Abstract
Background. Crohn’s disease (CD) is a non-specific inflammatory bowel disorder for which no definitive cure is available. The primary management strategy is pharmacological treatment aimed at alleviating symptoms. However, many patients ultimately require surgical intervention to manage complications arising from the disease.
Objectives. The aim of this study was to investigate disease-related factors that may increase the risk of early postoperative complications in patients with CD.
Materials and methods. A meta-analysis was conducted based on studies examining early surgical and medical complications following abdominal surgery for CD. The analyzed risk factors included disease duration prior to surgery, history of previous surgeries, presence of concurrent perianal disease, intra-abdominal abscess during surgery, and Montreal classification subtypes A1–3, L1–4, and B1–3. A systematic review was performed using 4 major databases: PubMed, Cochrane Library, Academic Search Ultimate (EBSCO), and Google Scholar. Outcomes were assessed using the odds ratio (OR) and response ratio (R), together with 95% confidence intervals (95% CIs). Egger’s test was used to evaluate publication bias. Heterogeneity was assessed using the I2 statistic, with I2 > 50% indicating significant variability.
Results. A total of 51 articles met the inclusion criteria. The analysis identified several significant risk-increasing factors: history of previous surgeries (OR = 1.39; 95% CI: 1.23–1.57), Montreal classification group B3 (OR = 1.26; 95% CI: 1.11–1.42), disease duration before surgery (R = 1.10; 95% CI: 1.02–1.18), and group L2 (OR = 1.38; 95% CI: 1.11–1.72). Conversely, factors associated with a reduced risk of postoperative complications included group L1 (OR = 0.81; 95% CI: 0.71–0.92) and group B2 (OR = 0.81; 95% CI: 0.71–0.91).
Conclusions. This meta-analysis aggregated data from a broad spectrum of patients and treatment settings across multiple institutions worldwide. Although some risk of bias and heterogeneity was observed, the findings nevertheless highlight the importance of considering disease subtype and progression when assessing the likelihood of postoperative complications in patients with CD. This knowledge may be valuable for optimizing treatment strategies.
Key words: Crohn’s disease, postoperative complications, risk factors, abdominal surgery, disease progression
Introduction
Crohn’s disease (CD) is classified as a non-specific inflammatory bowel disease characterized by transmural inflammation of the gastrointestinal tract that may occur in any segment, from the mouth to the anus. As the exact etiology of the disease remains unknown, no definitive cure is currently available.1 The primary approach to managing CD involves pharmacological treatment aimed at alleviating symptoms. Nevertheless, many patients ultimately require surgical intervention, including major intra-abdominal procedures such as adhesiolysis, resection of the small or large intestine, strictureplasty, or drainage of intra-abdominal abscesses, to manage complications arising from the disease.
Although CD can affect any segment of the gastrointestinal tract, it most commonly involves the ileocecal region. Inflammation may lead to intestinal strictures and adhesions in some cases, whereas in others it may result in fistulas or intra-abdominal abscesses. Many patients also develop perianal abscesses and fistulas, which present additional clinical challenges. This heterogeneity of disease presentation is reflected in the Montreal classification system, which evaluates the following parameters: age at disease onset, disease location, and disease behavior. The Montreal classification was first introduced at the World Congress of Gastroenterology in Montreal in 2005.2
Research suggests that the severity of CD may be influenced by its specific subtype.3, 4, 5 Indicators such as perianal manifestations, early age at diagnosis, and the ileocolonic subtype may be associated with a more severe disease course.6, 7 Severe disease courses are often accompanied by chronic inflammation, significant malnutrition, and deficiencies in energy and protein, as well as the need for long-term steroid therapy, all of which are recognized risk factors for postoperative complications. Factors associated with a complicated disease course may therefore contribute to an increased risk of unfavorable postoperative outcomes.8
Objectives
When considering treatment options, it is essential to evaluate the likelihood of complications. The aim of this study was to investigate disease-related factors that may influence the risk of early postoperative complications.
Materials and methods
This paper forms part of the research conducted for the doctoral thesis of one of the authors, entitled “Multifaceted Assessment of Perioperative Risk Factors in Patients with Crohn’s Disease (CD).” The findings presented in this study adhere to the guidelines established in the MOOSE (Meta-analysis of Observational Studies in Epidemiology) and PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) 2020 statements.
Information sources and search strategy
The first author (J.Ł.) conducted a systematic literature search using the PubMed, Cochrane Library, Academic Search Ultimate (EBSCO), and Google Scholar databases between November 21 and December 4, 2021. The search included the following terms: “Crohn’s disease,” “Crohn’s disorder,” “IBD,” “regional enteritis,” combined with “complications,” “outcomes,” “morbidity,” “recurrence,” “relapse,” “remission,” “treatment outcome,” “treatment failure,” and “reoperation,” as well as “postoperative,” “perioperative,” “risk factors,” “contributing factors,” “predisposing factors,” “surgery,” and “resection”. Different combinations of these terms were used with the logical operators “AND” and “OR”. The search included articles published up to December 4, 2021. The authors did not contact researchers to obtain unpublished studies. On July 15, 2025, the databases were re-examined after a 3.5-year interval using the same search strategy to update the literature review. Articles published in all languages were considered. To broaden the search results, the “related articles” function in PubMed and the reference lists of the included studies were also screened.
Eligibility criteria
The selection of articles for the meta-analysis was guided by the PICO framework. Participants: Patients aged >15 years, regardless of race or sex, undergoing abdominal surgery for CD. Intervention: Disease-related risk factors, including disease duration prior to surgery, history of previous surgical procedures, presence of concomitant perianal disease, intra-abdominal abscess during surgery, and disease subtypes A1–3, L1–4, and B1–3 according to the Montreal classification (Table 1). Comparator: Presence vs absence of specific risk factors. Outcome: All complications occurring within 30 days after surgery, as defined by the ACS-NSQIP.9 (Table 2)
Complications were stratified into 5 severity grades (1–5) according to the Clavien–Dindo classification system (Table 3). Articles were included in the analysis if they met the following criteria: reported exact event numbers, provided odds ratios (ORs) with corresponding 95% confidence intervals (95% CIs), reported mean values for the analyzed risk factor in the complication group, and included identifiable corresponding data for the control group. The following study designs were eligible for inclusion: observational studies, randomized controlled trials (RCTs), non-randomized controlled trials, prospective cohort studies, retrospective cohort studies, case–control studies, and cross-sectional studies.
Studies were excluded if they contained insufficient data or represented duplicate publications involving the same group of patients. In addition, studies involving animals, pregnant patients, or individuals diagnosed with indeterminate colitis or ulcerative colitis were excluded. Studies were also excluded if they focused on procedures such as cholecystectomy, organ transplantation, or surgeries related to perianal disease. Furthermore, studies with a follow-up period shorter than 30 days, unclear follow-up duration, or lacking outcome data for a comparison group were excluded from the analysis.
Data collection process
The primary author used Mendeley Desktop v. 1.19.8 (Mendeley Ltd., London, UK) and Microsoft Excel 2010 (v. 14.0.7268.5000; Microsoft Corp., Redmond, USA) to review the selected studies. The extracted data included the study title, authors, year of publication, study design, country of origin, type and date of surgical procedures performed, as well as postoperative complications and their associated risk factors. Any discrepancies were resolved through discussion with the 5th author (M.M.), who served as the principal investigator. Articles written in languages other than English were translated using Google Translate (Google LLC, Mountain View, USA).
Quality assessment
The quality of the included studies was assessed by the first author (J.Ł.) using the Newcastle–Ottawa Scale (NOS). Studies with a score of ≥5 points were considered to be of high quality.
Statistical analyses
Statistical analyses for the meta-analysis were performed using Statistica v. 13.1 (StatSoft Inc., Tulsa, USA). Given the anticipated heterogeneity among studies, a random-effects model was applied. All statistical tests were two-tailed, and p < 0.05 was considered statistically significant. The results were presented in forest plots illustrating 2 effect measures. Odds ratios were calculated for dichotomous outcomes and presented with 95% CIs. Continuous variables, expressed as means with standard deviations (SDs), were analyzed using the response ratio (R), defined as the ratio of the mean value in the treatment group to that in the control group, together with 95% CIs. A chronological cumulative analysis was performed to evaluate how the cumulative effect estimate and its standard error (SE) changed over time with each successive publication. Statistical heterogeneity among studies was assessed using the I2 statistic, with values >50% indicating substantial heterogeneity. Publication bias was assessed using Egger’s test, with p < 0.1 considered indicative of significant asymmetry, and the results were visualized using funnel plots. Sensitivity analyses were conducted to evaluate the influence of individual studies on the pooled effect estimates by sequentially excluding each study from the analysis.
Evaluation of the strength of evidence
The strength of evidence was determined based on 3 criteria, as previously described.10 These criteria included Egger’s test p ≥ 0.1, a total study population exceeding 1,000 patients, and I2 < 50%. Level I strength of evidence (high quality) was assigned when all 3 criteria were satisfied; level II (moderate quality) when 2 criteria were met; level III (moderate quality) when 1 criterion was met; and level IV (low quality) when none of the criteria were fulfilled.
Results
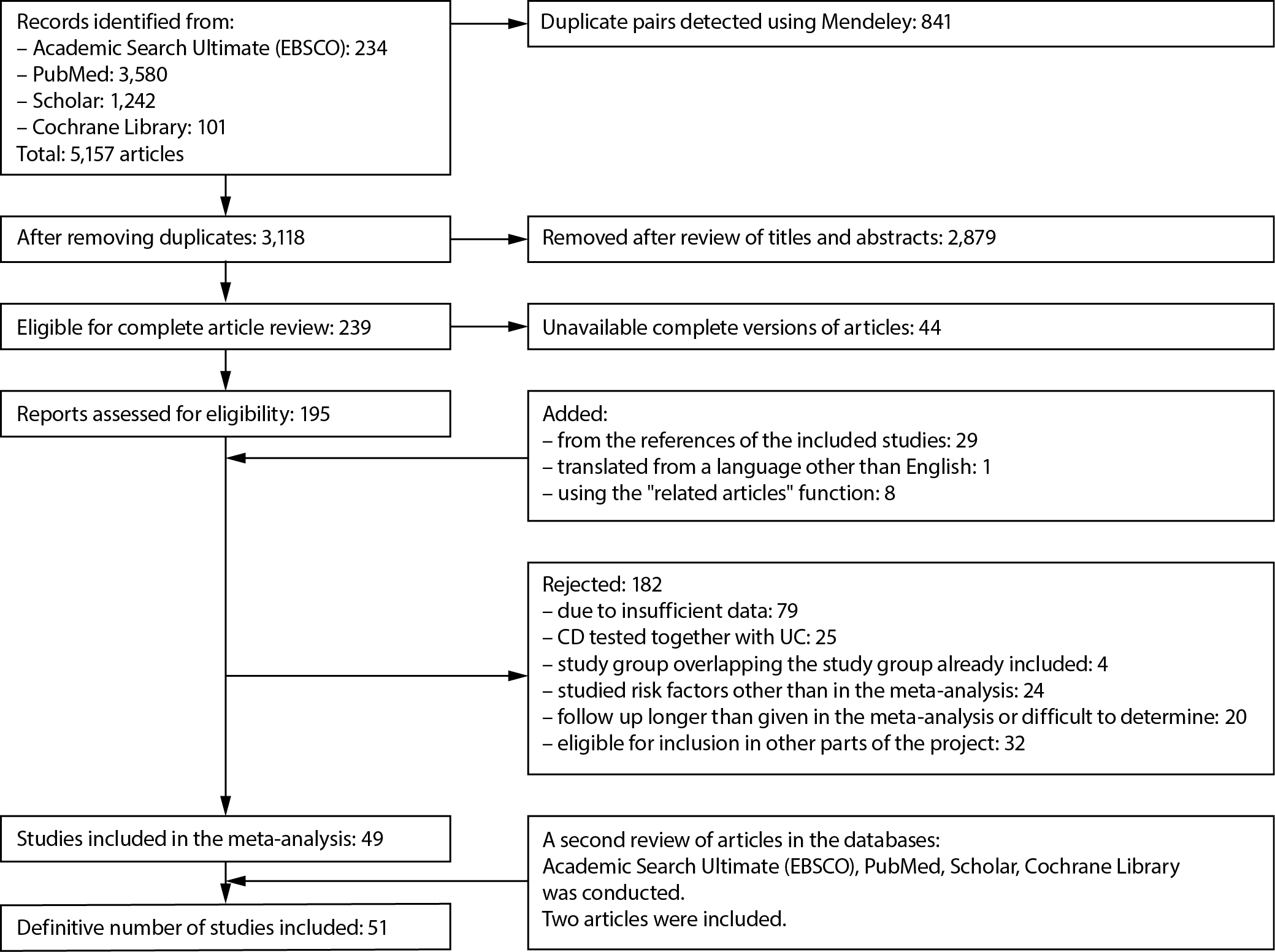
A comprehensive literature search identified a total of 5,157 publications. Of these, 51 articles met the inclusion criteria and were included in the meta-analysis. A detailed overview of the number of records identified, screened, excluded, and included in the analysis is presented in Figure 1. Detailed characteristics of the included studies, together with their quality assessments using the NOS, are presented in Table 4.11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61 No randomized controlled trials were identified; therefore, the meta-analysis was based on cohort studies comprising data from 21,283 patients. The mean age of participants across studies ranged from 29 to 46 years.
Considerable heterogeneity was observed among the included studies. Notably, studies conducted by Celentano et al.,14 the Italian Society of Colorectal Surgery (SICCR),15 and Yu et al.25 focused on cohorts of patients aged ≥16 years. In contrast, Wang et al.,11 Tiberi et al.,16 and O’Brien et al.17 exclusively analyzed severe postoperative complications classified as >3a according to the Clavien–Dindo classification. Mege and Michelassi31 assessed postoperative complications based on rates of acute hospital readmissions. In another study, Mege et al.42 included only patients undergoing ileocecal resection with anastomosis and a temporary protective stoma.
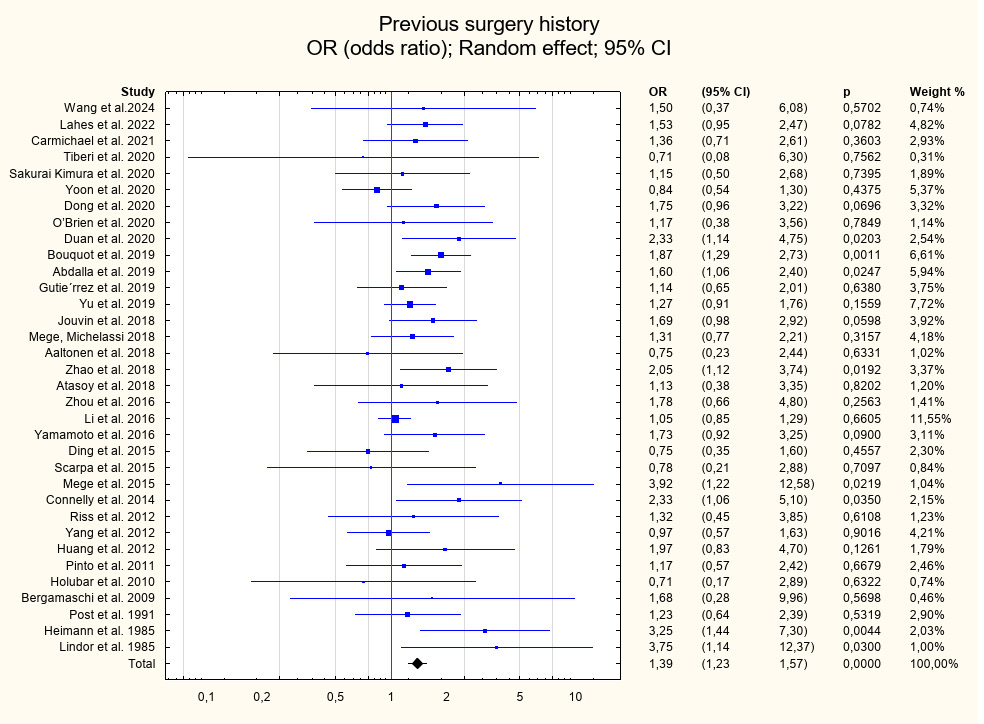
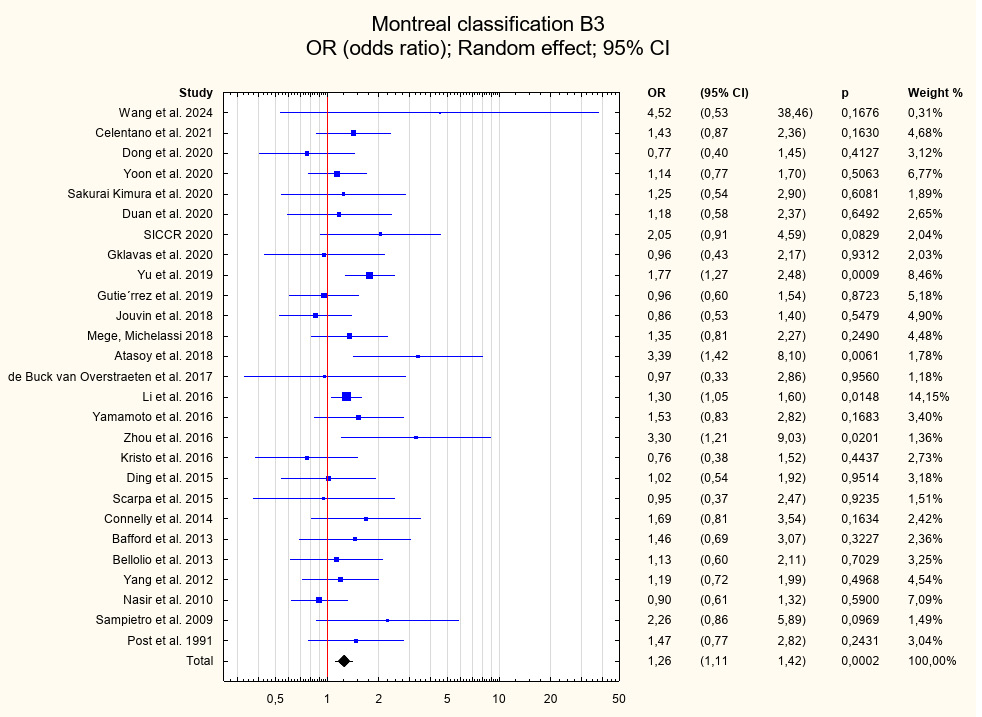
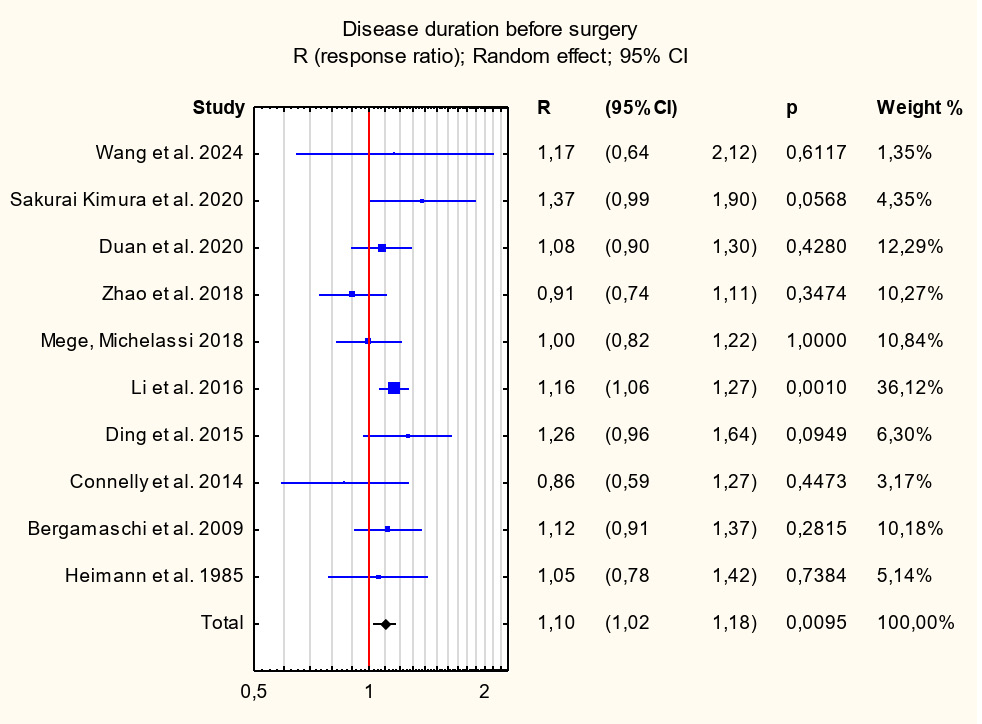
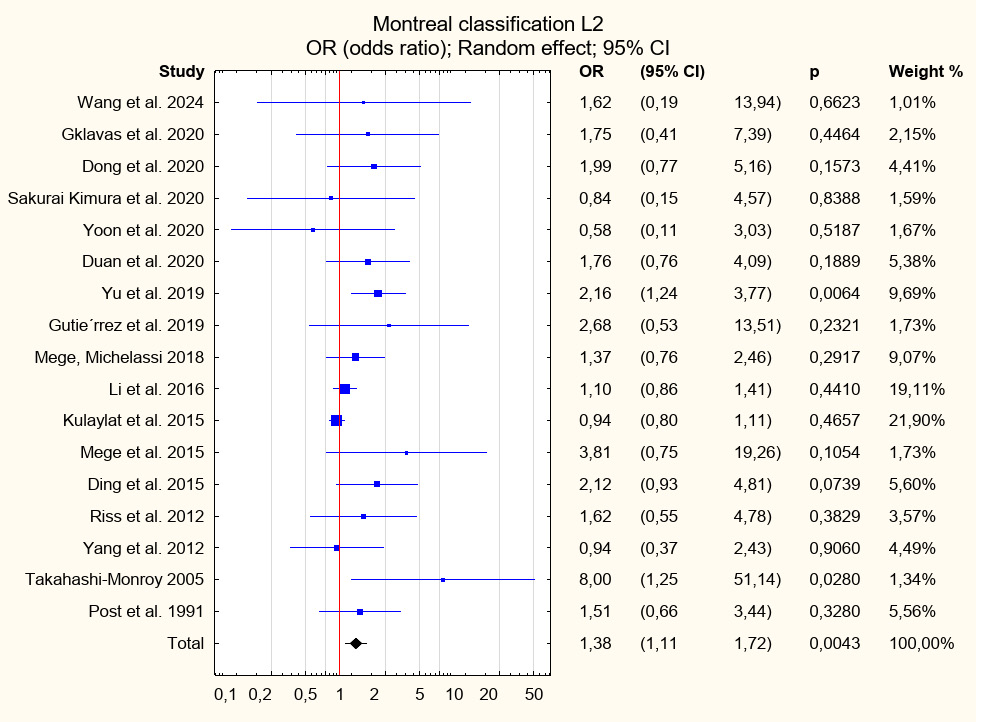
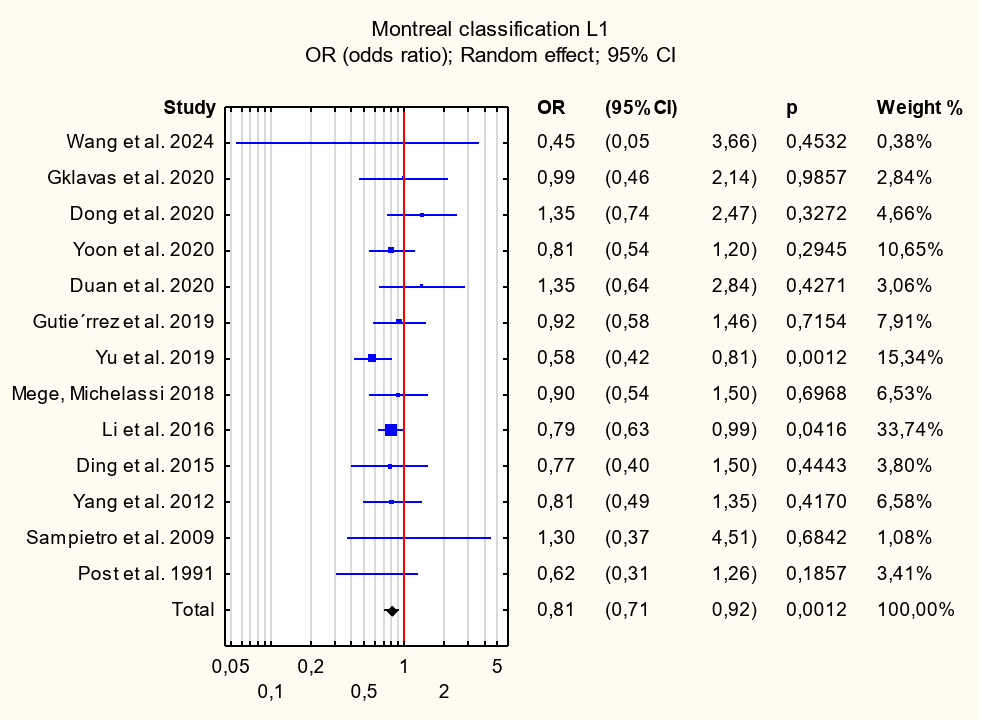
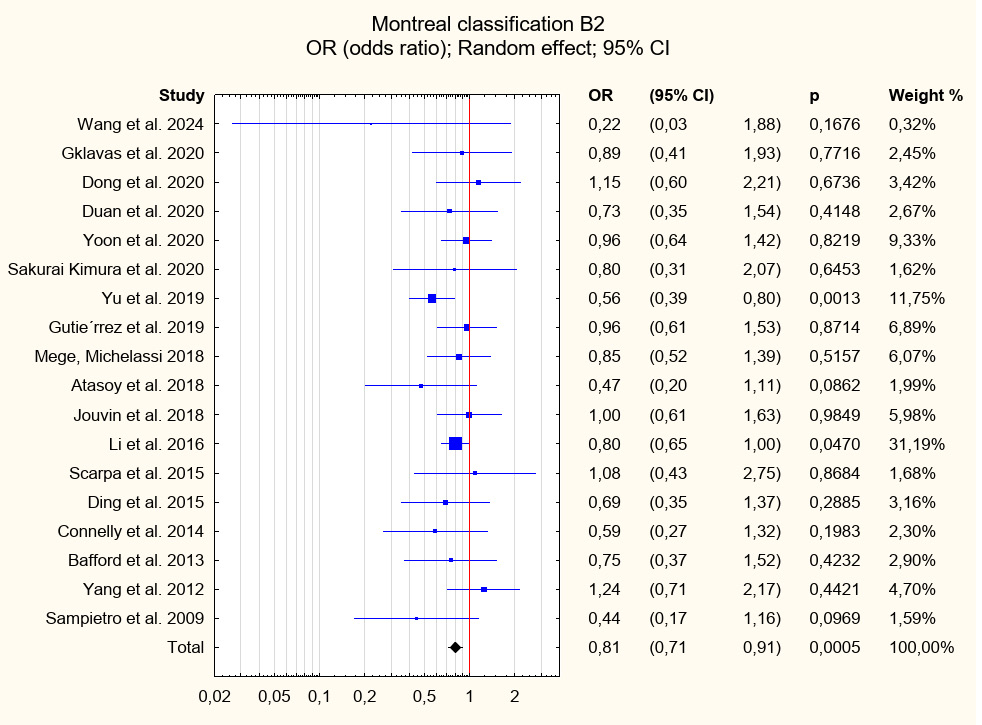
Among the patient-related risk factors associated with early postoperative complications and identified with level I strength of evidence were previous surgical interventions (OR = 1.39; 95% CI: 1.23–1.57; p < 0.001), Montreal classification group B3 (OR = 1.26; 95% CI: 1.11–1.42; p < 0.001), and disease duration prior to surgery (R = 1.10; 95% CI: 1.02–1.18; p = 0.01). A factor identified with level II strength of evidence that showed statistical significance was Montreal classification group L2 (OR = 1.38; 95% CI: 1.11–1.72; p = 0.004). Conversely, factors associated with a decreased risk of postoperative complications and identified with level I strength of evidence included Montreal classification group L1 (OR = 0.81; 95% CI: 0.71–0.92; p = 0.001) and group B2 (OR = 0.81; 95% CI: 0.71–0.91; p = 0.001). For other risk factors, including intraoperative intra-abdominal abscess, concomitant perianal disease, and Montreal classification groups A1–A3, L3, L4, and B1, no statistically significant associations were observed. Further details of the analyses are presented in Table 5, Figure 2, Figure 3, Figure 4, Figure 5, Figure 6, Figure 7, and Supplementary Fig. 1–8.
Following the initial analysis, an additional analysis of risk factors associated with severe postoperative complications (defined as Clavien–Dindo grade ≥IIIa) was performed. However, the available data were sufficient to analyze only prior surgical interventions, which did not show a statistically significant association (OR = 1.55; 95% CI: 0.82–2.96; p = 0.181).
Discussion
Crohn’s disease is an inflammatory disorder of the gastrointestinal tract that may involve any segment of the digestive system and is often accompanied by extraintestinal manifestations. The etiology of CD is complex and not fully understood, with multiple factors contributing to its development. These include genetic predisposition, such as variants in the NOD2/CARD15 genes,62 autoimmune responses to intestinal bacterial antigens,63 environmental factors including smoking, dietary habits, and stress,64, 65, 66 as well as dysbiosis of the intestinal microbiota.67 A key characteristic of CD is its chronic course, which may lead to various complications related both to the disease itself and to its management, particularly surgical treatment.68
This meta-analysis aimed to identify disease-related risk factors associated with the occurrence of complications after surgery in patients with CD. Disease duration prior to surgical intervention emerged as a statistically significant predictor of early postoperative complications (class I evidence; R = 1.10; 95% CI: 1.02–1.18; p = 0.01).
Previous studies have also demonstrated the association between longer disease duration and postoperative complications. For example, Hossne et al.69 reported that disease duration was a significant risk factor for postoperative complications, a finding similarly observed by Sakurai Kimura et al.22 The available literature further supports the notion that prolonged disease duration is associated with an increased risk of surgical complications.70 This relationship may be explained by the fact that patients with a longer disease course are particularly susceptible to complications resulting from chronic inflammation, malabsorption, and subsequent nutritional deficiencies, all of which may impair the healing of surgical wounds and intestinal anastomoses.71 Prolonged inflammation may also contribute to the development of complications requiring additional surgical interventions, such as fistulas or intestinal strictures.23, 68, 69, 71 Moreover, repeat surgical procedures are generally associated with a higher risk of complications due to the compromised quality of abdominal tissues, which makes safe surgical dissection and preparation more challenging.72, 73
Additionally, the presence of Montreal classification group B3 (penetrating disease with fistula formation) was identified as a statistically significant risk factor for postoperative complications (level I evidence; OR = 1.26; 95% CI: 1.11–1.42; p < 0.001). Montreal classification group L2 was also identified as a significant risk factor for postoperative complications in this cohort (OR = 1.38; 95% CI: 1.11–1.72; p = 0.004). However, there is no clear consensus regarding the Montreal classification as a prognostic indicator of disease progression. A study by Tajra et al.74 found no association between disease progression as defined by the Montreal classification and the risk of postoperative complications. Conversely, Lin et al.75 suggested that categorizing patients based on colonic involvement may provide a more reliable predictor of clinical outcomes than the Montreal classification. This issue warrants further investigation, particularly in light of reports suggesting that the Montreal classification may be associated with a statistically significant increase in the need for emergency surgical interventions.74
In our study, Montreal classification groups L1 (OR = 0.81; 95% CI: 0.71–0.92; p = 0.001) and B2 (OR = 0.81; 95% CI: 0.71–0.91; p = 0.001) were associated with a reduced risk of postoperative complications. Historically, it has been suggested that patients presenting with stricturing disease experience a milder clinical course both before and after surgery compared with those with penetrating disease manifestations such as fistulas and abscesses, although the supporting evidence remains limited. The Montreal classification for CD reflects this assumption by assigning the most severe disease behavior category to fistulizing (penetrating) disease regardless of the presence of strictures. Patients are classified as having stricturing disease only when strictures occur in isolation. Consequently, the Montreal classification may underestimate the true prevalence of strictures. One proposed mechanism suggests that fistulas and abscesses develop at sites where transmural bowel inflammation coincides with increased intraluminal pressure in intestinal segments proximal to a stricture.76
Another variable identified in this analysis as a statistically significant risk factor for postoperative complications was a history of prior surgical interventions (level I evidence; OR = 1.39; 95% CI: 1.23–1.57; p < 0.001). Patients with a long disease duration and a history of previous surgical procedures represent a distinct subgroup that poses particular challenges in surgical management.70 This observation is supported by the findings of Kotze et al.,70 who reported that a longer interval between the diagnosis of CD and surgical intervention is significantly associated with an increased risk of postoperative complications. Each subsequent surgical procedure may lead to the formation of intra-abdominal adhesions,77 which in patients with CD may be particularly extensive and technically challenging during surgery.70, 78 Clinicians involved in the surgical management of inflammatory bowel disease are well aware that extensive adhesions substantially increase the risk of intraoperative full-thickness intestinal injury.77, 78 This represents a significant challenge, as the surgical management of CD should aim to avoid unnecessary intestinal resections whenever possible.70, 78
The analysis of risk factors such as intraoperative intra-abdominal abscess, perianal involvement, and selected Montreal classification categories (A1–A3, L3, L4, and B1) did not demonstrate statistically significant associations with postoperative complications. In contrast, Bechara et al.79 reported that factors including ileocecal disease location, the presence of fistulas, and perianal disease involvement (L3B3p) were associated with an increased risk of surgical recurrence of CD.79
A possible explanation for the lack of statistical significance observed for the aforementioned Montreal classification categories may be the considerable variability in disease progression associated with these phenotypes. In a study by Tajra et al.,74 the presence of strictures in the jejunum and ileum, perianal lesions in patients with CD, and age at diagnosis were reported to be significantly associated with an increased likelihood of surgical intervention. However, the authors did not evaluate the risk of postoperative complications.
According to Martinez-Sanchez et al.,80 CD with perianal involvement is associated with a poorer prognosis, particularly in cases with rectal involvement, and is linked to a higher frequency of biological therapy use and hospital admissions, especially among patients with complex perianal disease. However, the study did not demonstrate a corresponding increase in the number of abdominal surgeries in this patient group. Furthermore, the prevalence of biological therapy among patients with perianal disease was higher than among those without perianal involvement (42.8% vs 30.7%). The discrepancy between the findings of the present meta-analysis and those reported in the cited study highlights the need for further investigations involving larger patient cohorts to provide sufficient statistical power for a more comprehensive analysis.
Limitations of the study
Several important limitations of this study should be acknowledged. The primary methodological limitation was that the literature review was conducted by a single researcher, a constraint related to the doctoral nature of the project. To minimize potential errors, the process was supervised by the principal investigator, and all included studies underwent thorough verification. Nevertheless, the inherent risk of subjective bias associated with single-reviewer study selection cannot be excluded. Another methodological limitation was the re-examination and inclusion of additional studies 3.5 years after the initial literature search. Although this approach allowed the meta-analysis database to be updated, it may have introduced additional sources of bias. Furthermore, the aggregated data reflect treatment outcomes from diverse clinical centers, where unmeasured institutional factors may substantially influence therapeutic results. Differences in the interpretation of study endpoints across centers may further affect the reliability of the presented findings.
Conclusions
In summary, the findings of the present meta-analysis indicate that prolonged disease duration, penetrating disease behavior with fistula formation (Montreal classification group B3), and a history of prior surgical interventions are significant risk factors associated with an increased likelihood of postoperative complications in patients with CD. Further investigation of factors influencing surgical complications remains warranted. Future studies, particularly prospective investigations as well as those examining immunological, microbial, and genetic determinants, may help refine the identification of predictors of postoperative outcomes in this patient population. Addressing these challenges may play an important role in optimizing surgical management and reducing the risk of postoperative complications. In particular, a better understanding of the effects of chronic inflammation and the impact of gastroenterological therapies on postoperative healing – especially with regard to intestinal anastomoses – may facilitate the identification of more precise predictors of postoperative recovery. Ultimately, this may support the development of clinically applicable predictive models, enabling more personalized treatment strategies and improving patient outcomes and quality of life.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.18467011. The package contains the following files:
Supplementary Fig. 1. Meta-analysis of early postoperative complications for intraoperative abscess.
Supplementary Fig. 2. Meta-analysis of early postoperative complications for concomitant perianal disease.
Supplementary Fig. 3. Meta-analysis of early postoperative complications for Montreal classification group A1.
Supplementary Fig. 4. Meta-analysis of early postoperative complications for Montreal classification group A2.
Supplementary Fig. 5. Meta-analysis of early postoperative complications for Montreal classification group A3.
Supplementary Fig. 6. Meta-analysis of early postoperative complications for Montreal classification group L3.
Supplementary Fig. 7. Meta-analysis of early postoperative complications for Montreal classification group L4.
Supplementary Fig. 8. Meta-analysis of early postoperative complications for Montreal classification group B1.
Use of AI and AI-assisted technologies
Not applicable.