Abstract
Volume overload can be both the cause and effect of chronic kidney disease (CKD). Overhydration often accompanies renal insufficiency. In cardiovascular disease (CVD), fluid overload can also be the cause of renal function impairment. Beside salt restriction, loop diuretics are the first-line therapy. Frequently developed resistance can be overcome by switching to intravenous administration, adding albumin alone or in combination with other diuretics. Transient factors like infection or contrast media can impair diuretic response and contribute to congestion. Apart from conservative management, ultrafiltration (UF) and peritoneal dialysis (PD) are used. In huge congestion with inadequate diuretic effect, hemodialysis with UF plays an important role as a temporary or permanent remedy. An increasing amount of data indicates that sodium-glucose co-transporter-2 inhibitors (SGLT2i) have allowed for a breakthrough in controlling fluid volume in diabetic and non-diabetic patients with CKD. Sodium-glucose cotransporter 2 inhibitors show cardio- and renoprotective effects and have a positive impact on hard cardiovascular and renal endpoints.
Key words: chronic kidney disease, diuretics, dialysis, cardiorenal syndrome, SGLT2 inhibitors
Introduction
A patient exhibiting legs swelling, orthopnea and shortening of breath with known heart failure (HF) or diabetes mellitus (DM) can develop deterioration of kidney function. If correcting the redundant amount of fluid, regardless of the intervention, restores kidney function, we can assume that overhydration was the cause of renal injury. Progressively declining glomerular filtration rate (GFR) in the course of renal disease results in hypertension, sodium retention and fluid overload. In anuric patients, overhydration, as the effect of kidney damage, can be life threatening. The most frequent clinical situation is the mixture of the 2 examples mentioned, best described as cardiorenal syndrome (CRS) with overhydration as a net effect. In the last few years, new data about how overhydration correction is beneficial has been published.
Do we have enough
good quality data in CKD?
Circulatory system abnormalities occur frequently in patients with chronic kidney disease (CKD). Overhydration is one of the symptoms resulting predominantly from cardiovascular disease (CVD) cardiac insufficiency. Unfortunately, there is still not enough data of sufficient quality to fully understand the cardiorenal interactions. The problem should not be downplayed, because CKD in various stages affects over 850 million people worldwide, which doubles diabetes and exceeds twentyfold cancer occurrence. Because of the often insidious course of the disease, many patients are unaware not only of the existence of CKD itself, but also of the potential complications, including CVD. It should be emphasized that almost half of CKD patients die because of major cardiac events.1
Cardiorenal syndromes
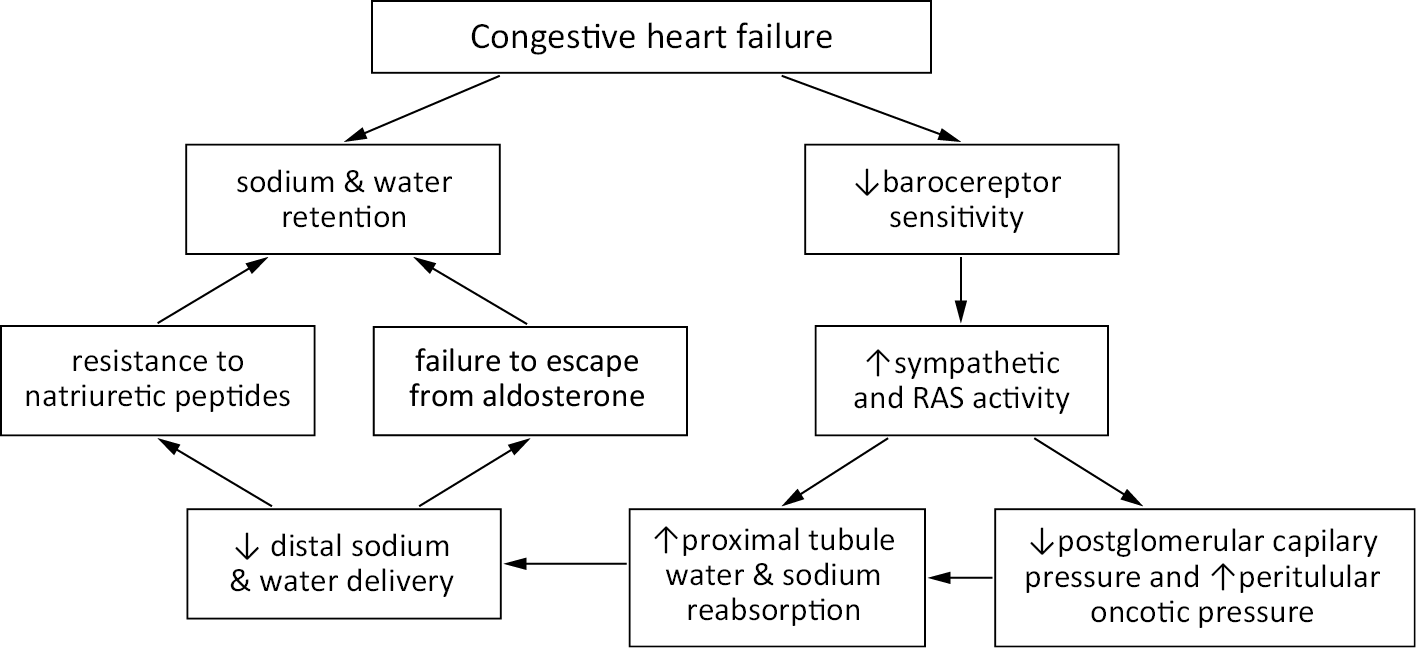
Overhydration can be perceived in the context of cardiorenal crosstalk, which is bidirectional. The systematic classification of the organ interactions was proposed at the consensus conference of the Acute Dialysis Quality Initiative in 2008.2 An interdisciplinary group of experts and opinion leaders categorized CRS into 5 classes on the basis of the primarily underlying pathology and its acute or chronic character: acute cardiorenal syndrome (type 1), chronic cardiorenal syndrome (type 2), acute reno-cardiac syndrome (type 3), chronic reno-cardiac syndrome (type 4), and secondary cardiorenal syndromes (type 5). Vicious cycle of sodium and water retention is a hallmark of most frequent CRS type 2 (Figure 1).
Although the cardiovascular mortality among hemodialysis patients is intuitively more understandable, it has been proven that even patients in earlier stages of CKD are at higher CVD risk.3 Among CVDs, there is the lethal triad: congestive HF, acute myocardial infarction (MI) and sudden cardiac death that take the largest toll.4 Already microalbuminuria, even with preserved GFR, speaks for cardiovascular mortality,3 and the influence of estimated glomerular filtration rate (eGFR) on the major adverse cardiovascular events risk is direct.5 Furthermore, CKD patient is less likely to progress to end-stage renal disease (ESRD) than to pass away because of CVD.6 On the basis of numerous analyses including large database examination consisting of over million patients,7 it should be concluded that CKD is one of the most significant risk factors for cardiovascular events.3 The up-to-date World Health Organization (WHO) charts to evaluate ten-year cardiovascular mortality ratio do not include CKD as a risk factor, while CKD is not less meaningful in this regard than encompassed DM.3, 8 This may lead to underestimation of its meaning.
Overhydration: An effect
Chronic kidney disease inevitably causes volume overload in more advanced stages. It leads to hypertension, arterial stiffness, the shift of the fluids to the third space, left ventricular hypertrophy, and the development of HF.9, 10 Volume overload is believed to be the most common mortality risk factor in CKD.10, 11 Hypervolemia is an adverse prognostic factor not only for patients undergoing renal replacement therapy, but also for those in pre-dialysis stadium. It is an independent risk factor for the development of cardiac dysfunction.12
Overhydration: A cause
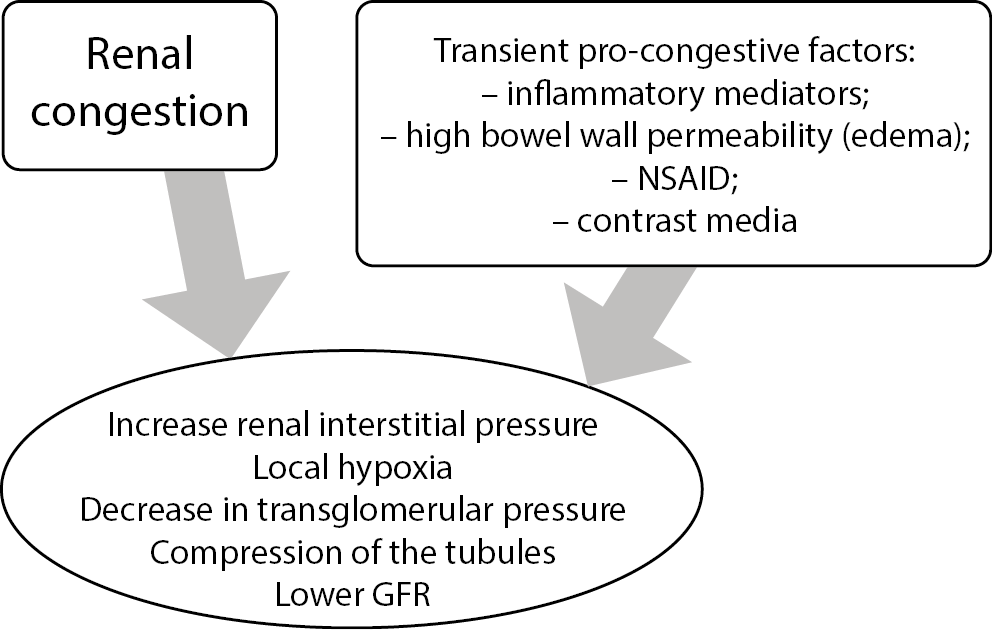
It is agreed that good delivery of blood to the kidneys is vital to preserve their function, but the proper outflow is not less important. “Congestive kidney failure”13 could be a good description of a pathology that takes place in patients with diastolic HF and CKD. It is believed that sodium retention is always connected to water expansion, a primary therapeutic target, and it increases central venous pressure (CVP). The association of CVP and kidney function impairment and higher all-cause mortality was described in a study where cardiovascular patients underwent the catheterization of the right heart.14 Although some doubts in blocking activated both renin–angiotensin–aldosterone system (RAAS) and sympathetic system exist, the need to lower CVP to treat congestion appears to be unquestionable.14 In chronic HF, diastolic dysfunction, which coexists with increased filling pressure, is often accompanied by kidney failure (Figure 1); the diastolic dysfunction progression worsens the mortality outcomes.15 The exact pathomechanism is yet not well understood, but the interstitial overpressure and venous congestion, described together as increased renal afterload, seems to play an important role.13 One must remember that transient exposition to inflammatory mediators, endotoxins or changed bowel wall permeability can be additional pro-congestive factors (Figure 2). Sometimes, a proper antibiotic course can break diuretic resistance by lowering inflammatory response.
Treatment
Regardless of the direction of interactions between overhydration and the kidneys, effective dehydration is crucial. The therapeutic strategy should be subtly tailored, because the window for fluid balance in cardiorenal patients is narrow. The universal recommendation for the majority of patients with CKD – low dietary sodium delivery – should be the first step.16 In more advanced CKD or HF exacerbation, such management is insufficient and pharmacological treatment should be introduced.
Loop diuretics
The most frequently used class of diuretics in HF are loop diuretics, which work in the loop of Henle; furosemide and torsemide are the most common among them. The inevitable, but also undeniable side effect of loop diuretics is the increased activity of sympathetic nervous system (SNS) and RAAS.
Loop diuretics relieve congestion but do not change mortality and rehospitalization rate in the short and long term.17 The decrease in its effect in the course of time is a common phenomenon, resulting in the reduction of sodium and chloride elimination; the causes are multiple. Pharmacodynamics and pharmacokinetics play an important role. Loop diuretics are highly bound to serum proteins and require secretion into the proximal tubule to be active. This Na/K/2Cl pump is located on the luminal side of the nephron. Therefore, loop diuretics must reach the tubular fluid to be active. Hence, in patients with severe renal insufficiency (e.g., GFR < 15 mL/min), larger doses of loop diuretics are required to achieve effective concentrations.18 The various loop diuretics differ in intestinal absorption, especially with an edematous bowel wall present in decompensated HF. Reduced drug excretion to the lumen of the tubule, reduced filtered load of sodium and increased RAAS and SNS activation due to HF-related underfilling and extracellular fluid volume (ECFV) depletion, which attenuates the peak effect of the diuretic, also contribute to the resistance.19, 20 The vasopressin release, enhanced by angiotensin II and baroreceptor-mediated mechanism in more advanced HF, increases nephron water reabsorption.21 The braking phenomenon, which impairs sodium loss in the course of time, distal tubular hypertrophy and hypochloremia, which plays a role in the activation of neurohormonal activity, can also contribute to diuretic resistance.22 The attempts to break the resistance encompass more frequent dosing, sometimes multiple times per day,22 as well as adjusting the dose to the renal function, intravenous administration of the drug and switching from furosemide to torsemide or bumetanide.16
The combination of different diuretics is another step to break the resistance. Blocking sodium reabsorption in different sections of nephron seems to be the key target.
Loop diuretic act from the lumen and are responsible for inhibition of Na/K/Cl2 cotransporter in thick ascending loop of Henle and macula densa. They inhibit the transporter by binding within the translocation pocket through the chloride-binding site. The use of loop diuretics leads to the increase of sodium concentration in distal parts of nephron and stimulates sodium reabsorption through the intensification of Na/Cl cotransporter activation, which is one of the resistance mechanisms.23
Thiazides and thiazide-like drugs also act from the lumen through the inhibition of Na/Cl cotransporter in distal convoluted tubule, and, as a result, decrease sodium reuptake. Although their effectiveness finishes with the drop in eGFR below 30 mL/min/1.73 m2, the combination of loop diuretics with thiazides to the certain point is justified.
Mineralocorticoid receptor antagonists (MRAs) – spironolactone and eplerenone – have been shown to improve morbidity and mortality in HF patients, for example in RALES study. They inhibit the aldosterone receptors in distal nephron and collecting tube, reducing Na channel and Na/K ATPase. One must remember that all the diuretics that target collecting tubule can cause hyperkalemia; therefore, they are not often used in HF patients. This, however, can be controlled by giving concomitant loop diuretic, oral bicarbonate (if acidosis is confirmed) or potassium binders in gut. Natriuretic doses need to be greater than 25 mg/day of spironolactone or 50 mg/day of eplerenone.
Tolvaptan
Tolvaptan, a selective antagonist for the vasopressin receptor V2, was investigated in the EVEREST trial to assess its usefulness for inpatient HF individuals. It diminished dyspnea, edema, body weight, and serum sodium level, but did not affect overall survival, mortality connected with CVD and hospitalizations for HF.24
Ultrafiltration
When diuretic response is poor or the resistance to diuretic therapy develops and the heart or kidney insufficiency deteriorates, ultrafiltration (UF) is a solution worth considering.25 In comparison to loop diuretics, it warrants decongestion with more efficient sodium loss and lower RAAS stimulation.19 Several randomized trials were conducted to investigate the potential benefits of UF strategy.
RAPID-CHF (Relief for Acutely Fluid-Overloaded Patients with Decompensated Congestive Heart Failure) and UNLOAD (Ultrafiltration vs Intravenous Diuretics for Patients Hospitalized for Acute Decompensated Congestive Heart Failure) trials showed superiority of UF in the loss of fluid over diuretic therapy.26, 27 In CUORE (Continuous Ultrafiltration for Congestive Heart Failure) trial, weight loss effect was similar in both UF and diuretic groups, but the increase in serum creatinine in the group treated with diuretics and a minor incidence of HF rehospitalizations in HF group were observed.28 CARRESS-HF (Cardiorenal Rescue Study in Acute Decompensated Heart Failure) study revealed similar weight loss effect in UF and diuretic patients, but also the worsening of serum creatinine results and greater adverse effects ratio in patients undergoing UF.29 Also, in AVOID-HF (Aquapheresis vs Intravenous Diuretics Hospitalizations for Heart Failure) study, greater adverse events ratio in UF patients were underlined, while no significant difference in three-month rehospitalization between the diuretic and the UF group was observed. The trial was terminated prematurely due to slow enrollment.30
Sustained low-efficiency dialysis (SLED) is carried out in patients with NYHA (New York Heart Association) class IV with good clinical effect. Although this solution requires central vascular access, it offers better hemodynamic stability in critically ill patients. The single SLED session, in comparison to conventional hemodialysis, lasts longer (6–12 h) and uses lower blood (50–200 mL/min) and dialysate (200–400 mL/min) flow. Fluid volume is removed slowly over a longer time, ensuring hemodynamic stability.31, 32
Peritoneal dialysis
The usefulness of peritoneal dialysis (PD) in the management of volume excess in patients with refractory HF and renal failure is justified by greater sodium removal compared with traditional diuretic strategies. Recently, both nephrologist and cardiologist appreciate this modality in CRS.33, 34
For CRS type 1, tidal PD was found to be a safe and effective mean of removing toxins and large quantities of excess fluid. In a small randomized study, it was superior to UF therapy when the preservation of renal function, improvement of cardiac function and net fluid loss was considered.35 Moreover, a higher rate of adverse events was noticed in the UF arm. A prospective Brazilian study also confirmed the high-volume PD (prescribed Kt/V = 0.50/session) as effective in CRS type 1 patients, allowing adequate metabolic and fluid control.
In chronic HF with CKD, when eGFR falls below 25 mL/min/1.73 m2, peritoneal UF may improve daily functioning and reduce hospitalizations. A systematic review prepared by Ronco group assessed the efficacy of PD in patients with refractory CHF.34 Twenty-one studies encompassed 673 patients; the authors suggested that in patients with refractory CHF, PD can be an effective and safe treatment option, leading to heart function improvement and better weight control.
Wojtaszek et al. showed the efficacy of peritoneal UF with nightly, 12-hour, 7.5% icodextrin exchange in the long-term treatment of refractory HF in the majority of NYHA class IV patients.36 One overnight icodextrin exchange appeared to be a promising therapeutic option as an adjunct to pharmacological management. The authors emphasized that the treatment can have a great impact on the quality of life and the total treatment costs.
In recent years, even more studies confirmed PD as a safe and feasible palliative treatment for refractory CHF in type 2 CRS.37 Pavo et al. analyzed multiple factors to identify patients with refractory HF and congestive right ventricular dysfunction who would benefit most from PD.38 Patients with more pronounced backward failure, less marked residual renal functional impairment and those not dependent upon others for assistance are likely to profit most from PD.
In summary, PD should be taken under consideration when refractoriness to conservative therapy appears. Typical clinical situations, such as persistent right heart congestion, ascites with intensified diuretic treatment, ≥2 hospitalizations within 6 months because of cardiac decompensation despite optimal medical treatment, or acute renal failure during intensified conservative treatment of cardiac decompensations are those in which PD should be considered.
The special group of patients: Diabetics
Diabetes mellitus is the most frequent cause of CKD in the world and is an independent death risk factor in the course of CVD.39 According to post hoc ACCORD trial analysis, in diabetic patients, CKD and/or CVD worsens the all-cause mortality and cardiovascular events prognosis.40 Therefore, modern anti-diabetic drugs are expected to be not only a good hypoglycemic effect, but also nephro- and cardioprotective result are expected.
Because of its beneficial characteristics, such as low price, safety of use and possible benefits on cardiovascular system, metformin is the first-line drug.41 It does not have to be ceased in CKD patients, even if the eGFR falls below 45 mL/min/1.73 m2, and, under appropriate surveillance, can be used up to 30 mL/min/1.73 m2.39 However, for the vast amount of patients, monotherapy is not sufficient. Taking into account the immense cardiovascular risk, the studies went in the direction of seeking more pleiotropic solutions. Two groups of medicines should be distinguished here: sodium-glucose cotransporter 2 (SGLT2) inhibitors and incretin agent – glucagon-like peptide-1 (GLP-1) receptor agonist.42 These novel antidiabetic drug groups reduce cardiovascular death risk, with SGLT2 inhibitors (SGLT-2i) influencing HF risk, and GLP-1RAs reducing MI risk.42
Sodium-glucose co-transporter-2 inhibitors
The EMPA-REG OUTCOME trial was a study focused on assessing cardiovascular endpoints. The researchers randomized over 7020 type 2 DM (T2DM) patients with high cardiovascular risk and examined the influence of empagliflozin. During the study, the primary outcomes, such as death from cardiovascular causes, non-fatal MI or non-fatal stroke, and all-cause mortality among patients receiving different doses of empagliflozin (an SGLT2 inhibitor) and placebo, were assessed. It was concluded that the administration of the SGLT2i, which acts in the proximal tubule by increasing glucosuria, in 2 doses, 10 mg and 25 mg, significantly reduced hard endpoints (by 14% and 32%, respectively) compared to placebo. The exact mechanism of action is not well understood, but the protective effect of the drug lies probably in changes in lipid levels and a reduction in the following: sympathetic activity, blood pressure, albuminuria, uric acid retention, oxidative stress, and insulin resistance.43, 44
Slowing down the progression of CKD was the additional hypothesized result of empagliflozin. The CKD patients in stages G1-3b were assessed for the eGFR during the trial and it was observed that after the initial transient drop in eGFR in the first 4 weeks of observation, the progression of the disease was stabilized in patients receiving empagliflozin, while eGFR declined over time in the group receiving placebo.45 The eGFR slope analysis from the EMPA-REG OUTCOME trial evaluated GFR variability in 3 different periods, that is, shortly after initiation of the therapy, during chronic maintenance period and after drug cessation. Their observations – eGFR relative decline in the 1st stage, inhibition of the eGFR loss in the 2nd period, and eventually eGFR increase in the last phase after drug cessation – support the hypothesis that there is a renoprotective effect of empagliflozin, reduction in intraglomerular pressure caused by the drug and the SGLT2i effect on hemodynamic changes and its reversibility.46
EMEROR Reduced [ejection fraction] trial assessed the effect of empagliflozin on HF, regardless of the diabetic status, on patients with ejection fraction of 40% and less. In this trial, 3730 patients with NYHA class II–IV HF were randomized into 2 groups – placebo and 10 mg empagliflozin once daily. It turned out that the patients receiving empagliflozin had lower risk of meeting primary outcomes, such as cardiovascular death or hospitalization for HF, than the patients receiving placebo (19.4% compared to 24.7%). The study showed not only a positive cardiovascular, but also renal effect of empagliflozin. The secondary outcome, the rate of the decline in the eGFR over the duration of treatment period, was slower in the empagliflozin group than in the placebo group (−0.55 mL/min/1.73 m2 compared to −2.28 mL/min/1.73 m2). Also, a composite renal outcome, that is, the beginning of renal replacement therapy (chronic dialysis or renal transplantation) or a profound, sustained reduction in the eGFR occurred in less patients in the empagliflozin than in the placebo group (1.6% compared to 3.1%). It is worth mentioning that 48% of enrolled patients had eGFR of less than 60 mL/min/1.73 m2.47
The effect of empagliflozin on patients with HF with preserved ejection fraction is still being studied in similarly designed EMPEROR Preserved [ejection fraction] trial.
Empagliflozin is beneficial for CKD patients because of the influence on fluid management. This is important, as CRS frequently coexists with overhydration. Positive impact of empagliflozin on HF with both preserved and reduced ejection fraction (HFpEF and HFrEF) and hypertension is explained by natriuresis enhancement.48 The resistance to physiological natriuretic peptide and administered diuretics in HF is common; the sodium-hydrogen antiporter 3 or, in other words, sodium-hydrogen exchanger (NHE 3), in the proximal renal tubule, whose activation increases in HF, is hypothesized to be the culprit.49 The NHE3 protein, which co-localizes with SGLT2,50 is responsible for the majority of sodium reuptake in the tubules. The SGLT2i interacts with NHE3 by its inhibition and causes natriuresis. The simultaneous administration of loop diuretics can prevent the compensative increase in sodium reuptake in different parts of the nephron.51
Another proposed mechanism for empagliflozin efficacy is the hypothesis of renal congestion and SGLT2i grip point focused on intracellular water. Because of local SGLT2i-induced hypovolemia in the kidney, the interstitial fluid passes easily to the vascular space to compensate for the fluid loss. Due to this assumption, the circulating volume, and thus, the organ perfusion and arterial filling, would not be affected. In addition, the interstitial renal volume overload reduction decreases and protects renal function.52
The other gliflozin worth mentioning is dapagliflozin. DECLARE-TIMI 58 (Effect of Dapagliflozin on the Incidence of Cardiovascular Events – Thrombolysis in Myocardial Infarction 58) trial was established to define the dapagliflozin cardiovascular safety profile. In this trial, 17,160 T2DM patients were randomized into groups receiving the drug or placebo. Dapagliflozin treatment lowered the rate of HF hospitalizations and cardiovascular deaths, but did not influence major adverse cardiovascular events (MACE), defined as cardiovascular death, MI or ischemic stroke, in the group of patients with primary atherosclerotic CVD or at risk of one.53 Promising results were observed in Dapagliflozin and Prevention of Adverse outcomes in Chronic Kidney Disease (DAPA-CKD) phase III trial, focused on primary renal outcomes. In DAPA-CKD trial, the composite endpoint (≥50% sustained decline in eGFR, onset of ESKD, or cardiovascular or renal death) in a comprehensive group of CKD patients with eGRF of 25–75 mL/min/1.73 m2 with and without DM had been assessed.54 At the end of March 2020, the trial was terminated prematurely due to the overwhelming efficacy in renal insufficiency patients. Similarly designed EMPA-Kidney trial is being conducted, but the results we will be available in 2022.
Other SGLT2i were also assessed in the trials. Focused on cardiovascular outcomes, CANVAS (Canagliflozin Cardiovascular Assessment Study) program encompassed 2 trials of 10,142 T2DM patients with high CVD risk. It was designed to assess the canagliflozin benefit–risk balance. The program showed the superiority of SGLT2i over the placebo group in cardiovascular events reduction, but the adverse effects – amputations at the level of toe or metatarsal – was more frequent in the canagliflozin group.55 CREDENCE (Canagliflozin and Renal Endpoints in Diabetes with Established Nephropathy Clinical Evaluation) study was focused on canagliflozin primary renal outcomes. Included patients had eGFR of 30–90 mL/min/1.73 m2 and an estimated daily proteinuria at the level of 0.3–5 g. The study showed that both kidney failure and the risk of cardiovascular events were lower in the group receiving canagliflozin compared to the placebo group. The study was ceased after planned interim analysis, as the demonstration of efficacy was obtained already after randomizing 4401 patients.56
The summary analysis of the SGLT2i mentioned above is as follows: empagliflozin, canagliflozin and dapagliflozin were recently collected in a meta-analysis. It was concluded that SGLT2i reduce HF hospitalizations, for which the evidence is the strongest, MACE and all-cause mortality. The evidence is weaker in the group of patients with eGFR < 60 mL/min/1.73 m2; yet, the impact on lowering adverse renal effects is observable even in this group of patients.57
Among the desirable features of SGLT2i, weight loss, blood pressure lowering, uricosuric effect, and lack of the hypoglycemia risk are worth mentioning. Gliflozins reduce hyperfiltration in the early stages of diabetic nephropathy, as they restore tubuloglomerular feedback (TGF).58
CVD-REAL3 was an observational study to assess the SGLT-2i effects on kidney function in comparison to other glucose-lowering drugs in real-world clinical practice. It was concluded that initiation of the therapy with SGLT2i, that is: dapagliflozin, empagliflozin, canagliflozin, ipragliflozin, tofogliflozin, and luseogliflozin, was associated with a lower risk of major kidney events and a slower decline in kidney function than the initiation of other anti-diabetic medications.59
The recommendations of American Diabetes Association suggest that second-line DM therapy should be chosen on the basis of the existence of atherosclerotic CVD (ASCVD), HF or CKD comorbidity. Heart failure or CKD predominance implies SGLT2i usage to reduce CKD progression and/or HF, while ASCVD predominance suggests SGLT2i or GLP-1 receptor agonist usage with suspected cardiovascular benefits.60, 61
Incretin drugs
Glucagon-like peptide-1 (GLP-1) receptor agonists are the other anti-hyperglycemic agents that reveal nephroprotective properties, as they prevent macroglobinuria occurrence and the impairment in renal function.62 The putative renoprotective effect of the GLP-1 receptor agonists is direct and indirect (dichotomic). Indirect actions consist, among others, of better glycemic and blood pressure control, weight loss, and improvement in coronary flow and left ventricular wall motion. Direct effects encompass the decrease in renal and systemic inflammation, reduction of oxidative stress, decrease of renal hypoxia, and sodium and water modulation.63 Natriuresis seem to be the main mechanism of decreasing overhydration. It was hypothesized that natriuretic effect of GLP-1 primarily involves inhibition of NHE3 activity in the brush border of the renal proximal tubule.64 Other authors, who studied the impact of GLP-1 agonists on healthy saline loaded men, suggest that the natriuretic effect is rather induced through a tubular mechanism in distal nephron segments. This mechanism is believed to be secondary to suppression of angiotensin II (ANG II) and independent of renal hemodynamics. Such hypothesized mechanism of action supports the existence of a GLP-1–renal axis.65 Improving tubuloglomerular feedback and ameliorating glomerular hyperfiltration in patients with T2DM would also be anticipated.64
In LEADER (Liraglutide Effect and Action in Diabetes: Evaluation of Cardiovascular Outcome Results) trial, 9340 diabetic patients were randomized. The time-to-event of death from cardiovascular causes, nonfatal MI or nonfatal stroke was assessed. The results showed that the time to the first occurrence of the event was shorter in liraglutide than in the placebo group.66 There were also other trials evaluating GLP-1 agonists, like ELIXA67 with lixisenatide, SUSTAIN-668 with semaglutide, AWARD-769 with dulaglutide, EXCEL70 with exenatide, and LIRA-RENAL71 again with liraglutide. Although there is evidence for GLT-1 agonists to reduce albuminuria, the evidence for hard renal endpoints are still to be found.62 Surprisingly, in contrast to SGLT2, there are no GLP-1 receptors in the renal tubuli or other kidney compartments.
Summary
Cardiovascular disease resulting in HF is a common CKD complication, but cardiac insufficiency can also cause renal function deterioration. Cardiorenal syndromes present a common symptom: overhydration. Because the therapeutic target is not always met by means of classical loop diuretics because of resistance, alternative management of fluid overload is required. Ultrafiltration reduces symptoms effectively, but there is no evidence that it improves long-term survival. Peritoneal dialysis seems an attractive solution in refractory CRS type 2 and in selected patients in CRS type 1 treatment. Tolvaptan turned out to diminish the symptoms, but did not prove to influence hard endpoints. The interesting therapeutic option emerges for diabetic and non-diabetic patients with CRS. The SGLT2i are particularly noteworthy, showing reno- and cardioprotective effects, for instance, by reducing overhydration (mainly by NHE3 inhibition). Liraglutide, a GLP-1 receptor agonist, probably shares the NHE3 inhibition effect with SGLT2i and also has cardio- and renoprotective properties.
Although the new agents seem promising in overhydrated patients with CRS, further studies are required to thoroughly assess their potential. To date, the management of CKD patients with HF and fluid overload remains challenging.