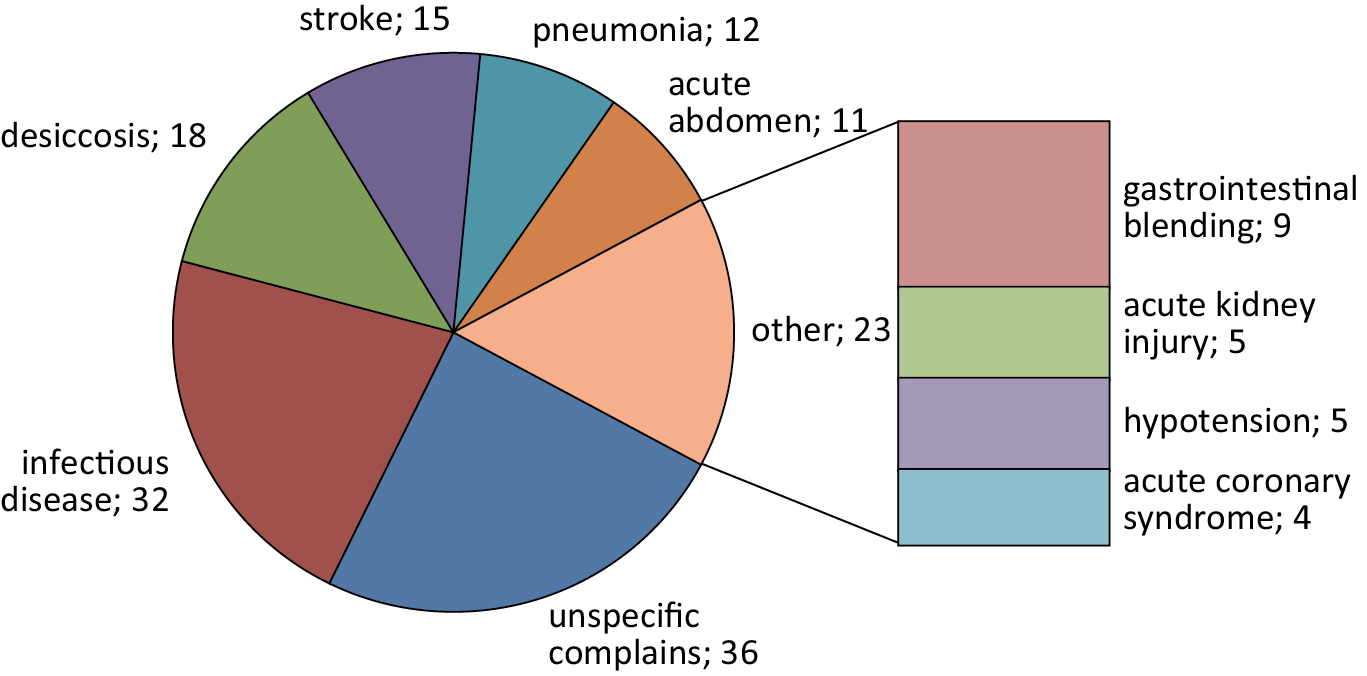Abstract
Background. Early recognition of sepsis and a prompt initiation of goal-directed therapy is important for sepsis survival. Little is known about the impact of early recognition of sepsis in the out-of-hospital setting when paramedics are the 1st medical professionals arriving on the scene.
Objectives. To characterize the impact of sepsis recognition by paramedics in the 1st out-of-hospital contact and to establish a predictive model by combining preclinical patient characteristics.
Materials and methods. In this retrospective single-center cohort study, we included a total of 263 patients diagnosed with sepsis after admission to the emergency department and correlated them to the emergency medical protocols of the paramedics who have seen the patient out-of-hospital.
Results. Only 25 patients were correctly diagnosed by paramedics out-of-hospital. If sepsis was diagnosed, the median time to antibiotic administration was significantly lower (136.50 min compared to 206.98 min, p = 0.0069) and mortality was reduced from 22.8% to 8% (p = 0.0292). We have identified predictors for prognosis and calculated a predictive model with a modified quick Sepsis-related Organ Failure Assessment (qSOFA) score, which fits the needs for out-of-hospital usage and results in a better discrimination of vitally threatened patients (receiver operating characteristic (ROC) area under curve (AUC) of 0.641 compared to 0.719), as compared to the standard qSOFA.
Conclusions. Sepsis recognition by paramedics at the 1st out-of-hospital contact significantly reduces sepsis mortality. The qSOFA and modified qSOFA are suitable tools for sepsis recognition, and have an impact on mortality and disease management when used.
Key words: paramedic, emergency medicine, qSOFA, sepsis, out-of-hospital
Background
Sepsis is a severe disease associated with high rates of mortality and morbidity.1 The treatment of septic patients is often expensive.2 Moreover, despite advances in intensive care, the diagnosis of sepsis and its underlying disease remains challenging and time-consuming.3 The availability of an early predictive model of sepsis for the initiation of goal-directed therapy is necessary. Guidelines from the Surviving Sepsis Campaign have helped to establish structures for the successful treatment of sepsis, especially in hospitalized patients.4 However, only limited capabilities exist in preclinical emergency services for diagnostic procedures.5
In some projects, so-called sepsis kits have been established to improve sepsis survival in a preclinical setting. However, randomized controlled trials evaluating these kits are not sufficiently available and have been difficult to establish.6 The quick Sepsis-related Organ Failure Assessment (qSOFA) score has been developed recently using guidelines for the rapid assessment of septic conditions. This scoring system uses respiratory rate, disturbance of consciousness, and systolic blood pressure (SBP) as parameters. They can be easily assessed during preclinical emergency service.7 There is an ongoing scientific debate on the performance of this score as a predictor of outcome.8 Other scoring systems like the Mortality in Emergency Department Sepsis (MEDS) score and the Modified Early Warning Score (MEWS) may perform better, but are more difficult to establish under preclinical conditions.9, 10 The qSOFA criteria might be also useful in some differential diagnostic aspects that are important for preclinical medicine.
The treatment outcome parameters for sepsis patients are established and described. Among these, the time to antibiotic treatment, lactic acid levels and hemodynamic parameters are the most important.11 Research in the field of sepsis at this interface is nearly non-existent for out-of-hospital emergency service systems, thus further research is needed in this area.12 The development of a risk assessment tool for death in patients with preclinical sepsis may be helpful in identifying sepsis patients at the earliest possible time point, and aid in providing adequate and rapid diagnostics work-up and surveillance.
Objectives
It was hypothesized that early sepsis recognition by paramedics at 1st out-of-hospital contact would have an impact on the mortality and morbidity of sepsis. We have also searched for predictive parameters that can be assessed by paramedics, and aimed to establish a predictive model for sepsis prognosis by combining out-of-hospital patient characteristics.
Materials and methods
Study design
This study is a retrospective single-center study. Sepsis survivors and non-survivors who were rushed to the hospital by ambulance were identified and used as the 2 cohorts for this study.
Setting
Sepsis patients interacting with emergency medical services (EMS) in the county of Northeim, Germany and the Helios Albert-Schweitzer-Hospital, Northeim, Germany, were considered for inclusion in the study.
Participants
The patient group was identified by hospital release documentation ICD-10 code A41 (sepsis) and R65 (systemic inflammatory response syndrome – SIRS) after final release or death in hospital. Data were collected from the clinic information system (CIS) of the hospital and the records of the EMS. A total of 263 patients were identified. Data were obtained from the period of January 1, 2012 to December 31, 2018. The exclusion criteria were age under 18 years and missing documentation in the CIS system. All parameters were obtained according to the Declaration of Helsinki and to the rules of the European Union. The study was approved by the ethics committee at the University of Göttingen (approval No. 4/8/17). Data were collected in a Microsoft Excel 2013 (Microsoft Corp., Redmond, USA) table and pseudonymized.
Variables
The primary outcome parameters for mortality rate were death in hospital and length of hospital stay for morbidity rate. The secondary parameters were as follows: age, sex, coronary heart disease, diabetes, residential care, hypertension, chronic kidney disease, liver cirrhosis, malignancy, immunodeficiency, septic urinary tract infection, septic pneumonia, other foci, Allgower’s index, preclinical respiratory rate >22/min, preclinical heart rate, preclinical Glasgow Coma Scale (GCS) <14, preclinical lowest SBP, preclinical qSOFA, preclinical O2 saturation, preclinical temperature >38.5°C, preclinical diagnosis of sepsis (yes or no), hospital respiratory rate >22/min, hospital heart rate, hospital O2 saturation, hospital GCS <14, preclinical lowest SBP, hospital qSOFA, length of intensive care stay, length of hospital stay, death in hospital, denial of resuscitation order, preclinical antibiotics, hospital antibiotics, time of antibiotics administration, MEWS, Manchester triage category, laboratory tests like thrombocyte count, bilirubin, creatinine, lactic acid level, high sensitive troponin, leucocyte count, procalcitonin and blood culture results, need for dialysis, and type of antibiotic treatment.
Sample size
The required sample size was not calculated since this was a retrospective proof of concept study.
Statistical analyses
Descriptive statistics were calculated using IBM SPSS v. 27 (SPSS Statistics for Windows; IBM Corp., Armonk, USA). Values are presented as number (n) and percentage, means ± standard deviation (SD), or median ± interquartile range (IQR). Detailed explanations are included in the figure captions.
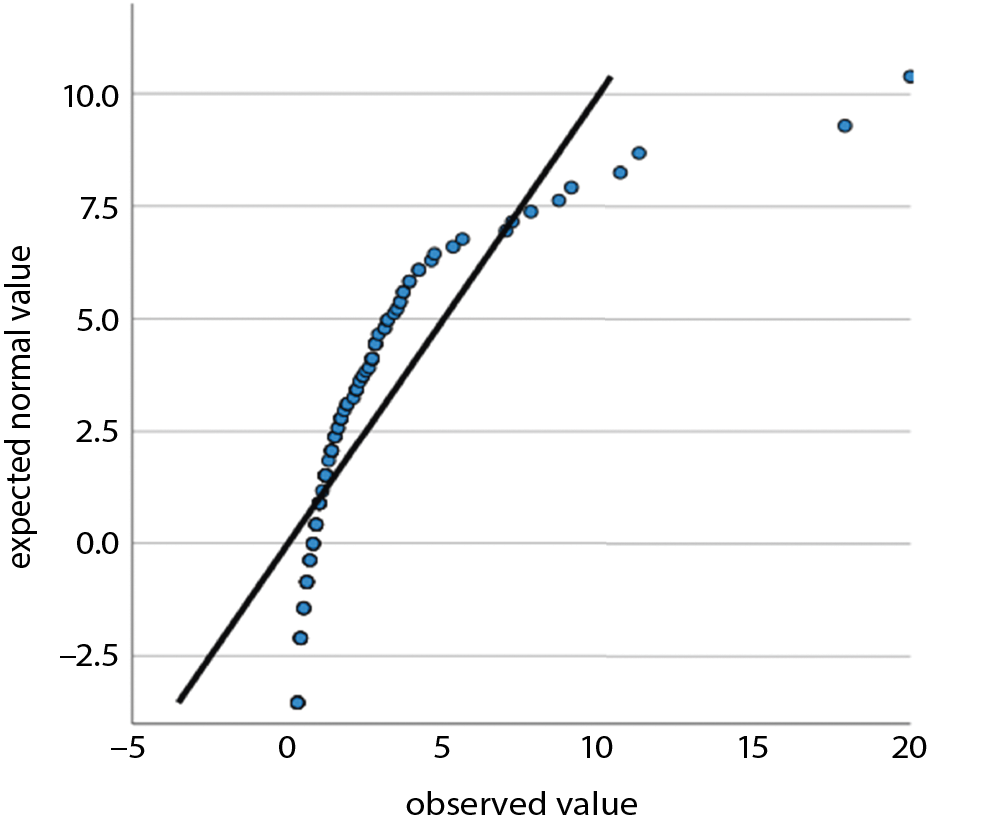
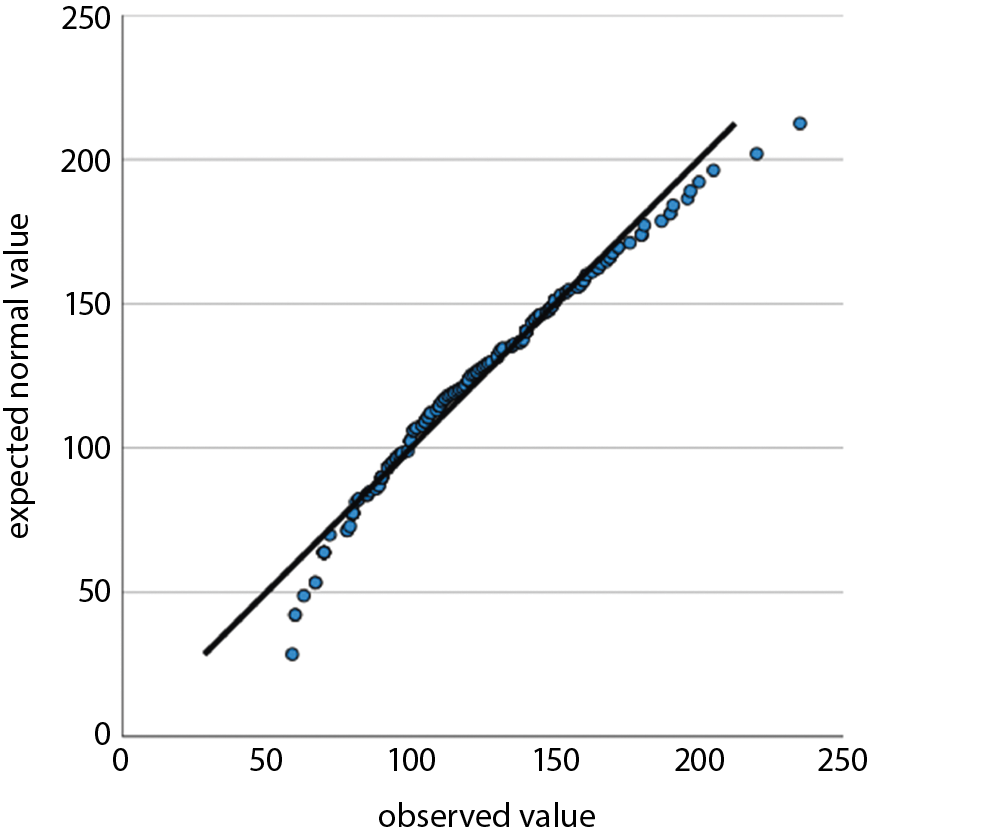
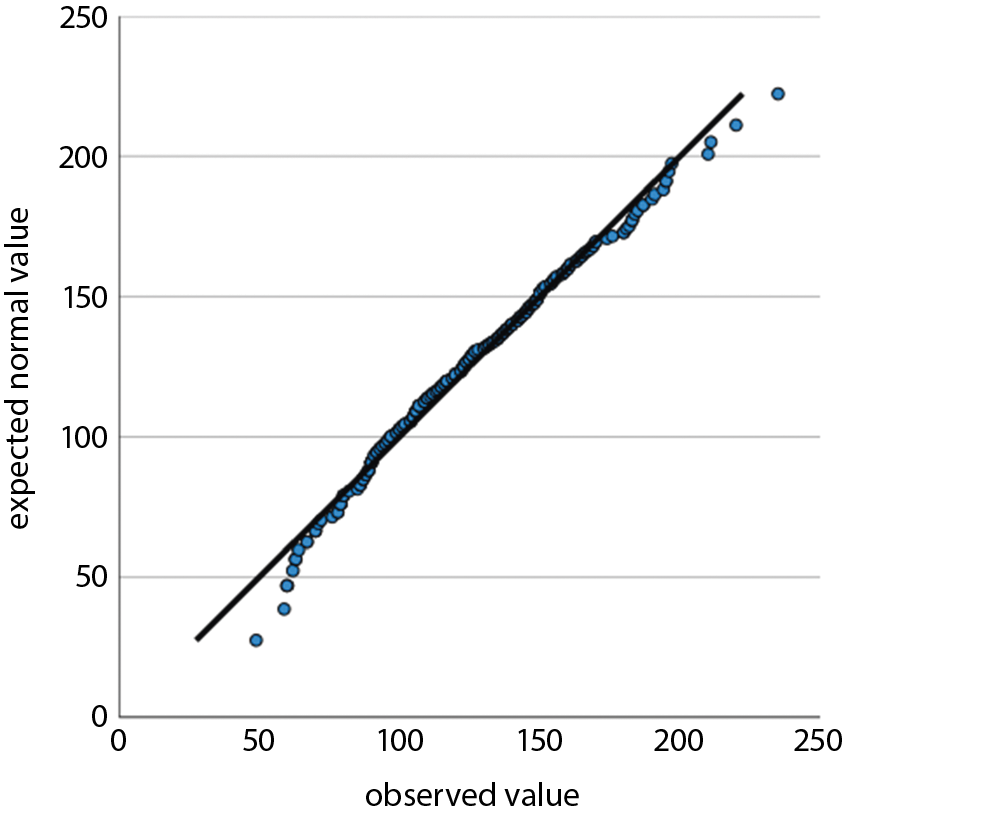
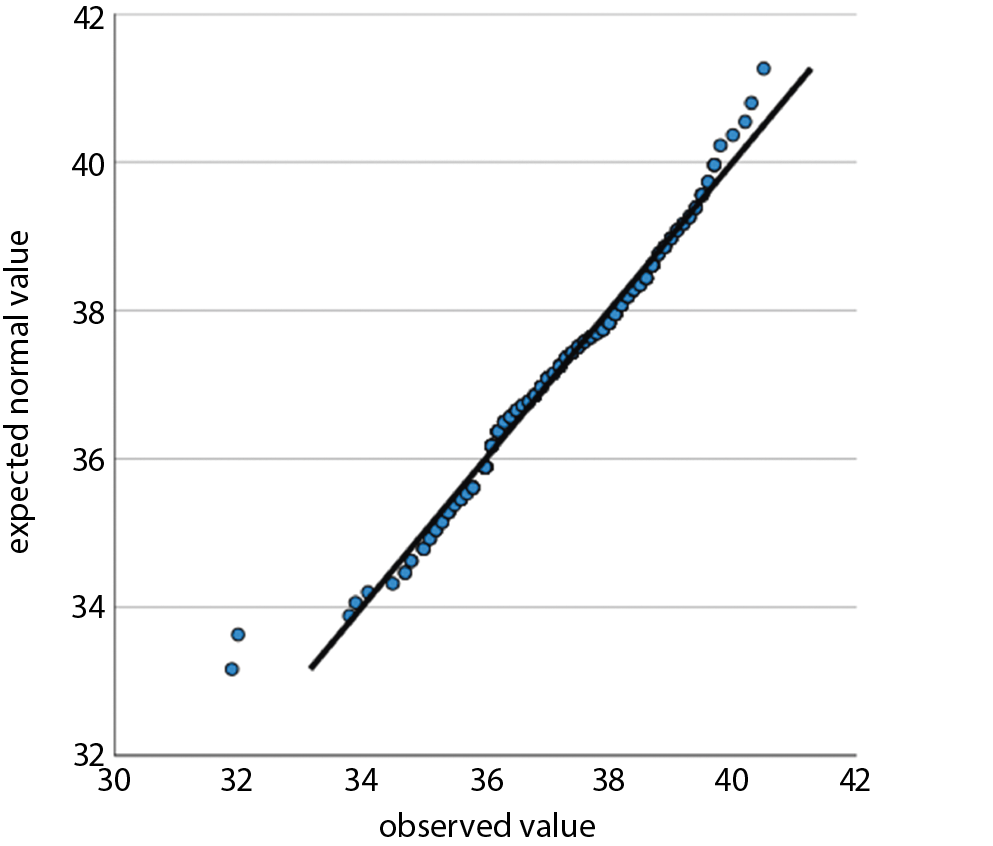
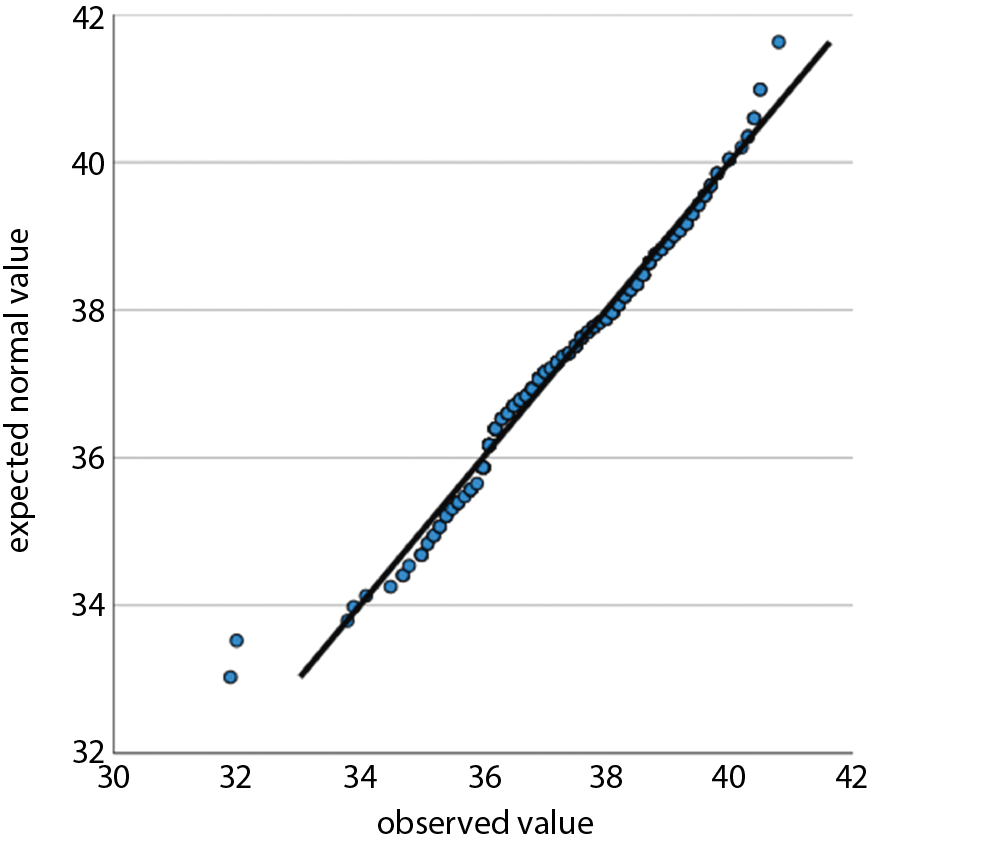
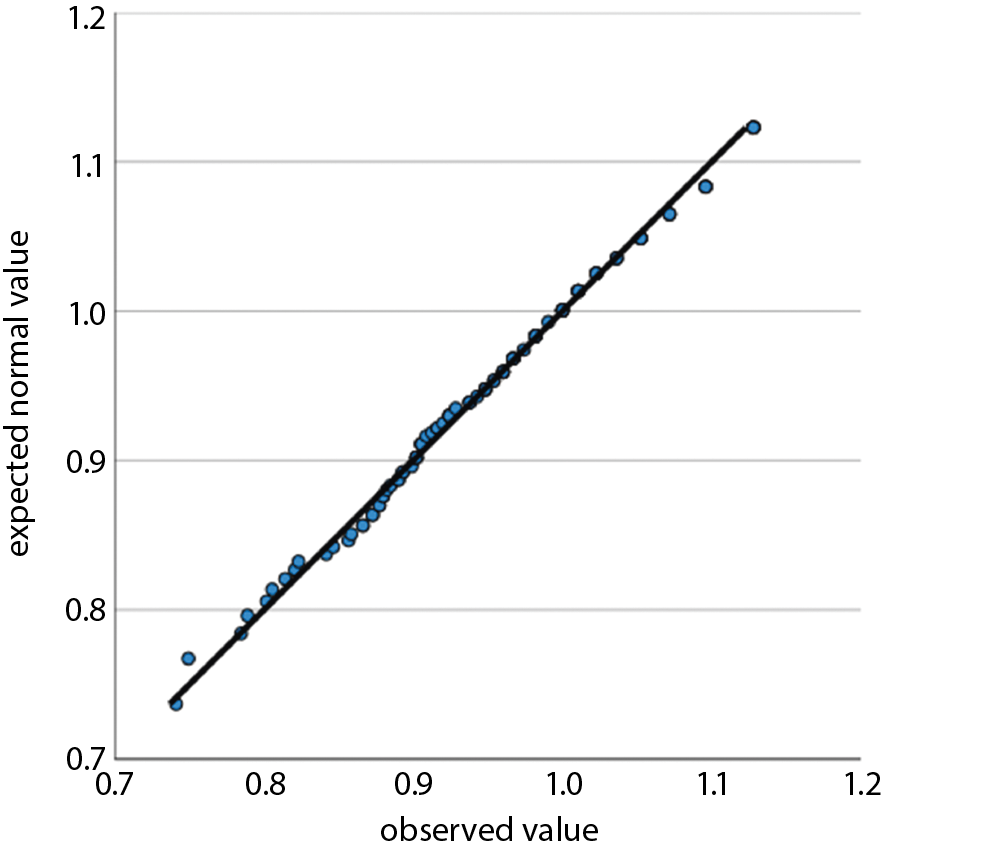
For tests of statistical significance, t-tests were performed, with a p < 0.05 considered significant. For comparisons of n in contingency tables, χ2 tests were performed where applicable, with a p < 0.05 considered significant. We used Q–Q plots to determine a normal distribution for the following variables: age, preclinical SBP, emergency department SBP, preclinical temperature, and emergency department temperature. Non-normality was seen for lactate. After Box–Cox transformation with lambda = 0.1, a normal distribution was achieved and a two-tailed unpaired t-test was performed. For emergency department vigilance, a non-normal distribution was seen. After Box–Cox transformation with lambda = 6.7934, a normal distribution was approximated, but it still did not satisfy the Shapiro–Wilk test. Therefore, a Mann–Whitney U test was used to test for significance in this case.13
A receiver operator characteristic (ROC) curve analysis was performed to calculate the correlations of qSOFA and our modified qSOFA with death due to sepsis. Points on the ROC curve nearest the upper left corner were chosen for cut-off values as this resulted in optimal sensitivity and specificity. The predictive model Psepsisdeath, indicating the probability for sepsis death by means of multivariate logistic regression, was calculated separately.13 The Psepsisdeath was calculated by using variables in our modified qSOFA with the following parameters: age, preclinical GCS, and preclinical SBP. For immunodeficiency, 1 indicated immunodeficiency and 0 indicated no immunodeficiency. The predictive factor X was generated as the weighted sums of the predictive factor values, weighed with their regression coefficients from the final binary logistic regression analysis13 (algorithm: X = 0.024 * [age] – 0.088 * [out of hospital GCS] – 0.021 * [out of hospital SBP] – 0.558 * [1 if immunodeficiency is positive, otherwise 0] + 0.430). The probability of sepsis death (Psepsisdeath) was calculated as follows (Equation 1):
For boxplots of predicted vs real mortality, we grouped patients into quartiles based on their individual mortality likeliness.
Results
We identified 263 sepsis patients who were seen between 2012 and 2018 by the EMS and brought to the emergency department. Sixty-one of them died of sepsis and 202 survived. Between these 2 groups, no differences were found for sex distribution, coronary heart disease, diabetes, dementia, residential care, hypertension, chronic kidney disease, dialysis, liver cirrhosis, or cancer history. Sepsis death was associated with age (83.10 compared to 79.51 years, p = 0.0385; Table 1) and the presentation of acute kidney injury (77.7% compared to 61.98%, p = 0.0346). For immunodeficiency status, a strong positive tendency was found, but it did not reach significance (11.5% compared to 21.8%, p = 0.0743; Table 1). The mean out-of-hospital qSOFA was 1.49 in the non-survivor group and 0.9 in the survivor group (p < 0.0001). It was also found that preclinical SBP (107.61 mm Hg compared to 124.37 mm Hg, p < 0.0001) and preclinical body temperature (36.53°C compared to 37.46°C, p < 0.0001; Table 1) were significantly lower in the sepsis death group. No differences were seen in preclinical average blood pressure (BP), heart rate, vigilance, respiration rate, or oxygen saturation. Interestingly, these findings were also true for the vital parameters taken in the emergency department, but a significant signal for vigilance, as measured with the GCS (12.80 compared to 13.68, p = 0.0048; Table 1), was observed between non-survivors and survivors.
Regarding the reason for sepsis (urinary tract sepsis, pneumonia, abdominal infection, other reasons), no significant differences were found between the sepsis death group and the survival group. A total of 114 positive blood cultures and 149 negative blood cultures were found. The main disease-causing agent was Escherichia coli, followed by Staphylococcus aureus and Klebsiella spp.
The laboratory parameters taken in the emergency department revealed a positive association with sepsis death and lactic acid levels (3.88 mmol/L compared to 2.02 mmol/L, p = 0.0008; Figure 1), whereas no correlation was found with thrombocyte levels, leucocyte levels, bilirubin concentration, creatinine levels, C-reactive protein (CRP) levels, or increased procalcitonin levels.
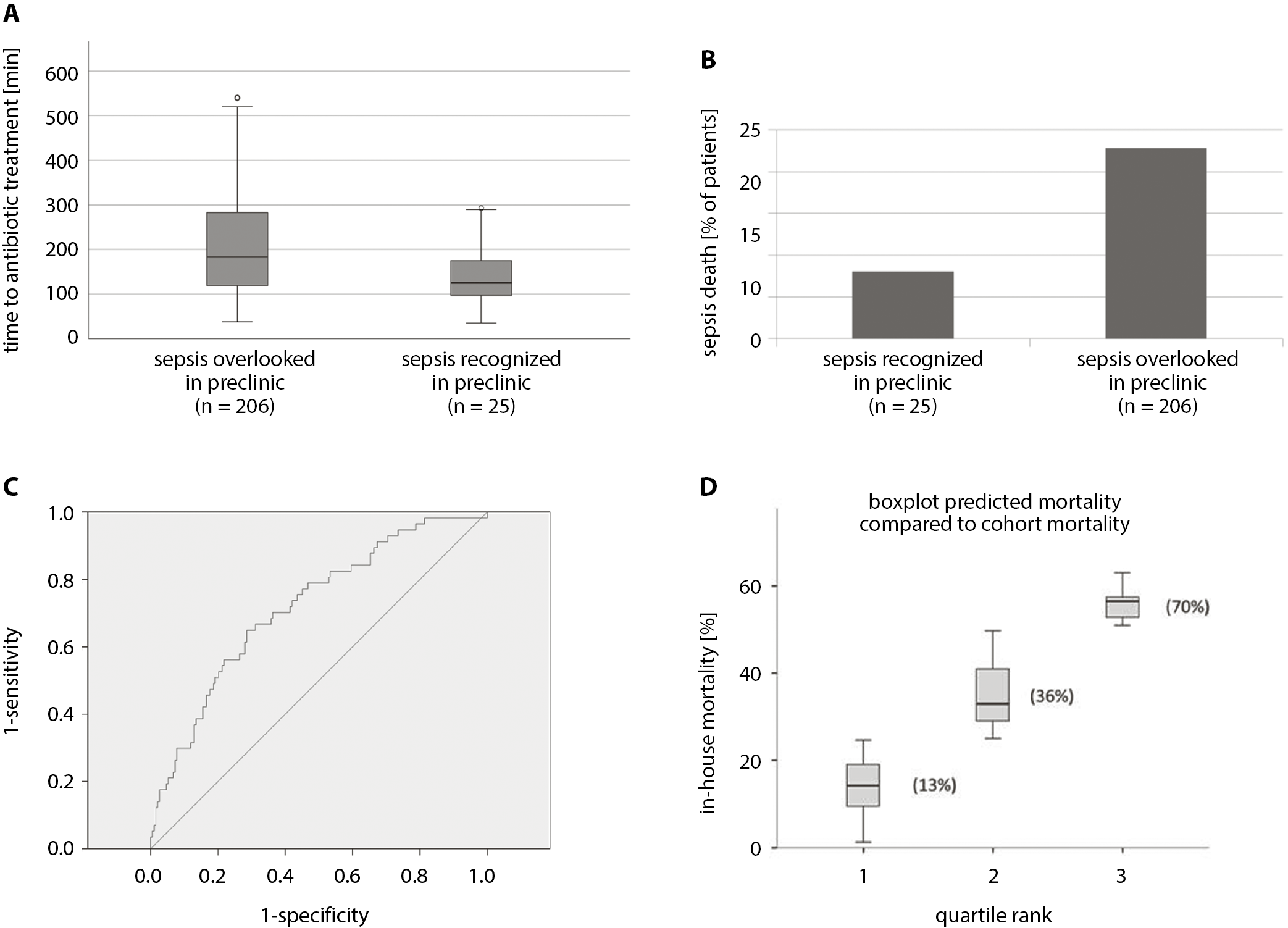
Sepsis was recognized by paramedics in only 25 patients and 238 did not have a suspected diagnosis of sepsis by EMS paramedics (Table 2). If sepsis was diagnosed by EMS, a noteworthy decrease in time to the first antibiotic administration as compared to a non-diagnosed sepsis was found (136.50 min compared to 206.98 min after the arrival of EMS on the scene, p = 0.0069; Figure 1). A remarkable increase in mortality was also found if sepsis was not initially diagnosed by EMS personnel (8% compared to 22.8%, p = 0.0292) before hospital admission (Figure 1).
If sepsis was not suspected, several misleading diagnoses were documented by paramedics upon delivery to the emergency department (Figure 2). The most often diagnosis was “unspecific complaints,” followed by “unspecific infectious disease” and desiccosis (Table 3).
Interestingly, most patients were transported by a type B ambulance (emergency ambulance) according to Euronorm (EN) 1789 (48%). Only 31% of patients were transported by a type C ambulance (mobile intensive care unit; EN 1789) and 21% had a type C ambulance accompanied by an emergency physician.
The duration of stay in the intensive care unit (ICU) did not differ between non-survivors and survivors, and no significant difference was seen after subdivision of the preclinical qSOFA 1 or 2–3 (3.5 compared to 5 days, p = 0.109). There was only a tendency for longer stays with a higher qSOFA (>1) on admission.
The out-of-hospital qSOFA and in-hospital mortality were correlated using a ROC. In our cohort, qSOFA correlated with an area under curve (AUC) of 0.641 (95% CI = 0.559–0.723) for predicting sepsis death.
If we use a modified sepsis score (with parameters age, out-of-hospital GCS, out-of-hospital SBP, and presence of immunodeficiency – Figure 2), the AUC was more exact with 0.719 (95% CI = 0.644–0.794), as compared to the original qSOFA. We have chosen to implement the aspect of immunodeficiency since this parameter showed a strong tendency, but lacked significance (p = 0.0743).
Next, we checked the abovementioned modified sepsis score (Figure 1C) by calculating the individual probability of sepsis death. Three groups were formed according to the mathematical division into quartile ranks (1st quartile – death probability 0–25%, 2nd quartile – death probability 25–50%, 3rd quartile – death probability 50–75%; Figure 1D). Since the 4th quartile (death probability 75–100%) encompassed no patients, it was excluded. The results of this calculation based on the abovementioned parameters are shown as boxplots representing the mean and IQR (25–75%) of the corresponding box for the specific quartile rank; therefore, representing the probability of death due to sepsis. The actual measured mortality in our cohort is shown in brackets (Figure 1D). A very good prediction of the real in-hospital mortality was found when the modified version of the out-of-hospital sepsis score was used.
Discussion
Our main finding was that, if sepsis was suspected by EMS, an improved outcome was detected. This finding is consistent with previously published data.14 On the other hand, there are reports that did not detect an improved outcome even if EMS was involved in sepsis cases.15 The reasons for these conflicting results remain unclear. One could speculate that situational awareness might play a role. The qSOFA is therefore a useful tool to establish this awareness in the out-of-hospital context. Further investigation after implementation of the so-called sepsis bundles and routine qSOFA scoring in the preclinical setting might help solve this problem.
In the majority of cases, sepsis was not recognized by EMS personnel. Mainly, unspecific complaints were given as admission diagnoses on arrival at the emergency department. It is well known that especially elderly patients being brought to emergency departments with unspecified complaints in 60% of cases have a serious disease.16 The most frequent reasons for misdiagnosis in the geriatric setting are concealed or less typical symptoms, multi-morbidity, as well as communication problems.17 In this context, it is not surprising that most patients were transported with type B EN1789 patient transport ambulances.
The overall sepsis mortality rate in the current cohort between 2012 and 2018 was 23.2%. Stevenson et al.18 reported a mortality rate of 29% for severe sepsis in the decade 2010–2020. Therefore, it can be concluded that the current cohort was not undertreated; in fact, these patients performed better than average. Looking at the demographics, it was found that age, acute kidney injury and immunosuppression (defined as any state of ongoing antitumor chemotherapy, medical immunosuppression, and hereditary or acquired immunodeficiency) influenced sepsis mortality. These findings are consistent with literature.19 A positive correlation of out-of-hospital qSOFA and emergency department qSOFA with mortality was found, which is also consistent with literature.20 A preclinical normal or slightly lowered body temperature (mean 36.4°C) was associated with sepsis death, as compared to a normal or slightly elevated body temperature (mean 37.5°C). While the role of fever in sepsis has been widely discussed and sparked controversy, the impact on mortality is generally low.21 Kushimoto et al.22 found that body temperature lower than 36.5°C to be associated with sepsis death, which is in accordance with our data.
Interestingly, a signal for the parameters average BP and GCS in the emergency department was found, which was not detected during patient transport. This time-dependent phenomenon shows the dynamic status of septic shock and is known in literature.23
The reasons for sepsis and sepsis death, as well as blood culture results, did not differ between the survivor and non-survivor groups, therefore excluding intensive care treatment bias in our cohort.
Elevated lactic acid levels were associated with death in the current cohort, which has also been described in the literature.24 The data showed no significant difference in the duration of stay in ICU between the non-survivors and survivors. Patients with a higher qSOFA had only a tendency for longer stays. Not surprisingly, survivors had a longer hospital stay as compared to patients in the non-survivor group. Again, this finding is consistent with previously published data.25 The ROC analysis revealed a good correlation of qSOFA with sepsis death in the current study. Brink et al.26 recently published a qSOFA AUC with similar results; therefore, we are confident that our data is reliable.
In the current study, the qSOFA was modified by integrating easy to determine preclinical parameters like age, GCS, SBP, and anamnesis of immunodeficiency. ROC analysis revealed an even better correlation of these preclinical parameters with in-hospital death as compared to the original qSOFA. Therefore, a relatively exact prediction model for the current cohort was established. With this score model, a preclinical tool could be developed that allows for increased awareness of a septic condition and might enable prompt medical treatment, critical for sepsis treatment. We plan to establish a web-based calculator, which might help to further evaluate this score and could be a helpful tool for out-of-hospital sepsis recognition. Further evaluation may have a clinical impact on in-hospital mortality. The EMS in Northeim had already introduced a sepsis bundle in 2018 after obtaining our results. Further scientific evaluation of the future developments may be interesting.
Limitations
Our study is limited by the retrospective single-center design. Therefore, our data are not able to prove that the recognition of sepsis by paramedics will always improve sepsis outcome. Multicenter randomized trials are needed to provide further evidence. We did not calculate the required sample size, since this was a proof of concept study. Thus, the current study may be underpowered.
We identified our cohort retrospectively by selecting cases that were documented with ICD-10 codes A41 and R65 as the main diagnoses. These diagnoses are based on systemic inflammatory response syndrome (SIRS) criteria. One might criticize that this is a selection bias, since qSOFA-positive patients who did not fit SIRS criteria might have escaped our attention. Although we cannot exclude selection bias, recent work27 showed that SIRS criteria are superior to the qSOFA in their ability to identify sepsis cases.
Conclusions
Preclinical recognition of sepsis by paramedics may result in an improved clinical outcome. Our data may help improve preclinical sepsis recognition by paramedics. Although further investigation is needed, our modified scoring system could be a promising tool.
Supplementary files
We have examined all the metric variables for normal distributions by using Q–Q plots. Normal distributions were seen for age, preclinical SBP, emergency department SBP, preclinical temperature, and emergency department temperature. Non-normality was seen for lactic acid (lactate). After Box–Cox transformation with lambda = 0.1, normal distribution was achieved and a two-tailed unpaired t-test showed a p = 0.0008. For emergency department vigilance, a non-normal distribution was seen. After Box–Cox transformation with lambda = 6.7934, a normal distribution was approximated but did not satisfy the Shapiro–Wilk test. Therefore a Mann–Whitney U test was used to test for significance. All results from regression analysis and Q–Q plots as well as and transformation results are provided as supplementary file.
Table S1. Statistical analysis for normal distribution
Legend:
df – degrees of freedom; Exp(B) – exponent (B); SBP – systolic blood pressure; SE – standard error; Sig. – significance; Wald – Wald test;
GCS pre – preclinical Glasgow Coma Scale
Table S1A. Model description
|
Model name |
MOD_1 |
|
|
Series or sequence |
1 |
age |
|
2 |
lactic acid |
|
|
3 |
preclinical SBP |
|
|
4 |
preclinical temperature |
|
|
5 |
SBP at the emergency department |
|
|
6 |
temperature at the emergency department |
|
|
7 |
lactate-transformed |
|
|
Transformation |
none |
|
|
Non-seasonal differencing |
0 |
|
|
Seasonal differencing |
0 |
|
|
Length of seasonal period |
no periodicity |
|
|
Standardization |
not applied |
|
|
Distribution |
type |
normal |
|
location |
estimated |
|
|
scale |
estimated |
|
|
Fractional rank estimation method |
Blom’s |
|
|
Rank assigned to ties |
mean rank of tied values |
|
Table S1B. Case processing summary (part 1)
|
Case processing summary (1) |
Age |
Lactic acid |
Preclinical SBP |
Preclinical temperature |
|
|
Series or sequence length |
263 |
263 |
263 |
263 |
|
|
Number of missing values in the plot |
user-missing |
0 |
0 |
0 |
0 |
|
system-missing |
0 |
153 |
6 |
9 |
|
Table S1C. Case processing summary (part 2)
|
Case processing summary (2) |
SBP at the emergency department |
Temperature at the emergency department |
Lactate-transformed |
|
|
Series or sequence length |
263 |
263 |
263 |
|
|
Number of missing values in the plot |
user-missing |
0 |
0 |
0 |
|
system-missing |
8 |
10 |
153 |
|
Table S1D. Estimated distribution parameters (part 1)
|
Estimated distribution (1) |
Age |
Lactic acid |
Preclinical SBP |
Preclinical temperature |
|
|
Normal distribution |
location |
80.342205 |
2.582727 |
120.521401 |
37.329921 |
|
scale |
11.8883485 |
3.0865752 |
32.6686808 |
1.5284038 |
|
Table S1E. Estimated distribution parameters (part 2)
|
Estimated distribution (2) |
SBP at the emergency department |
Temperature at the emergency department |
Lactate-transformed |
|
|
Normal distribution |
location |
124.909804 |
37.214625 |
0.953582 |
|
scale |
34.6133267 |
1.4401222 |
0.0855219 |
|
Table S2. Logistic regression analysis
Legend:
df – degrees of freedom; Exp(B) – exponent (B); SBP – systolic blood pressure; SE – standard error; Sig. – significance; Wald – Wald test;
GCS pre – preclinical Glasgow Coma Scale
Table S2A. Case processing summary
|
Unweighted casesa |
Number of cases |
Percentage |
|
|
Selected cases |
included in analysis |
252 |
95.8 |
|
missing cases |
11 |
4.2 |
|
|
total |
263 |
100 |
|
|
Unselected cases |
0 |
0 |
|
|
Total |
263 |
100 |
|
Table S2B. Classification table
|
Classification |
Observed |
Predicted |
|||
|
died: yes (1), no (0) |
percentage correct |
||||
|
0 |
1 |
||||
|
Step 0 |
died: yes (1), no (0) |
0 |
195 |
0 |
100 |
|
1 |
57 |
0 |
0 |
||
|
overall percentage |
77.4 |
||||
Table S2C. Variables in the equation
|
Variables included |
B |
SE |
Wald |
df |
Sig. |
Exp(B) |
|
|
Step 0 |
constant |
–1.230 |
0.151 |
66.724 |
1 |
0 |
0.292 |
Table S2D. Variables excluded from equation
|
Variables excluded |
Score |
df |
Sig. |
||
|
Step 0 |
variables |
age |
4.510 |
1 |
0.034 |
|
immunodeficiency: yes (1), no (0) |
2.647 |
1 |
0.104 |
||
|
preclinical SBP |
15.542 |
1 |
0.000 |
||
|
GCS pre |
5.013 |
1 |
0.025 |
||
|
overall statistics |
23.429 |
4 |
0.000 |
||
Table S2E. Model coefficients test
|
Model coefficients |
χ2 |
df |
Sig. |
|
|
Step 1 |
step |
25.104 |
4 |
0.000 |
|
block |
25.104 |
4 |
0.000 |
|
|
model |
25.104 |
4 |
0.000 |
|
Table S2F. Statistical model summary
|
Model summary |
–2 Log likelihood |
Cox & Snell R square |
Nagelkerke R square |
|
Step 1 |
244.351a |
0.095 |
0.144 |
Table S2G. Classification table
|
Classification |
Observed |
Predicted |
|||
|
died in the hospital: yes (1), no (0) |
percentage correct |
||||
|
0 |
1 |
||||
|
Step 1 |
died: yes (1), no (0) |
0 |
192 |
3 |
98.5 |
|
1 |
50 |
7 |
12.3 |
||
|
overall percentage |
79.0 |
||||
Table S2H. Variables in the equation (calculation for significance)
|
Variables for equation |
B |
SE |
Wald |
df |
Sig. |
|
|
Step 1a |
age |
0.024 |
0.015 |
2.566 |
1 |
0.109 |
|
immunodeficiency: yes (1), no (0) |
–0.558 |
0.465 |
1.439 |
1 |
0.230 |
|
|
preclinical SBP |
–0.021 |
0.006 |
12.949 |
1 |
0 |
|
|
GCS pre |
–0.088 |
0.070 |
1.585 |
1 |
0.208 |
|
|
constant |
0.430 |
1.699 |
0.064 |
1 |
0.800 |
|
Table S2I. Exponent (B) summary
|
Exponent (B) summary |
Exp(B) |
|
|
Step 1a |
age |
1.024 |
|
immunodeficiency: yes (1), no (0) |
0.572 |
|
|
preclinical SBP |
0.980 |
|
|
GCS pre |
0.916 |
|
|
constant |
1.538 |
|