Abstract
The triglyceride-glucose (TyG) index, a simple surrogate marker integrating insulin resistance and atherogenic dyslipidemia, has attracted growing attention as a potential tool for addressing residual risk in cardiovascular disease (CVD) prediction. Robust observational evidence consistently links the TyG index to hard cardiovascular endpoints and subclinical atherosclerosis, positioning it as a strong candidate for clinical translation. However, its adoption in mainstream guidelines is constrained by a critical limitation: the current evidence is exclusively observational and lacks interventional data demonstrating that lowering the TyG index improves clinical outcomes. This editorial argues that, although the TyG index represents a practical and readily available risk-stratification adjunct, it has not yet evolved into a validated therapeutic target. We propose a dual-pathway strategy: provisional recognition within guidelines as a risk-enhancing factor, accompanied by a clear call for dedicated cardiovascular outcome trials. Such an approach would capitalize on its present utility while upholding the evidentiary standards required to inform clinical practice.
Key words: triglycerides, blood glucose, insulin resistance, cardiovascular disease, risk assessment
Introduction
A consensus is emerging in preventive cardiology: the triglyceride-glucose (TyG) index is a remarkably strong, accessible, and independent marker of cardiovascular risk. Its calculation is seductively simple, its pathophysiological basis – integrating insulin resistance and atherogenic lipoproteins – is compelling, and its association with future events is consistently validated across diverse cohorts. It would seem, then, that its place in future clinical guidelines is all but assured.
However, the very ease of its adoption presents a danger. The translation of a risk marker into a guideline-recommended action is a process governed by rigorous evidentiary hierarchies, not merely by statistical association. The medical landscape is littered with biomarkers whose promising observational data never translated into improved patient outcomes when put to the interventional test. Thus, the critical question is not whether the TyG index is useful, but how it should be useful at this precise moment in its evidentiary evolution.
This editorial argues that we must resist the temptation to anoint the TyG index as a standalone intervention target prematurely. Its current, undeniable value lies in risk refinement – identifying the “hidden” high-risk individual within traditional intermediate-risk categories. I propose that guidelines should formally categorize it as a “risk-enhancing factor,” a tool to prompt more personalized clinician–patient discussion and enhance lifestyle counseling. Concurrently, and with equal urgency, the research community must prioritize the definitive randomized trials that can probe its causal role. The TyG index is ready for a supporting role in risk assessment today, but its starring role in therapeutic guidance awaits the next act of scientific validation.
The guideline dilemma and the TyG opportunity
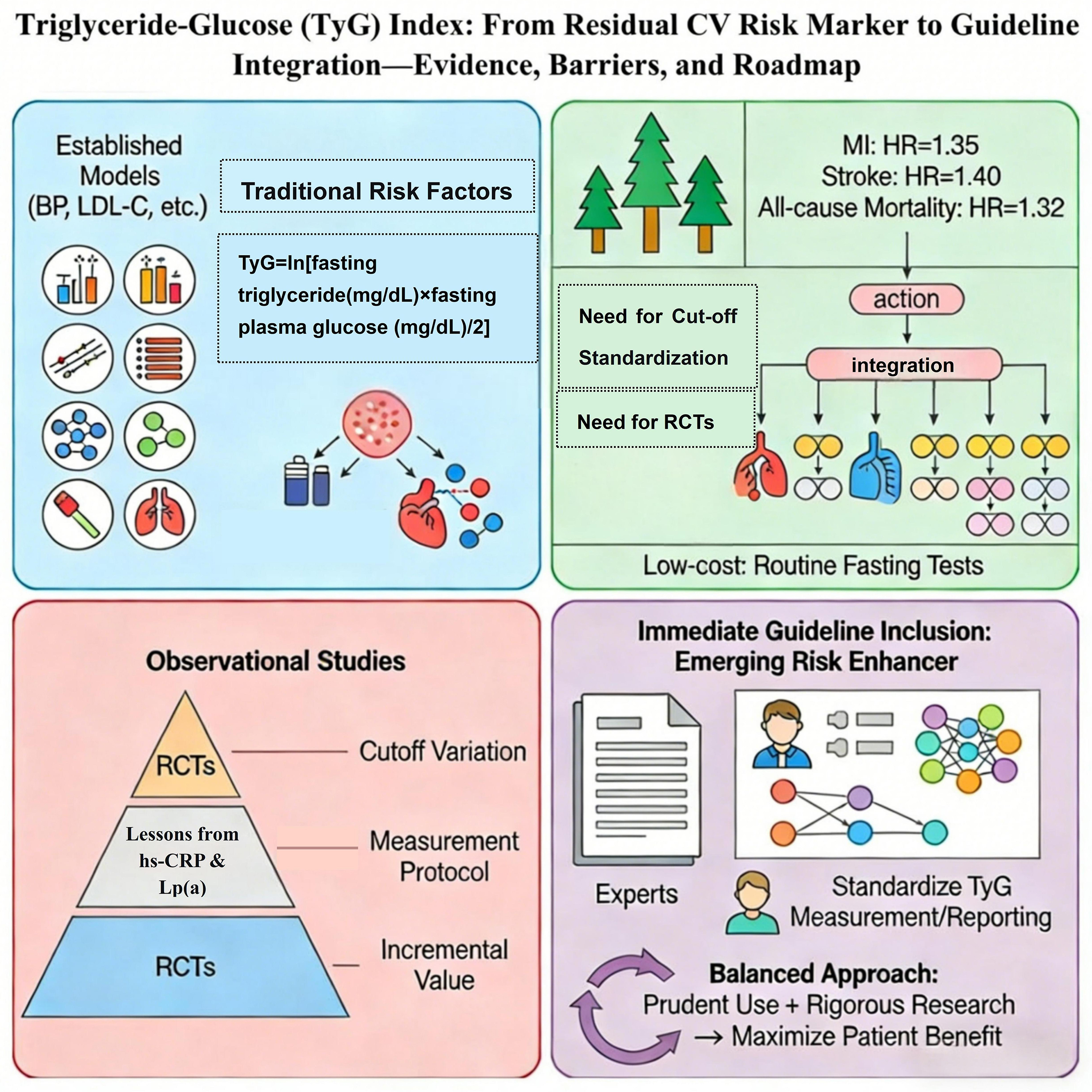
Despite the widespread application of existing risk prediction models based on traditional factors (e.g., blood pressure, low-density lipoprotein cholesterol [LDL-C]), the “residual risk” of cardiovascular disease (CVD) remains significant.1 This drives the search for novel biomarkers that can more comprehensively and earlier reflect the essence of metabolic dysregulation. Insulin resistance and atherogenic dyslipoproteinemia are core pathophysiological mechanisms driving this residual risk,2 yet current guidelines lack simple, cost-effective tools to routinely assess these dual pathways. The TyG index (Ln[fasting triglycerides (mg/dL) × fasting glucose (mg/dL)/2]) emerges precisely for this purpose. It ingeniously integrates these 2 mechanisms into a simple formula, representing a paradigm shift from assessing “single metrics” to evaluating “integrated pathways”.3 Faced with such a promising marker, what should be the stance of clinical guidelines: enthusiastic adoption or cautious vigilance?
Compelling evidence for TyG index usefulness
Large-scale, long-term follow-up studies have shown that the TyG index is significantly associated with myocardial infarction, stroke, and all-cause mortality.4, 5, 6 Its predictive power is independent of traditional risk factors and often superior to single glycemic or lipid metrics.7, 8, 9 Deeper investigation reveals that derivatives combining TyG with indices like body mass index (BMI) or waist circumference (WC) (e.g., TyG-BMI, TyG-WC) further enhance predictive accuracy, showcasing its excellent scalability.10, 11
The TyG index integratively reflects the 2 key drivers of residual cardiovascular risk. First, as a valid surrogate for insulin resistance, its elevation signals abnormalities in glucose and fat metabolism, accompanied by increased free fatty acids and a pro-inflammatory state.12 Second, elevated triglyceride levels per se are a marker for increased atherogenic triglyceride-rich lipoproteins (TRLs) and their remnants, which directly damage vasculature by inducing endothelial dysfunction and oxidative stress.13 Thus, through a simple calculation, the TyG index captures both the “upstream mechanism” (insulin resistance) and the “downstream effect” (TRLs).
The TyG index correlates strongly with subclinical atherosclerosis, providing a mechanistic bridge to hard endpoints. Substantial evidence confirms its strong association with key markers of subclinical atherosclerosis. At the vascular structural level, an elevated TyG index is independently associated with and is a significant risk factor for increased carotid intima-media thickness (CIMT).14 At the coronary level, cross-sectional and intravascular ultrasound evidence indicates that a higher TyG index is significantly associated with the presence, extent, and unstable features (e.g., spotty calcification) of coronary artery calcification (CAC).15 A meta-analysis of 41 studies concluded that individuals with a high TyG index are at significantly increased risk of coronary calcification, plaque progression, and multivessel disease.16 These findings indicate that the insulin-resistant and dyslipidemic state captured by the TyG index directly promotes the initiation, progression, and destabilization of atherosclerotic lesions. Therefore, the TyG index is not merely a risk marker but a mechanistic link connecting metabolic dysregulation to structural vascular damage.
Finally, the calculation of the TyG index relies solely on the most routine, low-cost laboratory tests, obtainable at any level of healthcare facility. This grants it immense public health value, particularly in resource-limited settings.
The “critical gap” hindering TyG index guideline inclusion
However, a sober recognition is essential: Observational association is merely the first step. Changing clinical practice through guidelines requires the highest level of evidence – demonstrating that intervention on the marker yields clear clinical benefit. Currently, all evidence supporting the TyG index remains observational, proving “association” but not “causation” or “intervention benefit”. This evidence chasm is precisely what separates a “risk marker” from a “clinical decision-making target.” The historical trajectory of high-sensitivity C-reactive protein (hs-CRP) offers a profound lesson: Despite an immense body of evidence linking it to cardiovascular risk, and a positive trial (JUPITER) designed based on this link,17 a subsequent trial specifically testing the “anti-inflammatory” benefit (CIRT) failed to improve cardiovascular outcomes.18 This exposes a critical issue: Even if a marker can be lowered by an intervention, it does not equate to the efficacy of a direct, pathway-specific intervention. Consequently, hs-CRP has not been widely adopted as an independent target to initiate therapy.
In contrast, the transformative pathway of lipoprotein(a) (Lp(a)) points the way forward. Although its causal link to cardiovascular risk was long established through large-scale genetic studies, its fundamental change in clinical stature began when drugs targeting Lp(a) reduction (e.g., Zerlasiran) demonstrated safe and potent lowering of its levels by over 80% in clinical trials, with several phase III trials with cardiovascular endpoints underway.19, 20 It is precisely this progress that has led to Lp(a)’s recent inclusion in multiple international guidelines. In contrast, the TyG index stands at this decisive threshold: It urgently needs its own ‘cardiovascular outcomes trial’ to answer the pivotal question – can actively lowering a high TyG index ultimately reduce the incidence of myocardial infarction and stroke? Only with positive results from such prospective randomized controlled trials (RCTs) can the TyG index complete its transformation from an excellent risk predictor to an actionable clinical decision-making tool.
Furthermore, the distribution and optimal risk cut-off points for the TyG index vary across races, ages, and genders. Guidelines require a universal or stratified standard. As the index fluctuates with lifestyle and medication, should guidelines recommend a single measurement or an average/trend from multiple readings? In modern risk prediction models incorporating over a dozen variables (e.g., the U.S. Pooled Cohort Equations), is the Net Reclassification Improvement (NRI) or Integrated Discrimination Improvement (IDI) gained by adding the TyG index significant enough to justify the added complexity and change risk stratification? Finally, its routine use necessitates evaluation of “clinical net benefit” to prevent unnecessary additional testing or over-treatment of borderline cases.
A pragmatic pathway towards guideline inclusion
I propose that current guidelines formally include the TyG index within chapters dedicated to “Emerging Risk Markers” or “Risk-Enhancing Factors.” Its role should be defined not as an independent standard to initiate therapy, but as a decision-aid for risk stratification and an alert indicator to intensify patient education. For instance, in individuals at intermediate traditional risk but with a significantly elevated TyG index, an escalation of intervention intensity could be considered.
There is a compelling need to launch TyG index-directed RCTs. These trials could enroll asymptomatic individuals at low-to-intermediate traditional risk but with a TyG index above a specific cut-point, randomizing them to standard care versus intensified intervention arms (e.g., early initiation of statins, GLP-1 receptor agonists, or intensive lifestyle management), with cardiovascular events as the primary endpoint.
We recommend that professional societies lead the development of expert consensus statements on the measurement and reporting of the TyG index to promote standardization.
Conclusions
Prudent integration of the TyG index into clinical context and the active pursuit of rigorous research are not mutually exclusive but embody the rigor and dynamism of evidence-based medicine. I embrace this promising tool but must validate it against the highest scientific standards, with the ultimate goal of patient benefit.
Use of AI and AI-assisted technologies
Not applicable.