Abstract
Background. The prevalence of fetal alcohol spectrum disorders (FASD) in Poland is about 2%, but the prevalence of prenatal alcohol exposure (PAE) is much higher than previously reported. In the absence of a single biomarker or imaging test that can confirm the diagnosis, the identification of FASD relies on clinical diagnostic criteria; therefore, the first Polish diagnostic standards were developed in 2020.
Objectives. To present the process of updating the national diagnostic guidelines for FASD in Poland and discuss the key revisions made based on clinical implementation feedback.
Materials and methods. The work was carried out by the members of the Council for FASD Prevention and Treatment at the National Centre for Addiction Prevention in 4 steps: 1) a pilot implementation study; 2) a structured literature review supporting the update; 3) a formalized expert consensus process; and 4) stakeholder consultation.
Results. The updated guidelines include the following sections: Introduction, Methodology, Diagnostic categories, Diagnostic scheme, Ethical considerations, Evaluation of PAE, Evaluation of facial dysmorphism, Evaluation of growth impairment, and Evaluation of the central nervous system (CNS). Appendices containing practical tools useful in the diagnostic procedure are an important element of the recommendations. Key amendments include the introduction of the partial fetal alcohol syndrome (pFAS) diagnostic category; the addition of biomarker analysis as a tool to confirm PAE; simplification of the assessment of facial dysmorphologies; clarification of CNS evaluation; and discussion of the ethical concerns associated with FASD diagnosis.
Conclusions. The updated national guidelines may improve the quality and standardization of FASD diagnosis not only in Poland, but also worldwide. The practical utility of each recommendation should be continuously monitored, validated, and updated.
Key words: Poland, diagnostic guidelines, fetal alcohol spectrum disorders (FASD)
Background
Fetal alcohol spectrum disorders (FASD) constitute an umbrella term describing a range of conditions resulting from prenatal alcohol exposure (PAE). The global prevalence of FASD is estimated at 22.77 per 1,000 individuals (range: 0–176.77 per 1,000).1, 2 The most recent study attempting to assess the prevalence of FASD among Polish children was conducted in 2013 and indicated that at least 2% of children in Poland are affected.3 However, more recent findings suggest that alcohol consumption during pregnancy may be considerably more common than previously reported.4, 5 Despite increasing awareness, FASD remains a significant public health concern both in Poland4, 6 and globally.7
In the absence of a single biomarker or imaging modality capable of confirming the diagnosis,8, 9 the identification of FASD – unlike many other medical conditions – relies primarily on clinical diagnostic criteria rather than laboratory findings. Consequently, standardized diagnostic guidelines remain the gold standard for diagnosing FASD. Since the first description of the dysmorphic features of fetal alcohol syndrome (FAS) by Jones and Smith,10 multiple diagnostic systems for FASD have been developed.
In North America, 4 principal diagnostic systems are currently used: the Institute of Medicine (IOM)/Hoyme et al. criteria,11 the 4-Digit Diagnostic Code,12 the Emory criteria,13 and the Canadian guidelines.14 However, inconsistencies among these systems have been documented.15, 16 Recently, the need for greater standardization of FASD diagnostic guidelines has been strongly emphasized.16 Over the past several years, a number of countries – including Germany,17, 18 Norway,19 Italy,20 and Australia21 – have developed their own national diagnostic guidelines. The first Polish diagnostic standards were developed in 2020.22 In 2023, the National Centre for Prevention of Addictions conducted a nationwide survey to assess their applicability in Polish FASD diagnostic centers.23 The results indicated that the standards were known in detail by about half (58%) of the study participants, mainly because their dissemination was limited (according to 90% of respondents). More detailed results of the survey indicated that the respondents agreed regarding the general value of the document: 100% of them rated the guidelines as practical, useful, and well structured.
The diagnostic scheme was positively assessed by 95% of the survey participants. Respondents’ answers differed regarding diagnostic categories (47% rated them negatively, 52% positively, and 1% left the question unanswered). Common reasons for negative assessments included the view that neurodevelopmental disorder associated with prenatal alcohol exposure (ND-PAE) constitutes overly broad criteria, the need for clearer differentiation between FASD diagnostic categories, and the lack of pFAS as a diagnostic category.
The part of the guidelines concerning PAE assessment (including the Alcohol Use Disorder Identification Test – Consumption (AUDIT-C) and Alcohol Use Disorder Identification Test (AUDIT) as part of it) was rated positively by 68% of the respondents, while the remaining 32% rated it negatively. Common reasons for negative assessments included the statement that biological parents often cannot be reached and that legal guardians rarely have knowledge about PAE; moreover, the diagnosis of PAE requires the ability to conduct a therapeutic conversation due to the mother’s feelings of guilt and shame.
Instructions on the physical examination, particularly in terms of the proposed tools (growth charts), were viewed positively by 89% of the participants. The proposed neuropsychological criteria were approved by 90% of the participants; however, 26% considered the neuropsychological criteria inadequate for young children. These results confirm the need to update and disseminate the FASD diagnostic guidelines among Polish specialists in order to improve healthcare for individuals affected by PAE.
Objectives
This paper summarizes the process of updating the national diagnostic guidelines for FASD in Poland and discusses key revisions made based on clinical implementation feedback. The main objective of this work was to improve the quality of FASD diagnosis and provide solid background for healthcare of individuals affected by PAE. The specific objectives concerned the following stages of work:
1. To evaluate functioning of the first guidelines (from 2020) in clinical practice;
2. To update the knowledge on FASD;
3. To refine and synthesize experts’ opinions and gain consensus on the guidelines update;
4. Stakeholder consultation.
In this paper, we briefly present these 4 stages of the updating process and an abbreviated version of the recently updated Polish diagnostic guidelines already published in the Polish version in its full form.23
Stage 1: Pilot implementation of the 2020 guidelines and their evaluation
The core element of the guidelines updating process was their pilot implementation and the assessment of their functioning and utility in a reference center. The general aim of this stage was to evaluate, in clinical practice, the functioning of the new Polish guidelines and their usefulness in assessing all key diagnostic dimensions in FASD: 1) PAE; 2) growth impairment; 3) dysmorphologies; 4) neurodevelopmental disorders.
Materials and methods
The study consisted of 2 parts. In the 1st part, the records of patients who presented for diagnostic purposes at the St. Louis Children’s Hospital FASD Diagnostic Center in Kraków, Poland, between October 2020 and February 2022 were analyzed. The diagnosis was made by a team of professionals, including a physician (child psychiatrist and/or pediatrician) and a psychologist, according to the Polish criteria. In addition, a diagnosis was made according to the IOM criteria11 based on the clinical data available at the time of analysis. During a 10-month period, 2 diagnostic teams used the guidelines: one with expertise in the field and another with expertise in childhood neurodevelopmental disorders, with minimal previous experience in the diagnosis of FASD. Before implementation, all team members participated in a structured 20-h training program delivered by the guideline co-authors (a physician and a psychologist), consisting of seminars and supervised practical exercises.
In the 2nd part of the study, qualitative data (interviews and written opinions) were collected from health professionals gathering information on PAE. These data provided insights into the practicality of the Polish guidelines and helped explain the differences in diagnosis based on IOM compared to Polish recommendations.
Sample
The records of 86 patients admitted to the clinic were analyzed, of whom 71 completed the diagnostic process. Fifteen patients did not complete the evaluation: some withdrew from the diagnostic process, some were lost to follow-up (the child’s status changed and new caregivers refused to continue the evaluation), and others were referred to other facilities (genetic, neurometabolic, or autism spectrum disorder evaluation) for differential diagnosis, e.g., due to the biological mother’s declaration that she had not consumed alcohol during pregnancy (confirmed no PAE, n = 4).
Among the patients, there were 40 boys and 31 girls. The median age was 96 months (range: 58–210 months). Ten children (14%) were raised by their biological parents (9 by their fathers and 1 by their mother), 31 (44%) were in adoptive families, 20 (28%) were in foster care, and 10 (14%) were living in institutional care (orphanage or childcare facility).
The 2nd group of study participants consisted of members of the FASD diagnostic team (n = 5) and professionals responsible for FASD screening (“screening experts”). The latter group (n = 8) comprised 2 family physicians, 1 neonatologist, 2 obstetrician–gynecologists, 2 school psychologists, and 1 educational psychologist, who applied the screening recommendations in their clinical or educational practice.
Measures
Data on PAE were collected using various methods: direct interview with the biological mother; interview with the biological mother’s relatives or a representative of an institution (adoptive agency, social services, orphanage); information passed to the diagnostic team through other persons; and/or review of the child’s health records. The evaluation of prenatal and postnatal growth impairment included all available data on birth weight as well as current and historical parameters (height and weight). The assessment of facial dysmorphic features was conducted by a clinical geneticist and pediatricians.
The neuropsychological assessment was adjusted to the child’s age and needs. In the case of children up to 3 years of age, the Children’s Development Scale (Dziecięca Skala Rozwojowa (DSR)) was used. For children aged 3 to 5 years, the Intelligence and Development Scales for Preschool Children, the Stanford–Binet Intelligence Scales (SB-5), and the Intelligence and Development Scales-2 (IDS-2) or Wechsler Intelligence Scale for Children (WISC–V) were used to assess emotional and social development.
For older children, the following tools were used: Intelligence and Development Scales (IDS-2), Stanford–Binet Intelligence Scales (SB-5), Raven’s Progressive Matrices, Delis–Kaplan Executive Function System (D-KEFS), WISC–V, Benton Visual Retention Test (BVRT), Rey Auditory Verbal Learning Test (RVLT), and the D2 Test of Attention. If needed, additional clinical assessments were performed, and data from interviews and observations, psychiatric examinations, and results of assessments conducted by other psychologists (if available) were taken into account. All participating professionals (n = 13) provided feedback through written opinion essays and participated in a focus group interview moderated by a member of the project team.
Data analysis
All data were analyzed and discussed by the diagnostic team. In the first step, the team compared each patient’s results in each diagnostic category with the Polish recommendations and classified the patient into one of the following categories: no FASD, FAS, ND-PAE, or FASD risk. The patient’s results were then compared with the IOM diagnostic recommendations, and the patient was assigned to one of the following categories: FAS, pFAS, ARND, or no FASD.
Qualitative feedback from professionals was analyzed thematically to identify recurring facilitators and barriers to guideline implementation. Caregivers of children diagnosed during the implementation period were surveyed separately to obtain patient and family perspectives on the diagnostic process.
Results
In 14 (20%) cases, a diagnosis of FAS was made; 19 (27%) received a diagnosis of ND-PAE; 28 (39%) were considered “at risk of FASD”; and in 10 (14%) cases, FASD was excluded. According to the IOM criteria, 14 (20%) of the patients were diagnosed with FAS, 10 (15%) with pFAS, and 14 (21%) with ARND, while in 33 (42%) cases no FASD diagnosis was made (Table 1).
As shown in Table 1, the consistency of diagnoses based on the Polish22 and IOM criteria11 was 60%. The inconsistencies were due to differences in neurodevelopmental assessment and the inability to confirm PAE. The assessment of growth parameters and dysmorphic features yielded consistent diagnoses.
The guidelines were positively evaluated by professionals and perceived as clear, concise, and clinically useful. Participants particularly emphasized the value of standardized tools supporting both physical and psychological assessments. However, the neuropsychological criteria required to establish an FASD diagnosis were frequently described as relatively restrictive compared with other international guidelines, especially regarding the required number of affected brain domains. Based on the findings from the Stage 1 study, the need for further refinement of selected components of the guidelines was identified.
Stage 2: Structured literature review supporting the update
In December 2023, an evidence-informed literature review was conducted to support the update of the guidelines and to evaluate the current state of knowledge relevant to FASD diagnosis. The PubMed database was searched using the terms “FASD”, “fetal alcohol spectrum disorders”, and “prenatal alcohol exposure”. A total of 150 results were retrieved. The search was further refined using targeted queries focused on dysmorphology and neuropsychological evaluation.
The identified publications were critically evaluated for clinical relevance and potential implications for physical and psychological assessment. Key findings and recent advances were summarized and presented to the working group to inform expert discussions and consensus-based revisions of the updated guidelines.
Stage 3: Formalized expert consensus process
The development of the updated guidelines was assigned to a multidisciplinary working group established on the initiative of the National Centre for Prevention of Addictions. The composition of the working group reflected the interdisciplinary nature of FASD diagnosis and included 6 physicians (a geneticist, pediatric neurologist, pediatric psychiatrist, pediatrician, pediatric gastroenterologist, and obstetrician–gynecologist), 4 psychologists, 1 pedagogue/social therapist, and 2 caregiver representatives.
Due to the limited number of national experts with extensive clinical experience in FASD diagnosis in Poland, the consensus process was based on a repeated expert panel methodology, consistent with an expert opinion consensus approach. The panel worked in an iterative manner, with successive rounds of discussion and refinement of the proposed amendments.
Draft amendments to specific sections of the guidelines were prepared by domain experts according to their areas of clinical expertise (pediatricians and geneticists – growth assessment and evaluation of dysmorphic features; child and adolescent psychiatrist, pediatric neurologist, and neuropsychologist – central nervous system (CNS) evaluation; obstetrician – PAE assessment; psychologist – diagnostic scheme; psychologist – diagnostic categories). These proposals were subsequently discussed during structured virtual and in-person meetings of the working group. An additional element of the guideline development process was a review by a psychologist, the first author of the previous edition of the guidelines.
Consensus was achieved through discussion, incorporation of feedback from all panel members, and revision of recommendations until agreement was reached. In cases of initial disagreement, alternative formulations were discussed and refined in subsequent rounds until a shared position was established.
Stage 4: Stakeholder consultation
A stakeholder meeting was held. The proposed guidelines were presented to the representatives of the diagnostic centers in Poland, a broader discussion was initiated, and the questions were answered.
Results of the process of the guidelines updating
The updated guidelines include the following sections: Introduction, Methodology, Diagnostic categories, Diagnostic scheme, Ethical considerations, Evaluation of PAE, Evaluation of facial dysmorphism, Evaluation of growth impairment, and Evaluation of the central nervous system.
Compared to the 2020 guidelines, a section describing Ethical considerations was added. The list of annexes was reviewed, and 7 annexes were included in the final document.
Annex 1: Feeding problems in infants and toddlers that require evaluation by a speech therapist.
Annex 2: Screening for CNS impairment suggesting the need for referral to a pediatric neurologist.
Annex 3: Behavioral and cognitive characteristics suggesting the need for referral for FASD diagnosis.
Annex 4: Differential diagnosis of FASD.
Annex 5: Structured anamnesis with the birth mother.
Annex 6: A list of standardized tools for neuropsychological evaluation.
Annex 7: Psychiatric evaluation in the diagnosis of FASD.
In all chapters, new information from the literature review was added to illustrate the current state of the art, presenting the crucial amendments made compared to the 2020 guidelines. The full printed guidelines are available.23
Diagnostic categories
In the 2020 guidelines, 3 diagnostic categories were recommended: FAS, ND-PAE, and a descriptive category “at risk of FASD.” The aforementioned categories were maintained in the new guidelines (Table 1). However, a new category was added: partial fetal alcohol syndrome (pFAS) (Table 1), which can be diagnosed in cases of both confirmed and non-confirmed PAE (Table 2).
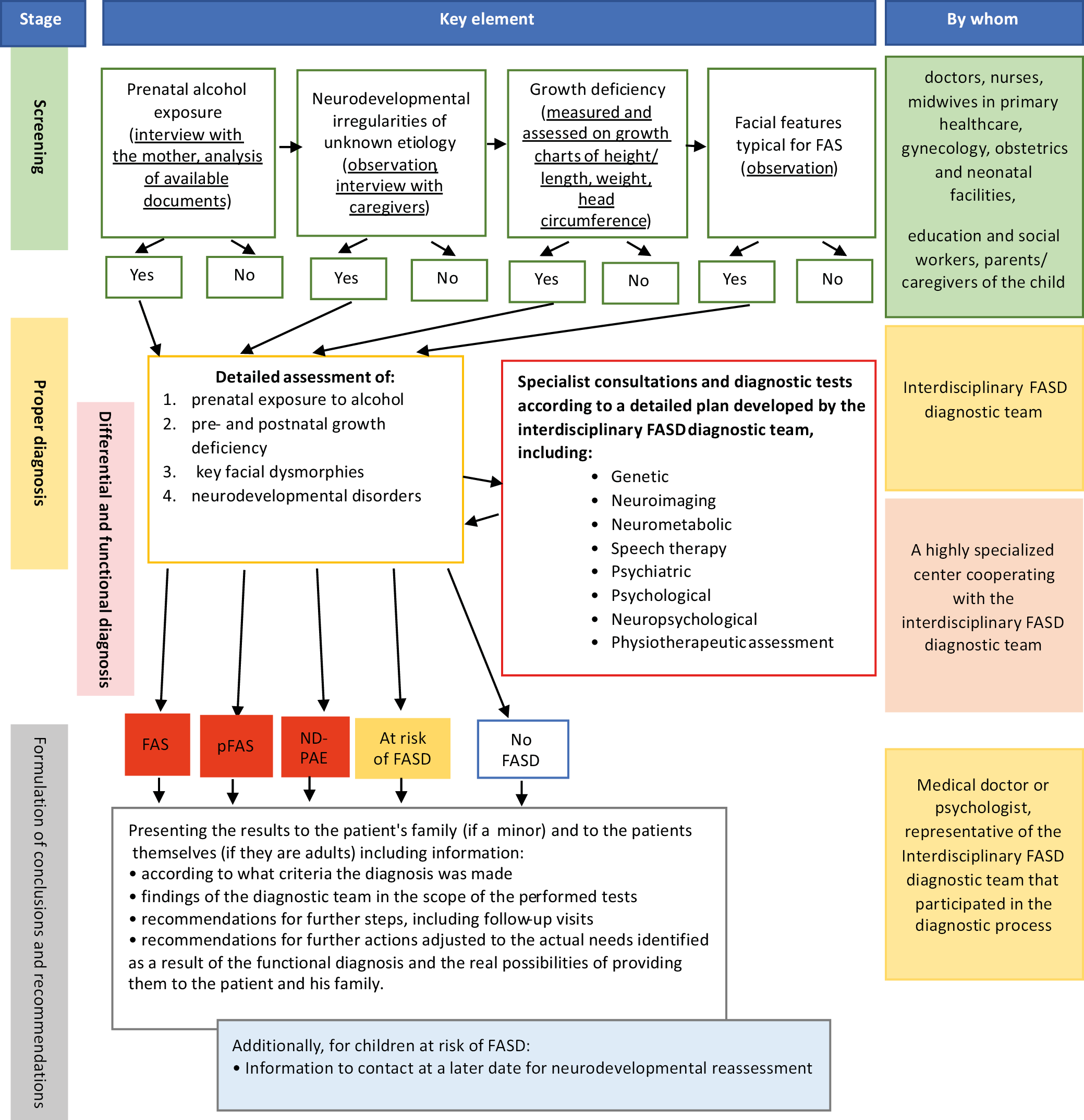
Diagnostic scheme
Consequently, the diagnostic scheme (Figure 1) was reviewed and updated to include the diagnosis of pFAS. The term “functional diagnosis” was removed from the scheme, as it was considered an integral part of the diagnosis itself; however, it was clarified that functional assessment is not equivalent to the diagnosis of FASD. The role of standardized tools was emphasized not only during the diagnostic process but also during screening. The pFAS category was retained for pragmatic reasons. In the Polish clinical and legal context, it allows for the classification of children with clear dysmorphic features who do not meet the full criteria for FAS but still require support.
This ensures that such children are not left without a diagnosis or access to adequate services. The authors emphasize that this is a transitional solution that can be revised in the future with broader implementation of ICD-11 and ND-PAE. However, the “at risk of FASD” category was maintained because the authors agreed that some children may be missing components of key diagnostic criteria at the time of presentation in the diagnostic setting. For these patients, it was considered beneficial to monitor them in case they meet the diagnostic criteria in the future.
Evaluation of prenatal alcohol exposure
According to the 2020 guidelines, there were 3 methods to evaluate PAE22: direct anamnesis obtained from the birth mother; indirect anamnesis obtained from a person who accompanied the birth mother during her pregnancy; and medical, judicial, or employment records, as well as broadly understood social welfare documentation. Due to the difficulty of obtaining reliable answers to questions about alcohol consumption during pregnancy through the interview method,5 a new section was added in the 2025 guidelines to the chapter illustrating the role of biomarkers in the evaluation of PAE.
Ethyl glucuronide (EtG) and phosphatidylethanol (PEth) were considered the 2 biomarkers with the most established role in confirming PAE. However, PEth can be detected in maternal blood only within a limited time frame of 4–6 weeks after alcohol consumption.24 On the other hand, EtG accumulates in human hair, and its detection can reflect alcohol consumption throughout pregnancy across all 3 trimesters.25 The promising role of biomarkers in the evaluation of PAE was highlighted; however, information regarding the requirement for mandatory maternal consent was also included.
Evaluation of facial dysmorphism
The evaluation of facial dysmorphism in the 2020 guidelines involved palpebral fissure measurement (using 2 methods: traditional with a ruler and a computerized method) and lip/philtrum assessment with the lip/philtrum guide.22 The aforementioned procedure was maintained in the 2025 guidelines. However, in the new guidelines, the use of only one selected method for assessing the length of the palpebral fissure was approved. In addition, a table with more subtle dysmorphic features was added to illustrate the complexity of the physical examination in the FASD diagnostic process.
Evaluation of growth impairment
The evaluation of prenatal and postnatal growth impairment was included in the 2020 guidelines. It involved birthweight and current and historical parameters (height and weight) analysis according to growth charts. The procedure remained unchanged in the 2025 guidelines. However, an amendment regarding growth charts was made: after the age of 3, the use of the 2010 Polish (OLA/OLAF) growth charts26 was recommended instead of the 1999 growth charts previously recommended.27
Evaluation of central nervous system
The evaluation of the CNS was the most comprehensive revision of the chapter. The levels of evaluation (neurological, neurodevelopmental, and neurobehavioral) were specified. Each level was characterized by potential pathologies that may indicate CNS impairment. In the neurological evaluation, microcephaly, CNS malformations observed in previously performed imaging, or epilepsy of prenatal origin were considered key neurological symptoms. However, it was underlined that the presence of features of cerebral palsy, focal symptoms, central deafness, or other neurological symptoms indicates the need for consultation with a pediatric neurosurgeon.
Neurodevelopmental evaluation encompasses the assessment of motor, sensory, and perceptual functions, as well as the assessment of the articulatory mechanism and oral praxis, primary orofacial functions, respiratory pattern, and the quality of speech and communicative competencies. Neurobehavioral evaluation consists of the assessment of the following domains: cognition (intelligence, executive functions, learning, memory, visuospatial competencies), self-regulation (emotional regulation, attention, impulse control), and adaptive functioning (communication, social cognition, motor functions, everyday coping strategies).
The evaluation of the 3 levels should be accompanied by a functional analysis, including the assessment of psychopathological symptoms and functioning in home and school settings. A section on concomitant psychopathological diagnoses was added. Significant CNS impairment was defined as the presence of: a) 1 key neurological symptom (microcephaly, CNS malformation, or epilepsy of prenatal origin); b) deficits in ≥3 neurocognitive areas; or c) ≥1 symptom in each domain (neurocognitive, self-regulation, adaptive functioning).
Discussion
The Polish FASD diagnostic criteria developed in 2020, although generally positively received, were poorly known among specialists and criticized mainly for the requirements for diagnosing PAE, which were difficult to apply in clinical practice.24 Pilot implementation of these guidelines (stage 1 of the updating procedure) largely confirmed both the expectations and concerns of Polish experts related to the assessment of PAE. First of all, in retrospective studies, i.e., when the diagnostic procedure is conducted in the case of a child aged several or more than a dozen years, collecting reliable information about the alcohol consumption of the pregnant mother is very difficult and often impossible. As a result, if PAE is considered “probable”, patients with neurodevelopmental disorders but without characteristic facial features are, according to the 2020 Polish recommendations, classified as “at risk” of FASD, whereas according to the IOM criteria, FASD is ruled out in these patients.
Another cause of inconsistent diagnoses, depending on the recommendations used, was the criteria for assessing the CNS. The 2020 Polish criteria were more restrictive in this regard than the IOM criteria. As a result, diagnostic consistency between the recommendations was 60%, which can be considered high. In studies comparing diagnoses based on the IOM criteria and the 4-Digit Code, consistency was 38%.28
In the absence of a single diagnostic or imaging test to confirm FASD, diagnostic criteria remain the gold standard. The proposed 2025 Polish guidelines are consistent with international recommendations; however, they also represent an attempt to adapt existing scientific data to the Polish context (e.g., specific growth charts, validated psychometric tools for evaluation). From a content perspective, the Polish recommendations are in agreement with other widely used FASD diagnostic guidelines. Regarding diagnostic categories, they largely reproduce the nomenclature from the guidelines developed by Hoyme et al.11
However, alcohol-related neurodevelopmental disorder (ARND) is instead termed ND-PAE to avoid a definitive causal interpretation. Nevertheless, this category does not fully overlap with DSM-5 ND-PAE,29, 30 in which a diagnosis of ND-PAE can be made with or without the presence of dysmorphic features. More similarities can be found between the definition in the 2025 Polish guidelines and the Canadian guidelines.14 The addition of a new diagnostic category, pFAS, may be viewed as controversial. However, the authors return to a system in which children presenting with 2 out of 3 dysmorphic features are treated as a separate group.
The category was added following suggestions from professionals who pointed out that patients presenting with 2 dysmorphic features, growth restriction, and neurobehavioral problems but with unknown PAE are left without a formal diagnosis. The “at risk” category was maintained, as professionals underlined its utility in cases where some information is lacking. Children assigned to this category remain in the diagnostic process and can be transferred to other categories if new information emerges, such as confirmed PAE (previously unknown) or the development of neurobehavioral problems.
The dysmorphological evaluation and its principles do not differ substantially among the available diagnostic guidelines, which are based on the lip and philtrum guide and palpebral fissure measurements.11, 12, 14 An important difference between the 2025 and 2020 Polish guidelines is that the process was simplified by allowing only 1 evaluation method instead of two. However, no other diagnostic guidelines11, 12, 14 provide for double verification of palpebral fissure length, and, in the opinion of professionals, this approach hindered the implementation of the guidelines.
Neuropsychological evaluation is a key component of FASD diagnosis. The undisputed advantage of the Polish guidelines is that they are accompanied by a list of validated and available psychometric tools recommended in Poland. In the amended recommendations, the role of structural brain defects such as microcephaly is emphasized. This is especially important in diagnosing young children, among whom subtle neuropsychological symptoms may not be evident. Regarding alcohol exposure, the 2025 Polish guidelines, in light of the latest research, are the first to suggest the use of biomarkers in the process of PAE assessment.
Furthermore, the path to population screening is still distant, and biomarkers need to be evaluated in terms of cost-effectiveness and technological availability. Therefore, the use of biomarkers remains a methodologically justified possibility that may be incorporated into the diagnostic process following further expansion of medical technology. Moreover, the use of biomarkers is only possible with the informed consent of the mother, which is consistent with previous international publications on this matter.8, 31
The authors of the 2025 guidelines emphasize that they should be reviewed and updated within a 5-year period; however, their applicability will be continuously monitored. The 2020 guidelines were created in the hope that they would be the first step toward establishing an integrated system of care for children with FASD in Poland. The authors of the 2025 guidelines emphasize that systemic, government-coordinated actions are urgently needed, as relatively few steps have been taken in the past 5 years and FASD diagnosis is still not covered by the public healthcare system. Although data on alcohol consumption among Polish women are alarming,4 and only some preventive efforts seem to be effective,6 FASD remains a marginalized health issue. The authors of the guidelines believe that the updated version will be widely distributed not only to diagnostic centers but also to primary care offices, regional centers, and obstetric/neonatology departments. Within the framework of the project funded by the National Centre for Prevention of Addictions, printed copies of the guidelines were distributed nationwide.
In the 2025 guidelines, an effort has been made to simplify the diagnostic pathway – this could potentially reduce the entry barrier for new diagnostic settings. An additional advantage of the 2025 guidelines is their potential to improve daily clinical practice. By introducing a simplified diagnostic process and recommending standardized tools available in Poland, they can streamline diagnostic work not only in referral centers but also in smaller facilities. This, in turn, could reduce regional disparities in access to FASD diagnosis in Poland (Table 3).
Limitations
Although the update of the 2020 guidelines22 is an important step toward improving the system for addressing FASD-related problems, the limitations of our work must be acknowledged. As recommended in the 2020 guidelines, evaluation in the clinical setting was the first step in verifying the guidelines (Stage 1). However, feedback was collected at only a single specialized facility. Therefore, it is uncertain to what extent the conclusions from this study can be generalized.
Second, although a literature search was conducted to update the guidelines with the most recent evidence and methods (Stage 2), most modifications were made based on feedback from professionals in the field (Stage 3). Therefore, in the summary of the 2025 guidelines, it was suggested that future updates should be entrusted to scientific societies.
Conclusions
The 2025 guidelines have been positively received as a unified diagnostic tool, and their implementation in clinical practice has provided valuable feedback that prompted the re-evaluation of several recommendations. Based on this experience, the updated guidelines are now expected to be widely disseminated and supported by structured training for health professionals, which may further reduce the diagnostic gap, improve consistency between centers, and strengthen interdisciplinary collaboration, thereby increasing momentum for systemic solutions to this complex health issue.
The 2025 Polish guidelines not only standardize diagnostic procedures but also set the stage for coordinated national strategies in the prevention, diagnosis, and care of individuals affected by FASD.
Data Availability Statement
The datasets supporting the findings of the current study are openly available in Zenodo at https://doi.org/10.5281/zenodo.18470451.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.