Abstract
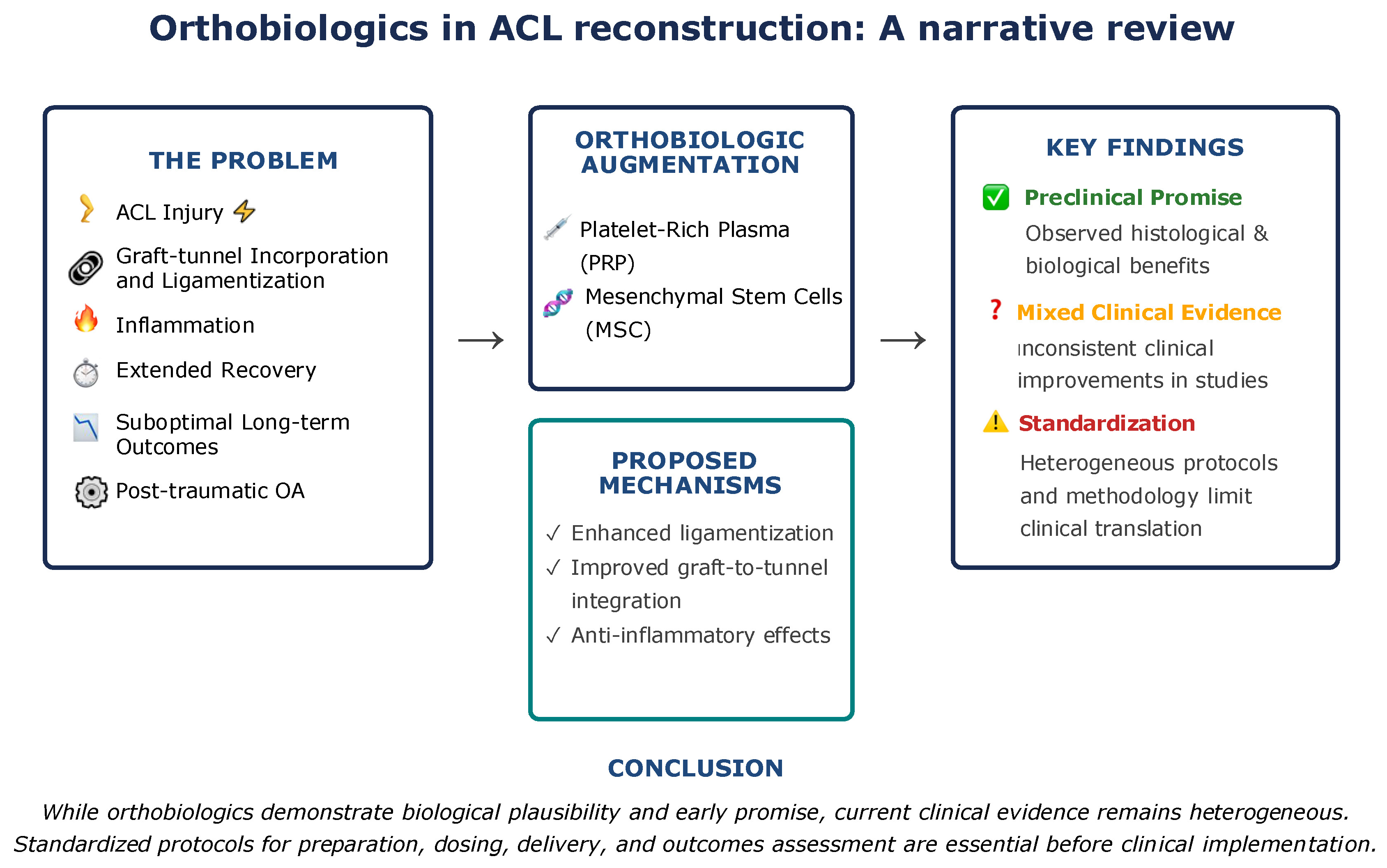
Anterior cruciate ligament (ACL) injury is one of the most frequent injuries in sports medicine. Despite advancements in surgical techniques, suboptimal results in terms of delayed graft incorporation into bone tunnels and ligamentization, as well as inferior clinical outcomes, still present significant challenges in treating these injuries. Orthobiological modalities such as platelet-rich plasma (PRP) and stem cells have emerged as a potential adjunct to ACL treatment, with a theoretical advantage in enhancing local biological processes and subsequent improvements in clinical outcomes. Embase, Cochrane, and PubMed databases were searched for relevant literature. The aim of this narrative review was to summarize the currently available evidence on the efficacy of orthobiologics, or the lack thereof, in ACL repair and reconstruction (ACLR), with emphasis on graft incorporation into bone tunnels, ligamentization, and overall clinical outcomes. These modalities exert their theoretical effect by delivering growth factors, anti-inflammatory cytokines, and regenerative cells to the target site, which enhance local biological processes. Although some research has shown positive effects of orthobiologics in ACLR, clinical outcomes remain inconsistent, without consensus on preparation methods and administration protocols. The lack of homogeneous, high-quality methodology in existing studies limits the possibility of drawing definitive conclusions on the efficacy of orthobiologics in ACLR. Although orthobiological modalities provide theoretical benefits in improving outcomes, until more robust evidence is available regarding their efficacy and optimal application methods, their routine use warrants caution and cannot yet be recommended. Future research should focus on improving and standardizing clinical and research protocols before drawing any definitive conclusions on this matter.
Key words: tissue engineering, ligament repair, anterior cruciate ligament reconstruction, orthobiologics
Introduction
Orthobiologics are a multifaceted group of biologically derived products aimed at enhancing the healing and regeneration of musculoskeletal tissues, including bone, cartilage, tendons, and ligaments.1, 2 These agents include platelet-rich plasma (PRP), bone marrow aspirate concentrate (BMAC), amniotic suspension allograft (ASA), adipose-derived products, which include micro-fragmented adipose tissue (mFAT) and stromal vascular fraction (SVF), mesenchymal stromal cells (MSCs), as well as various growth factor-based preparations. Orthobiologics act through a variety of mechanisms, including modulation of inflammation, stimulation of resident cell proliferation and migration, promotion of angiogenesis, and support of extracellular matrix remodeling.3 In the context of orthopedic surgery, they are increasingly being investigated and applied as adjuncts to conventional procedures with the aim of accelerating recovery, improving tissue integration, and potentially enhancing long-term outcomes.4 Their role in ligament repair and reconstruction, and in particular in anterior cruciate ligament (ACL) surgery, is of growing interest due to the biological complexity of ligament healing and the need for effective strategies to restore function and prevent complications.5
Anterior cruciate ligament injuries represent one of the most common and clinically significant traumas in sports medicine, with an estimated 200,000 cases annually in the USA alone.6, 7, 8 Despite advancements in surgical techniques, autograft selection, and rehabilitation, outcomes following ACL reconstruction (ACLR) remain poor in terms of functional outcomes such as full return to pre-injury activity levels, as well as long-term prevention of osteoarthritis.9 In theory, the graft within the bone tunnels should undergo complete mineralization and be fully replaced by bone tissue, with restoration of the native fibrocartilage transition zone at the original ACL insertion sites.10, 11 As the field moves towards personalized biologic strategies, it is critical to revisit the fundamental biology of graft healing and the ligamentization process. Ligament and tendon tissues are similar, but some differences do exist.12 For instance, compared to tendons, ligaments exhibit greater metabolic activity, contain cells with more rounded nuclei, and display higher DNA content.5 They also possess a greater proportion of type III collagen and proteoglycans, a lower overall collagen content, distinct profiles of non-reducible collagen cross-links, and a different distribution of collagen fibril diameters.5, 13
Ligamentization refers to the series of biologic processes that result in tendon graft transformation into a ligament-like structure after ACLR.10, 13 This biologic remodeling process has been observed histologically and radiographically and is divided into 3 overlapping phases: early necrosis and hypocellularity, remodeling and revascularization, and the maturation phase.14, 15 The 1st phase is characterized by the apoptosis of graft cells, in which inflammatory cells infiltrate first, followed by MSCs. These MSCs differentiate and secrete extracellular matrix, including collagen strands, which leads to peripheral granulation tissue being formed.14, 15 The risk of loosening in bone tunnels is increased during the first phase, with the transition spot of 2 inhomogeneous, different types of tissue being the locus minoris resistantiae.16 In the remodeling phase, angiogenesis factors are secreted by MSCs and inflammatory cells. In the meantime, collagen fibers acquire a more regular orientation, and Sharpey’s fibers are formed at the points of tendon insertion into the bone.16 In this second phase, the intra-articular part of the graft, due to the occurring changes, is the weakest link of the biologic construct.11 In the maturation phase, the graft becomes more similar to the natural ligament, the percentage of collagen decreases, and collagen fibers are organized in a more parallel fashion, while neovascularization penetrates the central part.12, 13 All studies have shown that ligamentization lasts significantly longer in humans than in animals.12, 14, 15 It has been demonstrated that all of these phases last longer than previously thought; thus, the early phase is considered to last for the first 6 months, the remodeling phase from the 7th to the 12th month, and the maturation phase from the 13th to the 24th month post-surgery.13 Despite its nomenclature, the remodeled graft does not fully replicate the ultrastructure or mechanical integrity of the native ACL.10, 13 Histological analyses in both animal models and human studies have shown that, while tendon grafts remain viable and undergo progressive vascularization and cellular infiltration, complete recapitulation of native ACL architecture, including zonal insertion and collagen cross-linking patterns, is rarely achieved.12, 15 Magnetic resonance imaging (MRI) studies demonstrate variable signal intensities reflecting differential maturation patterns based on graft type, source, and surgical techniques.17 For instance, contrary to popular belief, hamstring autografts tend to show more favorable signal progression compared to bone–patellar tendon–bone (BPTB) grafts by 12 months postoperatively.18
Objectives
The relative lack of studies on the topic of orthobiologics in ACLR raises concerns about the justification for their use in clinical practice. We conducted a comprehensive literature search in the Embase, PubMed, and Cochrane databases using various combinations of the following search terms: “orthobiologics,” “orthobiological augmentation,” “tissue engineering,” “anterior cruciate ligament reconstruction,” “anterior cruciate ligament repair,” “platelet-rich plasma,” and “stem cells”. Two authors (M.O. and E.R.) independently screened the literature for articles on the use of orthobiologics in ACLR and repair. After the initial search, all articles were re-evaluated by both authors, with discussion to determine whether each study was suitable as a source of information for this paper. The aim of this study was to collect and qualitatively synthesize the currently available evidence on the influence of orthobiological modalities on the local biological processes after ACLR and, where available, repair.
Orthobiologics in ACL injuries
Biologic augmentation using orthobiologics, such as PRP, stem cells, and other molecular or cellular agents, has been increasingly explored as a strategy to enhance graft-to-bone tunnel incorporation and ligamentization, modulate the inflammatory milieu, and promote tissue regeneration.1, 19 The application of orthobiologics in ACLR represents a logical extension of the principles used in osteoarthritis treatment. While mechanical restoration of joint function is achievable through surgery, biological healing of the graft or repaired ligament remains a critical determinant of long-term success.13, 20, 21 In this context, orthobiologics may offer valuable support by improving graft ligamentization, enhancing tendon-to-bone integration, and reducing local inflammation that may otherwise impair healing.22
Several factors may impede or enhance ligamentization. Excessive inflammation, faulty angiogenesis, micromotion within the bone tunnels, and individual biological variability contribute to inconsistent graft remodeling.10 Both ACL injury and ACLR result in an elevation of pro-inflammatory cytokines in the synovial fluid, which further attract pro-inflammatory cells, creating a vicious circle.11 Previously, different cytokines and growth factors, such as bone morphogenetic protein (BMP), fibroblast growth factor (FGF), and transforming growth factor beta (TGF-β), have been utilized to increase the speed of angiogenesis, suppress inflammation, and boost the osteogenic potential of the bone tunnels.11, 16 Orthobiologics are being investigated for their capacity to modulate these factors, aiming to accelerate and qualitatively improve the ligamentization trajectory.1
Understanding the biological timeline and limitations of native healing is essential for interpreting clinical outcomes and designing biologic augmentation strategies. The rationale behind the use of orthobiologics in ACLR hinges on their potential to influence 2 key biological processes: graft-to-bone tunnel incorporation and intra-articular graft remodeling, or “ligamentization”.1 In ACLR, orthobiologics can enhance the biological environment at the graft–bone interface and within the intra-articular space, potentially accelerating the remodeling process and supporting more robust integration of the graft.23 Preclinical and early clinical studies have shown that orthobiologic augmentation may improve histological, biomechanical, and imaging-based outcomes, though more high-quality clinical evidence is still needed.24, 25 Although studies have emphasized the importance of graft incorporation when considering the success of ACLR, especially with regard to RTP time, structured protocols to quantify and target these biological processes have yet to be developed.26 On the other hand, in ACL repair, where the preservation and healing of the native ligament is the goal, orthobiologics such as MSCs or PRP may facilitate intrinsic healing by delivering pro-regenerative signals and modulating the early inflammatory response.23
Beyond their role in enhancing ligament healing, orthobiologics may also serve as disease-modifying agents by addressing a less frequently discussed but clinically significant issue: the onset and progression of post-traumatic osteoarthritis (PTOA).27 Both ACL injury and its surgical treatment are considered “cartilage-damaging” events.28 Anterior cruciate ligament rupture often occurs in conjunction with meniscal injury and subchondral bone bruising, and the resulting joint instability can lead to altered load distribution, chronic low-grade inflammation, and accelerated cartilage degeneration.29 Even after successful reconstruction, patients remain at increased risk of developing PTOA, likely due to residual joint kinematic alterations and the catabolic intra-articular environment induced by injury and surgery.30 In this context, orthobiologics have the potential to mitigate these deleterious processes. Adipose-derived MSCs and BMAC, in particular, have demonstrated anti-inflammatory and chondroprotective effects in preclinical models,31 and early clinical studies suggest that their intra-articular application may preserve cartilage integrity and slow osteoarthritic changes when used in the acute or subacute phase after ACL injury.32, 33 By addressing both the biomechanical and biological consequences of ACL trauma, orthobiologic augmentation could therefore not only improve short-term healing but also reduce the risk of long-term degenerative sequelae.
Orthobiologics in ACL reconstruction
PRP in ACL reconstruction
Platelet-rich plasma has shown its potential in various fields of orthopaedic surgery.1, 34, 35, 36 Biological processes that occur during the course from injury to rehabilitation are highlighted as potential targets to improve outcomes after ACL injury and reconstruction.37 Platelet-rich plasma is a derivative of peripheral venous blood. It is produced by a centrifugation process which results in the end product – a suspension of platelets with a concentration multiple times higher than that of peripheral blood.34 The effects of PRP on tissue healing and repair have been well studied.24 It is composed of various growth factors and cytokines such as vascular endothelial growth factor (VEGF), insulin-like growth factors, TGF-β, FGF, and EGF.37, 38, 39 Some of these have demonstrated positive effects on cell migration, angiogenesis, and matrix production in both in vivo and in vitro models.37, 38, 39
The theoretical mechanism by which PRP could impact biological processes in ACLR is through the influence on collagen expression and fibroblast stimulation through their growth factor receptors.37 This raises theoretical questions about its possible role in ligamentization, anti-inflammatory effects inside the joint post-surgery, and graft–tunnel integration, which could consequently lead to better clinical outcomes and even a decrease in return-to-play time after ACL surgery.37, 40 Considering the potential benefits, it is not surprising to observe a growing number of preclinical and clinical studies on this topic.24, 37, 38 There are various methods by which PRP can be applied during ACLR: coating the graft during its preparation, injecting the finished product into the graft, or simply performing intra-articular application after surgery.23, 39 Preclinical evidence suggests that PRP has the potential to improve both the histological state and biomechanical outcomes after ACLR.24 However, previous research that investigated this preclinical evidence did not manage to reach a univocal conclusion based on clinical research. Although PRP is considered to be safe and well tolerated for this purpose, there is no consensus on the appropriate activation methods, platelet concentration, or the treatment’s clear benefits.23, 39, 41
In the context of ACLR, PRP has been explored with regard to its effects on ligamentization, graft–tunnel interface changes, donor-site morbidity, clinical outcomes, and return-to-play time.23, 38, 41, 42 Some studies have supported PRP augmentation for improving graft maturity.24, 39 In the review by Kon et al., the majority of studies analyzed in the context of graft ligamentization have shown some benefits of PRP for improving graft maturity.23 In a prospective, single-blinded study on 50 ACL-reconstructed knees by Radice et al., researchers examined whether PRP can positively affect MRI findings after ACLR.43 The authors concluded that PRP can shorten the time required to achieve a homogeneous graft based on MRI findings by almost 50%.43 A similar topic was studied by Sánchez et al., who investigated whether the application of PRP has any advantages when used as an adjunct to ACLR.44 Of the 37 volunteers who participated in this research, 15 were treated conventionally, and 22 were treated with PRGF-augmented ACLR using hamstring tendons.44 This study was of a histomorphological nature, and all volunteers underwent second-look arthroscopy. The morphology of the graft was evaluated based on an arthroscopic score, while histological changes were observed in graft biopsy samples obtained during the second-look arthroscopy.44 PRGF-augmented ACL-reconstructed knees resulted in histologically more mature tissue in comparison to conventionally treated patients. Moreover, these knees had better scores based on arthroscopic findings.44 In their study, Seijas et al. evaluated patellar tendon remodeling on MRI scans.45 In total, 98 patients were included in this randomized study, with an age range of 18–65 years.45 Platelet-rich plasma was applied intra-articularly after the last suture. Magnetic resonance imaging scans were obtained after 4, 6, and 12 months. More patients in the PRP-augmented group were assigned higher remodeling scores. The authors concluded that PRP provides the possibility of accelerating the remodeling of patellar tendon grafts after ACLR.45 When the bone–tunnel interface was observed in terms of graft incorporation and tunnel widening, studies were almost univocal in concluding that PRP does not provide any benefits.23, 39, 42, 46
When observing the clinical outcomes of studies on PRP in ACLR, most agree that PRP does not have a clinically significant impact on improving outcomes after ACLR. A meta-analysis of 9 randomized controlled trials (RCTs) by Pinho et al., with a minimum follow-up of 12 months, included 502 patients, of whom 251 underwent PRP-augmented ACLR.47 Although the results showed a statistically significant improvement in the IKDC score in the PRP-augmented ACLR group, no clinically significant difference was observed based on the minimal clinically important difference.47 In addition, no significant differences were observed in Tegner or Lysholm scores.47 In the systematic review by Andriolo et al., the authors concluded that PRP does not offer any clinical benefits when used as an adjunct to ACLR.39 However, based on the results of a very limited number of studies, some minor benefits were noted with regard to healing mechanisms, which could result in earlier graft maturation and decreased donor-site morbidity.39
Somewhat different conclusions were reported in the review by Davey et al. The authors did not support PRP in ACLR whatsoever and concluded that PRP does not offer any benefits with regard to improving clinical and functional outcomes, postoperative pain, or donor-site morbidity.38 Although interest in this topic is rising, the heterogeneity of the studies and their methodologies makes it difficult to perform high-quality systematic reviews. Figueroa et al. did not manage to perform a quantitative analysis in their systematic review because of significant clinical heterogeneity among the included studies.42 After including studies available up to that date, the paper encompassed 266 ACLRs augmented with PRP and 250 without PRP.42 Based on a qualitative analysis of the papers, the authors concluded that there is some evidence that adding PRP into the graft or bone tunnels during ACL reconstruction could promote quicker graft maturation in comparison to reconstruction performed without PRP.42 When looking at other observed outcomes such as tunnel widening and clinical outcomes, it was concluded that PRP does not offer any benefits.42
In the research by Kon et al., the influence of PRP and PRF in ACL repair or reconstruction was examined. Twenty-three papers were included in this study.23 The authors evaluated clinical and functional scores, objective measurements, and return-to-play time.23 Platelet-rich plasma did not provide any superiority over not using it in affecting clinical outcomes or shortening the return-to-play time.23 A systematic review by Zhu et al. analyzed 14 RCTs that evaluated the effects of PRP on subjective patient-reported scores, clinical knee function assessments, and related complications.46 In this study, the injection of PRP into tunnels during ACLR provided short-term benefits after 3 months by improving visual analogue scale (VAS) and IKDC functional scores.46 Also, when PRP was applied at the BPTB harvest site, pain measured on the VAS decreased at 6 months postoperatively without further benefits. The authors concluded that PRP could offer some early pain reduction and improve subjective knee function in the short term, but it does not improve objective knee performance measurements or have beneficial effects on graft healing or preventing tunnel enlargement.46 Berdis et al. provided a possible solution to the efficacy problem of PRP by adding a collagen carrier to their grafts.41 The theory behind this is that plasmin-mediated degradation of the fibrin layer that forms during surgery leaves PRP with unfulfilled potential.41 In this study, 151 knees from 143 patients were evaluated.42 In total, 132 patients returned to their pre-injury activity level. The authors concluded that augmenting the graft with a collagen carrier when adding PRP can decrease the rate of reinjuries and revisions.41 In addition, a quicker return to the pre-injury activity level in comparison to other studies was reported. The downside of this study was the lack of a control group and long-term outcome analysis.41 A summary of clinical studies on the use of PRP in ACLR is shown in Table 1.
Looking at the currently available evidence, the implementation of PRP during ACLR in clinical practice is still controversial. Although there is some evidence suggesting that PRP may accelerate graft ligamentization and improve short-term outcomes, based on the lack of robust studies with homogeneous methodology, the evidence is still insufficient to recommend the introduction of PRP into the routine practice of ACLR.
Stem-cells in ACL reconstruction
Mesenchymal stromal cells are a population of non-hematopoietic cells found in bone marrow (BM-MSCs), adipose tissue, liver, amniotic fluid, embryonic placenta, umbilical cord blood, and other tissues, and they are increasingly recognized as a versatile biologic adjunct in ACLR. Beyond their intrinsic self-renewal and multilineage potential, MSCs can adopt fibroblastic and osteoblastic phenotypes within the bone tunnels, thereby recreating a native-like fibrocartilaginous transition zone between graft and bone. Concomitantly, MSCs secrete a broad repertoire of trophic cytokines and growth factors (including VEGF, TGF-β, IGF-1, and BMPs) that coordinate angiogenesis, modulate inflammation; and drive matrix remodeling across cartilage, bone, ligament, and tendon compartments. This combined differentiation and paracrine activity positions MSC therapy as a promising strategy to expedite graft ligamentization and improve osteointegration after ACL surgery.48, 49 Recently, an ongoing study by Wang et al. proposed an innovative use of the infrapatellar fat pad to produce micro-fragmented adipose tissue, which may help overcome the disadvantages of abdominal fat harvesting while preserving the biological properties of the resident stem cell population.50
Only 4 animal studies paint a cautiously optimistic but not uniformly positive picture of MSC augmentation in ACL graft healing.51 On the upside, the 2 experiments that delivered bone marrow MSCs (≈1–2 × 106 cells) directly into a hamstring autograft with a fibrin-based carrier produced the clearest gains: ultimate failure load jumped by 22% at 8 weeks52 and 38% at 12 weeks.53 Those mechanical improvements were matched histologically by denser, better-aligned collagen fibers and a richer Sharpey-like insertion, changes preceded by a twofold rise in CD31-positive vessel density, evidence that MSCs boosted both angiogenesis and matrix remodeling. However, not every protocol translated biological potential into physical properties. When MSCs were supplied as a collagen-sheet wrap54 or as tendon-derived stem cells layered around an allograft,55 investigators observed encouraging early biological findings (greater cellularity, vessel ingrowth, and improved fiber alignment) but no significant change in failure load or stiffness up to 6 months. Those findings underline 2 limitations: 1) cell-retention strategies matter (wrapping may prevent loss of cells in the joint space), and 2) histological maturation does not guarantee immediate mechanical benefits. Taken together, the data suggest that intra-graft injection with a bioadhesive carrier is the most promising route, but standardizing dose, follow-up length, and mechanical endpoints will be essential before these early successes can be translated into reliable clinical benefit.
Among the 5 available RCTs that examined BMAC during primary ACLR in humans, only 2 demonstrated measurable biological advantages, and just 1 translated these gains into superior patient-reported function.56 Lin et al. injected 3 mL of undiluted BMAC exclusively into the femoral and tibial tunnels of hamstring autografts and observed a ≈9-point improvement in IKDC at 24 months, accompanied by lower graft signal intensity on proton-density fat-suppressed MRI, which was interpreted as a surrogate of advanced ligamentization.57 Forsythe et al. likewise reported lower signal-intensity ratios on T2-weighted scans in BMAC-treated BTB allografts, indicating enhanced metabolic activity, although this did not translate into a clinically significant change in IKDC or Lysholm scores.58 The remaining 3 trials yielded neutral findings. Anz et al. (mixed BTB and hamstring autografts) and Lavender et al. (quadriceps tendon grafts augmented with demineralized bone matrix) found no differences in T2* relaxation times, graft volume, or functional scores after 24 months.59, 60 Bhamare et al., the only study with a 9-month endpoint, provided qualitative MRI observations but no quantitative data, reporting neither clinical nor radiological benefits.61 Importantly, none of the 5 RCTs recorded BMAC-related adverse events beyond routine postoperative effusions or minor infections, and re-rupture rates were comparable with controls, underscoring the procedural safety of aspirate use.56 A summary of clinical studies on the use of BMAC in ACLR is shown in Table 2.
Taken together, current level I evidence suggests that tunnel-focused BMAC delivery can accelerate MRI-defined graft maturation with minimal increase in complications but does not significantly improve ACLR outcomes (either statistically or clinically). However, the heterogeneity in graft type, cell dose, adjunct carriers, and imaging methodology, coupled with modest sample sizes, limits definitive conclusions. Larger, harmonized multicenter trials with standardized MRI protocols, cost-utility analyses, and follow-up extending beyond 2 years are warranted before BMAC can be advocated as routine augmentation in ACLR.
Limitations of the study
This narrative review has several limitations. First, the available and reviewed studies vary widely in terms of methodology, preparation protocols, and orthobiologic products used, as well as outcome measures. The narrative nature of this review limits the possibility of quantitative analysis, with the additional possibility of excluding recently published studies, which may affect the comprehensiveness of the study. Future clinical studies should insist on more robust and standardized methodological protocols to better define the role of orthobiologics in ACLR. This would also facilitate data quantification and the production of high-quality systematic reviews.
Conclusions
Despite advancements in surgical techniques, the biological aspects of graft healing remain a critical determinant of successful outcomes following ACL repair and reconstruction. Orthobiologic augmentation – including PRP and stem cell-based therapies – holds promising potential to enhance ligamentization, improve graft integration, and possibly mitigate post-traumatic osteoarthritis. While preclinical data and early clinical studies show biological and histological benefits, the current level of evidence remains heterogeneous and inconclusive regarding consistent clinical improvement. Standardization of orthobiologic protocols, dosing, delivery methods, and outcome measures is urgently needed. Until then, routine use of these therapies should be approached cautiously and selectively, guided by individual patient context and evolving high-quality evidence.
Use of AI and AI-assisted technologies
Not applicable.