Abstract
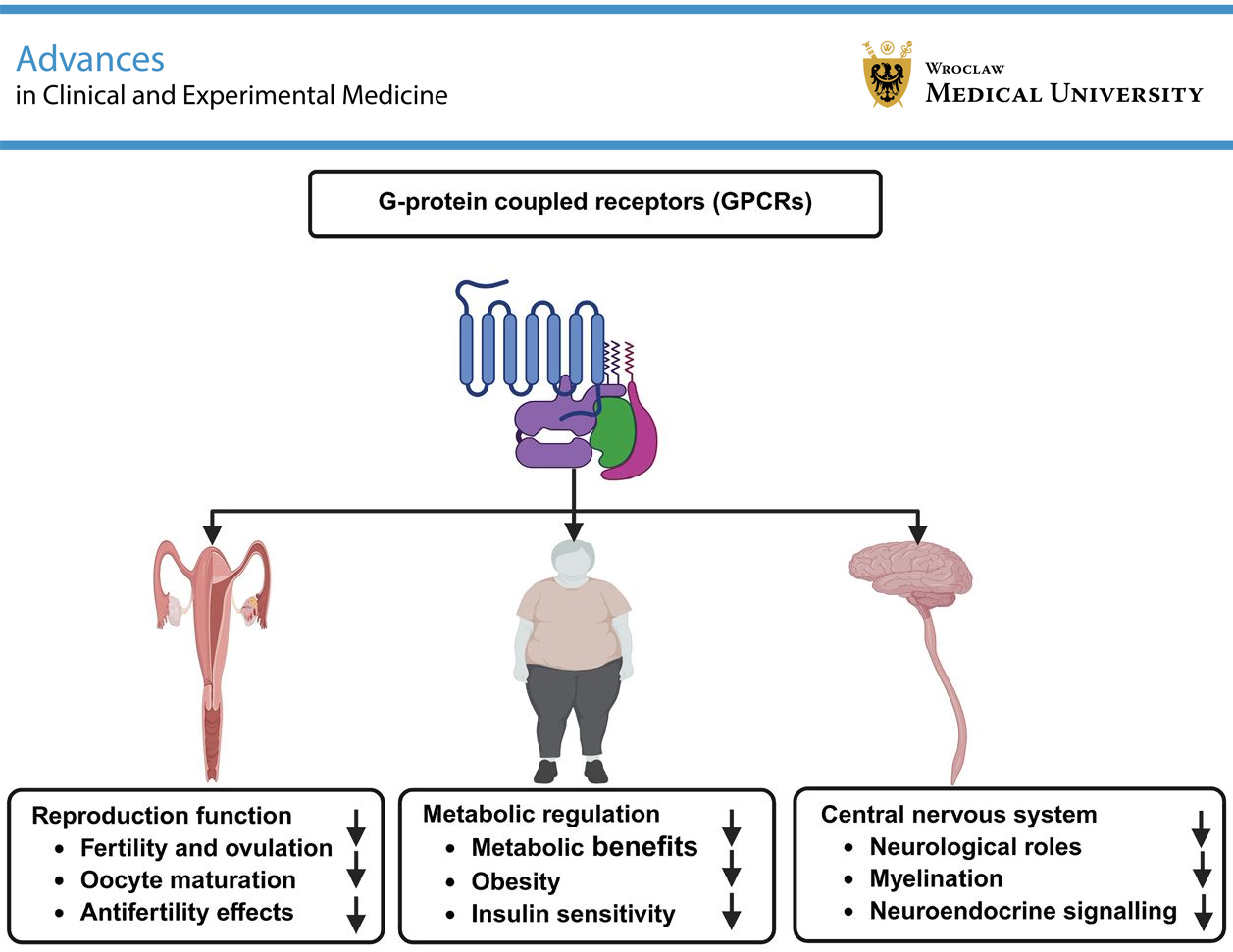
G protein-coupled receptors (GPCRs) constitute one of the largest and most versatile families of membrane proteins, mediating a wide range of physiological functions through intracellular signaling pathways. Among them, G protein-coupled receptor 149 (GPR149) is an orphan receptor that has recently attracted increasing attention due to its distinctive structure and emerging biological roles. Although initially identified as an “antifertility” gene with high ovarian expression, accumulating evidence suggests a broader physiological relevance, including roles in metabolic and neurological regulation. This narrative review summarizes and integrates current evidence on the structure, expression patterns, and functional roles of GPR149 across multiple biological systems, including reproduction, metabolism, and the central nervous system (CNS), while highlighting existing knowledge gaps and future research directions. GPR149 appears to act as a negative regulator of fertility and ovulation, influencing oocyte maturation and granulosa cell signaling. In metabolic regulation, GPR149 deficiency is associated with resistance to diet-induced obesity and improved insulin sensitivity. In the nervous system, GPR149 expression in glial and neuronal populations has been linked to myelination and neuroendocrine signaling, primarily through modulation of the MAPK/ERK pathway. Altered expression of GPR149 under diabetic and neurological conditions further supports its multifunctional role. Collectively, current evidence positions GPR149 as a multifunctional orphan GPCR involved in the regulation of reproductive, metabolic, and neural processes. Elucidating its ligand interactions and signaling mechanisms may open new therapeutic avenues for reproductive disorders, metabolic syndromes, and neurodegenerative diseases.
Key words: signaling pathways, neural development, GPR149
Introduction
G protein-coupled receptors (GPCRs) are a large and diverse family of membrane proteins that play a key role in transmitting signals from outside to inside the cell, thereby regulating numerous physiological processes, including those in the nervous system.1 They mediate a wide range of cellular responses to most neurotransmitters and hormones in the human body and exhibit extensive structural and functional diversity, enabling them to respond to various endogenous ligands.2 Because of their crucial roles in maintaining cellular and tissue physiology and homeostasis, GPCR-related signaling pathways are implicated in the pathophysiology of numerous diseases, including metabolic, immune, and neurodegenerative disorders, as well as cancer and infectious diseases.3, 4 G protein-coupled receptor 149 (GPR149) is a GPCR with a conserved C-terminal domain of unknown function and remains poorly characterized. GPR149 encodes a seven-transmembrane receptor belonging to the class A GPCR family. Although classified as a class A GPCR, the protein lacks the first 2 charged amino acids of the highly conserved Asp-Arg-Tyr (DRY) motif in the 3rd transmembrane helix, which is critical for efficient G protein-mediated signal transduction.5
Computational analyses predict that GPR149 is a class A rhodopsin-like GPCR consisting of 732 amino acids.6, 7 Well-defined orthologs of this receptor have been identified in various vertebrate species, including mice and humans.8 GPR149 possesses a highly conserved C-terminal domain of approx. 360 amino acids (in mice) that shows no similarity to other known proteins and may be involved in downstream signaling. However, its specific signaling pathways and endogenous ligands remain to be identified.6 GPR149 has been detected in the dorsal root ganglion and gonads of chicken embryos,9 in the ventromedial hypothalamus of mice,10 and in astrocytes differentiated from a rat glial progenitor cell line.11 GPR149 may form dimers with other GPCRs expressed in oocytes, which could influence its function or cellular localization.12 Its expression is low in newborn ovaries but gradually increases during folliculogenesis.8 GPR149 has been identified as an “antifertility” gene expressed in the ovary and oocytes, making it of particular interest. It was discovered through database searches for genes with expression patterns similar to that of GDF9.13 Using Gene Ontology (GO) classification, these genes were identified as GPCRs, among which GPR3, GPR149, and GPR175 were highlighted as potentially important.14, 15 Evidence indicates that GPR149 deficiency does not alter the number of OL precursor cells but instead promotes their differentiation into OLs, leading to earlier myelination.7 It has also been reported that GPR149 exhibits a distinct expression pattern in the kidney.16
Objectives
Despite being identified over a decade ago, GPR149 remains one of the least characterized members of the GPCR family. Its unique structural features, including a modified DRY motif and a highly conserved C-terminal domain, together with its tissue-specific expression in the ovary, brain, and kidney, suggest specialized functions that have yet to be elucidated. Early studies indicate a potential role for GPR149 in the regulation of ovarian folliculogenesis, OL differentiation, and myelination, highlighting its multifaceted physiological significance. However, to date, no narrative review has comprehensively summarized current knowledge on GPR149, including its structural characteristics, expression patterns, and potential biological roles. This review aims to synthesize existing evidence and provide insights into the unresolved aspects of GPR149, thereby offering a foundation for future functional and mechanistic studies.
GPR149 structure
The GPR149 gene encodes a protein belonging to the class A family of 7-transmembrane GPCRs. GPR149 contains a highly conserved C-terminal domain of approx. 360 amino acids that shows no similarity to other known proteins and may be involved in downstream signaling.6 This extended C-terminal region may play a role in receptor trafficking, intracellular signaling, or interactions with scaffold proteins such as β-arrestins. Similar extended C-terminal domains in other GPCRs have been implicated in receptor regulation and signaling bias. However, the specific signaling pathways and endogenous ligands of GPR149 have not yet been identified. Phylogenetic analysis of human GPCRs indicates that GPR149 shares the greatest sequence similarity with receptors that bind peptide ligands.6 GPR149 contains a modified DRY motif in the 3rd transmembrane helix, a sequence essential for efficient G protein-mediated signal transduction.17 Consequently, it is classified as a class A orphan GPCR.18 GPR149, also known as PGR10 (progesterone-responsive gene 10) or IEDA, is a membrane protein composed of 731 amino acids (UniProtKB: Q86SP6; GP149_HUMAN) that spans the membrane multiple times. The absence of conserved Asp–Arg residues within the DRY motif of GPR149 may affect its ability to efficiently couple to G proteins. Such variations have been associated with altered receptor activation, reduced G protein signaling, or a preference for non-canonical signaling pathways. GPR149 functions as an orphan receptor and belongs to the GPCR family. The GPR149 gene is located on human chromosome 3q25.2, a region that contains more than 1,100 genes, including a cluster of chemokine receptor (CKR) genes and several loci associated with human cancers.19 A key feature of GPR149 is its unusually long C-terminal region, consisting of approx. 368 amino acids.15, 18 Figure 1illustrates the structure of GPR149.
GPR149 in reproductive system
Edson et al.8 conducted a study investigating the relationship between GPR149 and fertility in a mouse model. They found that GPR149 is highly expressed in oocytes, with expression levels increasing during follicular development and decreasing after fertilization. The absence of GPR149 leads to increased fertility and ovulation. GPR149 was not expressed in granulosa cells but was highly expressed in germinal vesicle-stage and metaphase II oocytes. After fertilization, GPR149 expression decreased and became undetectable by the 2-cell stage. To investigate the role of GPR149 in oocyte growth and maturation, GPR149 knockout mice were generated. These mice were viable and exhibited normal folliculogenesis but showed increased fertility, enhanced ovulation, elevated levels of oocyte growth differentiation factor 9 (GDF9) mRNA, and increased expression of follicle-stimulating hormone (FSH) receptor and cyclin D2 mRNA in granulosa cells.8 These findings suggest that GPR149 may act as a negative regulator of fertility by suppressing pathways involved in granulosa cell proliferation and oocyte maturation. Another study reported that GPR149 is associated with reproductive traits, demonstrating high haplotype similarity among Chinese breeds.20 In mammals, GPR149 is highly expressed in the ovary and brain, whereas knockout mice exhibit increased fertility and enhanced ovulation.21 Notably, GPR149 is also highly expressed in the pituitary gland of domestic chickens.22
Thus, GPR149 appears to play a regulatory role in reproductive physiology by acting as a negative modulator of fertility and ovulation. Its expression pattern – characterized by high levels in oocytes during follicular development and a sharp decrease after fertilization – suggests a role in controlling oocyte maturation and early embryonic competence. Mechanistically, GPR149 is likely to function through G protein-mediated signaling pathways to suppress downstream signaling molecules involved in follicular growth and hormone responsiveness. The increased expression of oocyte GDF9 follicle-stimulating hormone receptor (FSHR), and cyclin D2 (Ccnd2) mRNAs in GPR149-null mice indicates that GPR149 normally inhibits pathways promoting granulosa cell proliferation and oocyte maturation.
These findings are consistent with previous studies demonstrating a regulatory role for GPR3 and GPR12 in spontaneous oocyte maturation.12 In addition, the ability of GPR149 to form dimers with other GPCRs may further modulate its signaling specificity, influencing receptor trafficking, ligand sensitivity, and downstream signaling. Although its endogenous ligand and precise signaling cascade remain unidentified, the conservation of the C-terminal domain suggests an evolutionarily preserved regulatory mechanism, potentially involved in intracellular signaling, scaffolding, or receptor localization.
Notably, the distinct expression of GPR149 in the pituitary gland of chickens suggests possible interspecies variation in its reproductive role, indicating that GPR149 may integrate neuroendocrine cues with ovarian function and exhibit species-specific regulatory properties (Figure 2). This observation warrants further comparative studies to determine whether the inhibitory effect of GPR149 on fertility observed in mammals is conserved in avian species.
GPR149 in metabolism
Wyler et al.6 investigated the role of GPR149 in metabolism and reported low levels of GPR149 expression in the gastrointestinal tract. The highest expression levels were observed in the islands of Calleja and adjacent nuclei, including the olfactory tubercle and the ventromedial hypothalamus. GPR149–/– mice were viable and born at the expected Mendelian ratios, with no apparent abnormalities. The absence of hypothalamic GPR149 expression in knockout mice was confirmed using quantitative polymerase chain reaction (qPCR). When fed a standard chow diet, GPR149–/– mice showed a tendency toward slower weight gain. Upon switching to a high-fat diet (60% fat), these mice exhibited significantly reduced diet-induced obesity compared with wild-type controls, whereas GPR149+/– mice displayed an intermediate phenotype. Furthermore, both GPR149–/– and GPR149+/– mice exhibited increased insulin sensitivity compared with control mice.
The authors concluded that GPR149 remains classified as an orphan receptor, as its endogenous ligand(s) and downstream signaling pathways have not yet been identified. The findings suggest that GPR149 acts as a regulator of energy balance, likely through central mechanisms involving the hypothalamus and other brain regions, possibly via Gi/o protein-mediated pathways affecting intracellular cAMP levels. GPR149 has also been proposed as a potential therapeutic target for metabolic disorders, including diabetes and its complications, such as diabetic nephropathy.16 These conditions are associated with dysregulation of intracellular cAMP signaling pathways involved in renal cell function, inflammation, and fibrosis. In addition, reduced expression of GPR149, GPR153, and GPR176 has been observed in the kidneys under diabetic conditions.16 Based on bioinformatic analyses, these receptors may couple to Gi/o proteins. GPR149 may therefore influence renal function and metabolic complications by modulating Gi/o-dependent signaling, highlighting a potential link between central nervous system (CNS)-mediated metabolic regulation and peripheral organ function (Figure 3).
This finding is significant, as alterations in Gi/o-mediated signaling have been implicated in key pathological mechanisms underlying diabetic nephropathy.16 Accordingly, reduced renal expression of these orphan Gi/o-coupled receptors in diabetes may contribute to the development and progression of diabetic nephropathy. Several G protein-coupled receptors, including GPR149, have also been reported to be differentially expressed in the habenular region.23 This observation suggests that GPR149 may modulate feeding behavior, reward circuits, or autonomic regulation, providing a broader central mechanism for its metabolic effects. Despite its initial classification as an orphan receptor, emerging evidence supports a role for GPR149 in metabolic regulation. GPR149 is expressed at relatively low levels in the gastrointestinal tract, while functional studies in knockout mice have shown that its deletion leads to reduced body weight gain and protection against diet-induced obesity. These findings indicate a role for GPR149 in glucose metabolism and insulin signaling, potentially linking CNS control of energy balance with peripheral metabolic outcomes. These results are consistent with previous studies demonstrating roles for GPR35 in adipose tissue energy homeostasis and inflammation,24 and for GPR12 in lipid metabolism.25
GPR149 in central nervous system
Mouse GPR149 is highly expressed in the CNS.8 It is strongly expressed in brain regions involved in energy regulation, including the ventromedial hypothalamus, olfactory tubercle, and islands of Calleja. GPR149 shows moderate to low expression in the nucleus accumbens, central amygdala, and ventral tegmental area. It is also clearly detected in striatal subregions, including the caudate-putamen and nucleus accumbens.26 This region-specific expression pattern suggests that GPR149 may contribute to the integration of metabolic, emotional, and motivational signals within the CNS (Figure 4).
GPR149 has been identified as one of the most highly expressed orphan receptors in vagal afferent neurons.27 Its enrichment in vagal pathways suggests a potential role in gut–brain communication and autonomic regulation, both of which are central to energy balance and homeostasis. GPR149 is also expressed at significantly higher levels in the brain than in other tissues.28 A short cDNA fragment of GPR149, designated R35, was first identified through RNA fingerprinting in 1997.29 Subsequently, the complete R35 sequence was obtained from chick embryonic day 8 (E8) dorsal root ganglia (DRGs), and its hydropathy profile revealed the characteristic structure of a seven-transmembrane receptor.9 This early identification in sensory ganglia supports a conserved role for GPR149 in neural signaling.
GPR149 and TRPM8 have been reported to be selectively enriched in peptidergic nociceptors in the DRG and trigeminal ganglia (TG). However, GPR149 shows higher enrichment in DRG peptidergic nociceptors, whereas TRPM8 is more highly expressed in TG peptidergic nociceptors.30 This differential distribution suggests that GPR149 may be more closely associated with spinal sensory processing than with cranial nociception. GPR149 expression increases during the early stages of astrocyte differentiation but not during OL differentiation in the CG-4.B8 subclone of the rat glial precursor cell line CG-4.11 Quantitative reverse transcription PCR (qRT-PCR) and double fluorescence in situ hybridization (dFISH) analyses have shown that GPR149 is highly expressed in oligodendrocyte precursor cells (OPCs) and decreases during oligodendrocyte (OL) differentiation.7 This pattern supports the hypothesis that GPR149 may function as an inhibitory regulator across multiple physiological systems, including neural and reproductive pathways. In addition, hypermethylation of the GPR149 locus has been associated with shorter progression time in clear cell renal cell carcinoma, although further studies are required to establish its prognostic value.31
Deletion of GPR149 in mice has been shown to result in transiently accelerated OL differentiation and increased myelination during development.7 Mechanistic studies indicate that GPR149 constitutively activates the MAPK/ERK pathway, and inhibition of ERK signaling nearly abolishes its suppressive effect on OPC differentiation into OLs. These findings suggest that GPR149 regulates myelin development and regeneration through MAPK/ERK signaling. Overall, GPR149 appears to act as a negative regulator of OPC differentiation, myelination, and remyelination. Targeting GPR149 may therefore represent a promising therapeutic strategy to enhance myelin repair in demyelinating disorders.7
Several studies have shown that multiple orphan GPCRs are highly expressed in the mouse prefrontal cortex, a brain region associated with learning and memory.26 GPR149 has also been linked to neuroendocrine signaling and is prominently expressed in inhibitory interneurons within the ventromedial hypothalamus of mice. This localization suggests that GPR149 may modulate inhibitory circuits involved in neuroendocrine and metabolic regulation. Moreover, genetic variants of GPR149 have been identified in studies investigating susceptibility to migraine.28, 32 The elevated expression of GPR149 is particularly noteworthy given its association with inhibitory interneurons and myelination – processes often disrupted in neurological conditions such as amyotrophic lateral sclerosis (ALS), where excitotoxicity and myelination abnormalities have been observed.33, 34 This overlap highlights GPR149 as a potential modulator of neural stability and circuit integrity.
Notably, GPR149 is expressed in several regions of the CNS, including the nucleus accumbens, central amygdala, ventral tegmental area, and striatal subregions such as the caudate-putamen. Its strong presence in vagal afferents and peptidergic nociceptors of the DRG suggests potential roles in sensory transmission and autonomic regulation.
In glial cells, GPR149 expression changes dynamically during differentiation, increasing during early astrocyte development and decreasing as OPCs mature. Functional studies have shown that deletion of GPR149 promotes OPC differentiation and accelerates myelination, indicating that the receptor acts as a negative regulator of myelin formation. Mechanistically, this effect appears to involve constitutive activation of the MAPK/ERK signaling pathway, as inhibition of ERK activity reverses the suppressive effect of GPR149 on OL maturation. These findings highlight GPR149 as a key modulator of myelin development and repair, with potential implications for demyelinating disorders. These results are consistent with previous studies demonstrating roles for GPR37 in neurodegeneration and protein processing,35 as well as in the regulation of OL differentiation and myelination.36 Furthermore, its expression in inhibitory interneurons of the ventromedial hypothalamus, together with reported genetic associations with migraine, suggests additional roles in neuroendocrine signaling and sensory processing (Table 1).6, 7, 8, 12, 23, 24, 25, 26, 27, 28, 31 Despite these emerging insights, the endogenous ligand(s) and precise signaling mechanisms of GPR149 remain unknown, underscoring the need for further mechanistic and translational studies targeting this orphan receptor.
Translational and therapeutic potential
GPR149 holds promise as a therapeutic target across reproductive, metabolic, and neurological systems. In reproduction, GPR149-null female mice exhibit hyperfertility, indicating that GPR149 normally suppresses ovulation. Thus, its inhibition may enhance fertility in clinical or assisted reproductive technology (ART) settings, whereas its activation could support contraceptive strategies, although ligand identification remains necessary.8, 37 Its oocyte-specific expression and pre-ovulatory peak suggest a modulatory role in gonadotropin responses. In addition, its potential as a biomarker of oocyte quality is supported by analogous findings in chickens related to reproductive timing.38 Beyond reproduction, GPR149 is expressed in brain regions associated with energy balance, and knockout studies demonstrate protection against diet-induced obesity and improved insulin sensitivity. These findings implicate GPR149 in glucose regulation and position it as a potential target for the treatment of metabolic syndrome and type 2 diabetes.6 In the CNS, GPR149 is highly expressed in OPCs and is downregulated during myelination. Deletion of GPR149 enhances remyelination both in vitro and in vivo, suggesting therapeutic relevance in demyelinating diseases such as multiple sclerosis (MS).6, 7
Although no GPR149-targeted compounds are currently available, its cell type-specific expression supports its potential as a biomarker. In reproductive settings, GPR149 levels may reflect ovulatory function, whereas in the nervous system, its downregulation during OPCs maturation may indicate remyelination.7 Single-cell RNA sequencing studies may further clarify its utility as a cell-state biomarker, particularly in rare neuronal populations such as those in the islands of Calleja.6, 39 In summary, the selective expression and multifunctional roles of GPR149 confer broad translational potential, ranging from therapeutic targeting in infertility, obesity, and MS to its future development as a biomarker in reproductive and neuroregenerative contexts.40, 41
Comparisons with other orphan GPCRs
GPR149’s therapeutic potential is supported by successful examples of other orphan GPCRs. KISS1R (formerly GPR54), once considered an orphan receptor, became central to reproductive health following identification of its ligand, leading to the development of kisspeptin analogs now in clinical trials for pubertal and fertility disorders.37 Similarly, GPR17, which is expressed in OPCs like GPR149, has emerged as a target in MS, with antagonists promoting remyelination and progressing to clinical trials.6, 42 These findings suggest that GPR149 could follow a comparable trajectory in neuroregenerative or reproductive therapies. In metabolic disease, GPR119 was deorphanized as a lipid-sensing receptor, and several agonists have advanced to clinical trials for type 2 diabetes. Although early candidates showed limited efficacy, ongoing combination strategies underscore the importance of continued therapeutic development.43, 44 A similar approach may be relevant for targeting GPR149 in obesity. Orphan GPCRs have also shown promise in oncology; e.g., GPR87 has been linked to cancer cell survival and is being explored as a potential therapeutic target.39 Collectively, these examples illustrate that, once characterized, orphan GPCRs such as GPR149 can become important clinical targets across diverse disease areas.
Limitations of the study
This narrative review provides an overview of the current knowledge on GPR149, integrating evidence from reproductive, metabolic, and neural studies. However, the lack of identified endogenous ligands and well-defined signaling mechanisms limits a comprehensive understanding of its physiological and pathological roles. Ligand screening for orphan GPCRs relies on a combination of experimental and computational approaches. Traditional reverse pharmacology involves expressing receptors in cell-based systems and screening ligand libraries using signaling readouts such as cAMP or reporter assays, although its efficiency has declined over time.40 High-throughput platforms, including β-arrestin recruitment assays (e.g., Tango and PRESTO-Tango) and luciferase-based reporters, have improved the detection of both basal and ligand-induced activity.39 Many orphan GPCRs exhibit constitutive activity, enabling assessment of baseline signaling through the use of promiscuous G proteins or yeast-based systems, which can guide the selection of appropriate functional assays and the identification of inverse agonists.39, 40 Structure-based approaches have gained increasing attention with the development of tools such as AlphaFold2 and cryo-electron microscopy (cryo-EM), which facilitate virtual ligand screening (VLS) by enabling receptor modeling and docking of large compound libraries, thereby streamlining candidate selection.39 These advances have also revealed intrinsic agonists and bound lipids that may help infer endogenous ligands.41 Overall, integrated in vitro and in silico strategies are increasingly used to deorphanize GPCRs. Further experimental and clinical studies are required to validate these approaches and clarify the therapeutic potential of GPR149.
Conclusions
GPR149 is a multifunctional orphan GPCR with important roles in reproduction, metabolism, and neural development. Current evidence highlights its function as a negative regulator of fertility, a modulator of energy balance and insulin sensitivity, and a contributor to myelination and neuroendocrine signaling. However, the absence of identified endogenous ligands and an incomplete understanding of its signaling pathways limit insights into its physiological and pathological roles. Future studies should focus on identifying GPR149 ligands, elucidating its downstream signaling mechanisms, and exploring its function in human tissues. Sequence analyses indicate that GPR149 is conserved across multiple mammalian species, suggesting an evolutionarily preserved function. Nevertheless, variations in specific regions, including the C-terminal domain and intracellular loops, may contribute to species-specific differences in receptor signaling and regulation. Further investigation of GPR149 as a therapeutic target in reproductive disorders, metabolic diseases, and demyelinating or neurodegenerative conditions may provide novel avenues for clinical intervention.
Use of AI and AI-assisted technologies
Not applicable.

















.jpg)
.jpg)
.jpg)
.jpg)