted to the device. The animals were numbered by marking their tails.
Knee osteoarthritis pain model
Anesthesia was achieved by injection of 100 mg/kg intramuscular ketamine (Alfamine®; Egevet, Izmir, Turkey; 100 mg/mL) and 8 mg/kg intramuscular xylazine (Alfazyne®; Egevet; 2%) through a 30-gauge needle. Under general anesthesia, the left knees of the rats were shaved and 0.2 mg of MIA (Sigma-Aldrich, St. Louis, USA) in 10 µL of sterile saline was injected. The solution was injected through the patellar ligament using a 27-gauge needle with the leg flexed at a 90° angle at the knee. After surgery, we waited 4 weeks for the formation of chemically induced OA.
Gait analysis
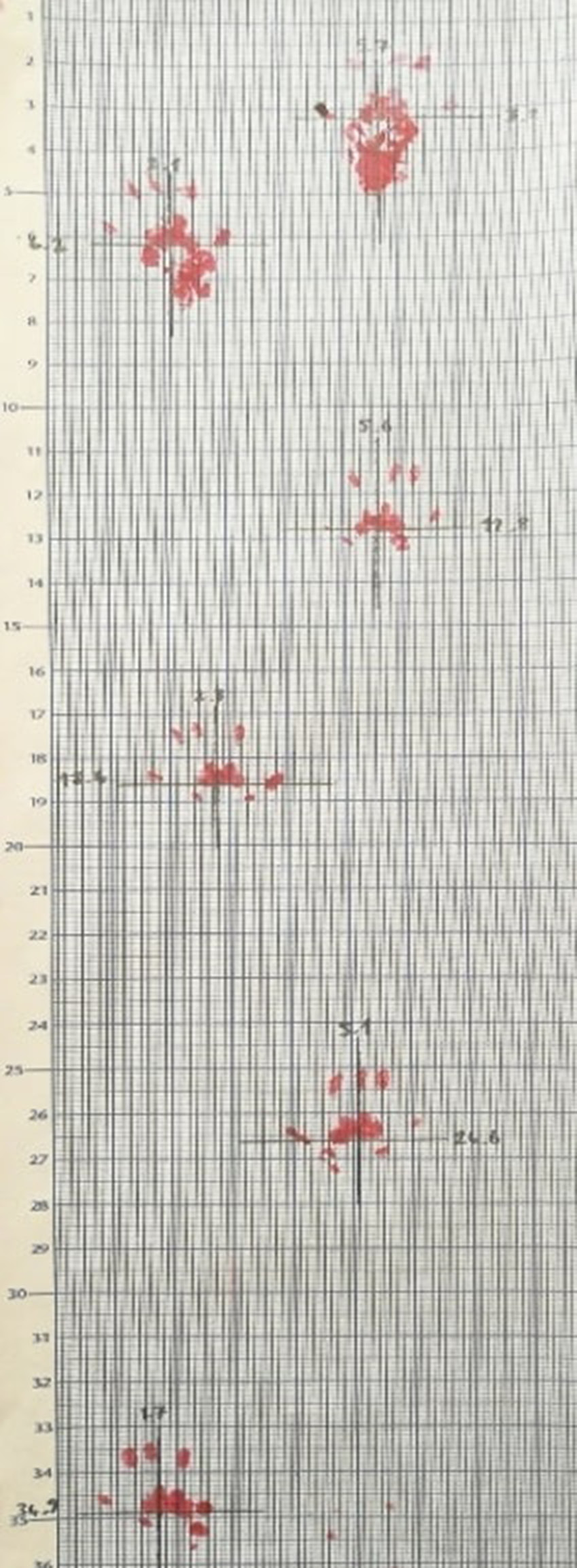
We selected 2 different systems for gait analysis. The 1st gait analysis system was a manual walking platform. Analysis with a manual platform involves the movement of the animal along a single inkpad followed by the measurement of the pattern of the ink prints on graph paper. Bait was placed at the end of the manual walking platform to encourage the rats. The rats walked in a dark room until they were accustomed to the manual walking platform (Figure 1A,B). Once the rats were completely used to the platform and started walking, their hind paws were painted with ink. The rats were walked on graph paper and the values were recorded. The main data, such as stride length, stride width, paw print length, paw print width, and paw print area, are then measured from this paper. Variables such as the angle between the paws may also be assessed, but it should be kept in mind that these are not independent measurements and are derived from step length and width.20, 21 Of note, the amount of ink is inconsistent between steps and trials and, therefore, the paw print areas are highly variable. Thus, modern high-speed videography has a higher probability of enabling robust analysis of spatial parameters.
The present study also used the CatWalk device for analysis using a machine learning-based approach. CatWalk is a gait analysis system for mice and rats that allows voluntary diagonal passage toward a target box over a glass surface in a darkened room, with the animals allowed to walk freely.22 A light from a lamp is aimed through the glass surface. When an animal’s paw touches the glass surface, the light beams are reflected downward. The entire run is recorded with a video camera. When the rat walks, the paw prints are automatically captured, recorded and analyzed.23 This system has been explained in detail elsewhere.24 The animals walked on graph paper on the manual walking platform (Figure 2) and the CatWalk device (Figure 3A,B) in sequence before injection. All base values were recorded for both platforms after this first walk. At this stage, the rats were administered MIA as mentioned above and 4 weeks were allowed for the formation of OA.
Four weeks after induction of OA, the animals were forced to walk again on the manual walking platform as well as the walking platform of the CatWalk device. Data collected both manually and via the CatWalk system before and after OA induction were categorized and analyzed. Data collected before and after OA induction were compared, and data collected manually and via CatWalk were analyzed for the presence of correlations.
With the analysis conducted using the CatWalk device, several parameters such as pre-treatment walking velocity, usage of the treated knee, comparisons with the other knees, run duration and average run speed, maximum contact area and intensity, stride length, paw print length, paw print width, paw print area, mean stance, and swing speed were analyzed by recording both baseline and OA values.
Histological analysis
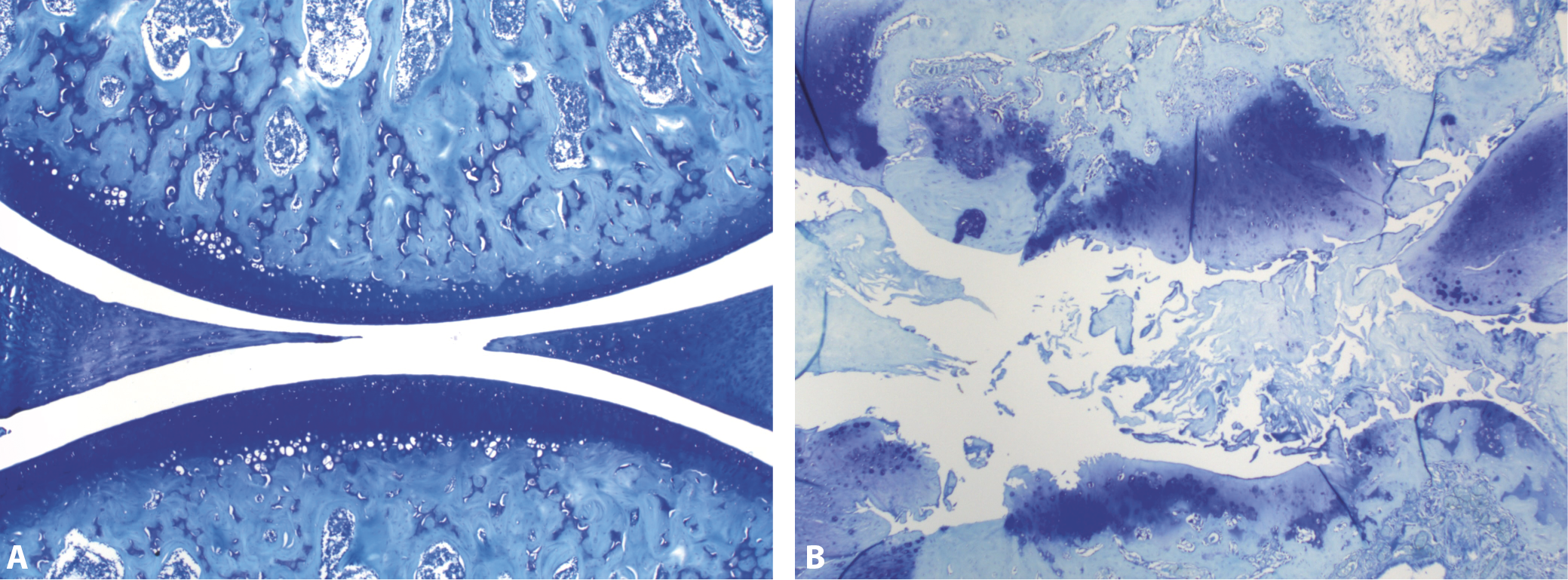
All animals were euthanized by cervical dislocation after the second walk. The complete knee joint was collected. The joint samples were fixed in 10% phosphate-buffered formalin and then decalcified in 5% formic acid. Decalcification was confirmed by radiography. Sections were then processed with routine histological techniques. The joints in paraffin blocks were cut to a thickness of 7 µm and finally stained with toluidine blue (Figure 4A,B) which enabled the researchers to evaluate loss of glycosaminoglycans in addition to morphology. The samples were then examined under a light microscope. The most severely affected regions of the joints were scored using the modified scoring system shown in Table 1. A total score for left knee joints was obtained and used in statistical analyses. Histological score data were expressed as median (minimum–maximum) scores.
Statistical analyses
Data are presented as median (minimum–maximum) values. The Wilcoxon rank-sum test, which is a non-parametric test, was used to assess whether or not there was a significant difference between both the base and after OA values of distance and histological OA scores between control and MIA-injected knees. The analyses were carried out using IBM SPSS Statistics for Windows v. 21 (IBM Corp., Armonk, USA). A value of p < 0.05 was accepted as significant in all statistical test.
Results
In histological sections, MIA-injected knees exhibited signs of OA. Surface erosion, fibrillation, clone formation, eburnation, tidemark invasion, and synovitis were common findings. The OA scoring system used for the rats in this study is shown in Table 1. While the average OA score in the MIA-injected knee was 21.4 (18–25), the OA score in the control knee was 0.7 (0–3). The OA scores in the MIA-injected knees were significantly higher compared to those in the control knees (p < 0.05).
Seven parameters were evaluated with the manual walking platform. The calculated values are shown in Table 2. After induction of OA, the) stride length of the left (affected) knee and the left paw print width and length values decreased; at the same time, the stride length of the right (unaffected) knee, right paw print width and length, and stride width increased. Among the data recorded from the manual walking platform, the values of stride width, left stride length and left paw print length were statistically significant.
The CatWalk software provided data for 9 different parameters. As the injections were administered to the left knee in all animals, values pertaining to the left hind limb were prioritized for analysis. The recorded values from the CatWalk device are shown in Table 3. After induction of OA, the run duration and mean stance values of the left knee increased; at the same time, the average run speed, maximum contact intensity, paw print length, paw print width, paw print area, stride length, and swing speed of the same knee decreased. Among the data recorded with the CatWalk device, the average run speed, maximum contact intensity, paw print length, paw print width, and swing speed values were statistically significant.
Discussion
There are many different systems that can be used for automatic gait analysis.26 These include DigiGait, TreadScan, the open-source Experimental Dynamic Gait Arena for Rodents, and CatWalk. In a study by Xu et al., the DigiGait and CatWalk gait analysis systems were compared and the advantages of each were reported.27 Five different automatic gait analysis systems were compared in a study by Jacobs et al., who mentioned that these devices play an important role in rodent OA models.28 In the present study, we compared manual platform data analysis and automated CatWalk gait data analysis.
The CatWalk gait analysis system allows simultaneous analysis of several factors by recording the steps of rats walking on a band, categorizing these steps and creating dozens of parameters.29 Data regarding all of the parameters, which are collected by the software, were statistically analyzed in this study. Upon the detection of significant differences, data were compared between groups. The CatWalk device can provide many more types of data, and the software is fast and easy to use. In this sense, it was observed that the CatWalk device has clear advantages over the manual platform method in terms of gait analysis. In addition to the parameters measured by classical methods, CatWalk can evaluate parameters that we think are important for OA within the walking analysis system.30
The MIA administration is an important method for chemical induction of OA.16 Other methods of chemical OA described in the literature include anterior cruciate ligament transection, destabilization of the medial meniscus and the use of collagenase.31 For example, Adães et al. used collagenase to create a rat model of OA,32 while Jacobs et al. used surgical medial meniscus and anterior cruciate ligament transection to create a similar OA model.33 Most importantly, the MIA OA model progresses with a pathology similar to that of degenerative OA.13, 34, 35 Thus, we preferred the MIA model for OA induction. Signs of OA were observed in the left hind limbs of all rats following MIA administration in our study.
In the analysis, the first marked evidence was the presence of differences between data collected prior to and after OA induction. Such differences were obtained from both the CatWalk and the manual platform. Therefore, the parameters used in the CatWalk gait analysis system can be considered to detect gait changes in the MIA-induced rat OA model, a successful and well-established OA model documented in the literature.36
In the measurements after OA induction, it was observed that the rats deliberately avoided putting weight on their left hind limbs and possibly felt pain.37 The average run speed and swing speed decreased and, secondarily to this, the run duration and main stance increased. Moreover, the decrease in the length, width, and area of the paw prints and stride length after OA induction suggest that the rats stepped on the platform less widely to reduce the pressure on the foot of the affected limb. This hypothesis is also supported by decreases in the maximum contact density values. Whereas paw print measurements on the manual walking platform are performed manually with the help of a ruler, paw print measurements in the CatWalk system are automatically analyzed and provided by the software. Thus, the CatWalk system is more convenient and more objective than the manual walking platform. Gabriel et al. used the CatWalk software to assess acute inflammatory pain model rats and reported that they obtained objective and detailed data.38 In another study, rats with complete Freund’s adjuvant-induced monoarthritis were analyzed using CatWalk software and clearer and non-biased results were also obtained.39 All of these results support that the parameters used by the CatWalk software are valuable for monitoring in vivo OA progression as well as responses to any intervention in OA.
Limitations
Our study has some limitations. First, the measurements from the manual walking platforms were obtained using a completely hand-held manner and may thus contain intra-observer and inter-observer measurement errors. One disadvantage of the CatWalk gait analysis system is that only rats and mice can be used. Also, rodents need a certain amount of time to adjust to the walking platform. Since the study was carried out in a dark room, the rodents behaved shyly in the environment and required a period of serious acclimatization.
Conclusions
In conclusion, the CatWalk gait analysis system is a useful, reliable and convenient method for monitoring gait changes in rat knee OA models. The CatWalk gait analysis system and its associated parameters, such as stride length, paw print width, paw print length, average run speed, run duration, maximum contact intensity, paw print area, mean stance, and swing speed, are appropriate for the evaluation of rat knee OA models.