Abstract
Oxaliplatin (OXI) is a platinum-based chemotherapeutic agent commonly used in the systemic treatment of colorectal, gastric, pancreatic, and esophageal cancers. Like other cytotoxic agents, OXI is associated with various adverse effects; however, OXI-induced peripheral neuropathy (OIPN) represents a particularly challenging clinical problem for both oncologic patients and their clinicians. The severity of neuropathic symptoms may necessitate dose reduction or premature discontinuation of therapy, which can ultimately shorten recurrence-free survival. This paper discusses the symptoms, mechanisms of onset, risk factors, preventive strategies, treatment options, and grading scales associated with OIPN. Both the prevention and treatment of OIPN require further innovative and effective research. It is hoped that the outcomes of ongoing trials will address both chronic and acute OIPN in the near future, as this condition remains a persistent clinical concern for patients with gastrointestinal cancers.
Key words: oxaliplatin, peripheral nervous system diseases, neurophysiology, oxaliplatin-induced peripheral neuropathy, high-resolution ultrasonography
Introduction
Oxaliplatin (OXI)-induced neuropathy is a common and challenging adverse effect of this platinum-based anticancer agent. This neuropathy can be classified into acute neuropathy (affecting 85–96% of treated patients) and chronic peripheral neuropathy (affecting 40–93% of treated patients), with sensory symptoms such as tingling and numbness of the limbs being more prevalent than motor symptoms.1 Cold-induced peripheral paresthesia, cold-induced pharyngolaryngeal dysesthesia, muscle cramps, dysphagia, jaw stiffness, laryngospasm, and voice and ocular changes (such as blurred vision, visual loss, color perception abnormalities, eye pain, ptosis, and excessive lacrimation) are typical features of acute OXI-induced neuropathy. According to Grothey, 15% of patients receiving a cumulative OXI dose of 780–850 mg/m2 develop sensory ataxia as a chronic manifestation of neuropathy.2 Nine of the 14 analyzed studies reported acute neuropathic symptoms in 4–98% of patients during the 1st treatment cycle (within 14 days of initiating OXI therapy). Symmetrical paresthesia of the limbs, mouth, pharynx, and larynx occurs or worsens in response to low-temperature exposure in acute neuropathy and typically resolves within a few days.3, 4 Acute OXI-induced neuropathy leads to dose reduction in 15–43% of patients, prolongation of infusion times in 12–22%, and premature treatment discontinuation in 6–21% of cases. These adjustments may, to some extent, mitigate the negative effects of neuropathy.5, 6, 7, 8, 9, 10
Persistent numbness, paresthesia, allodynia, and burning pain in the hands and feet are reported in approx. 40% of patients with chronic peripheral neuropathy.11 Chronic symptoms, particularly pain, lead to difficulties with tasks such as writing, buttoning, and walking. These impairments significantly affect quality of life, sleep, and daily functioning. Moreover, they represent a primary reason for treatment discontinuation, which consequently shortens recurrence-free survival.3, 12, 13, 14 As mentioned above, oncologists must learn to distinguish painful from non-painful peripheral neuropathy, as the painful form significantly worsens quality of life.15 Results from the population-based PROFILES registry in the Netherlands indicate that a more advanced stage of colorectal cancer, osteoarthritis, and higher levels of anxiety symptoms are associated with an increased risk of non-painful peripheral neuropathy. In contrast, a longer time since oncological diagnosis was positively correlated with a decreased risk of painful peripheral neuropathy. Additionally, painful peripheral neuropathy had a detrimental effect on quality of life and social functioning.15
It is noteworthy that OXI-induced neuropathy can worsen even several months after treatment has been discontinued. This phenomenon is referred to as “coasting.” Approximately 30% of patients experienced symptoms of peripheral neuropathy 6 months or more after chemotherapy cessation, while about 2/3 of patients reported symptoms 1 month following the completion of chemotherapy.16 According to an analysis of 27 trials, chemotherapy-induced peripheral neuropathy (CIPN) diminished by 26% per year after the completion of systemic treatment.17
There are various definitions of acute and chronic neuropathy. One definition suggests that chronic neuropathy occurs 12 ±2 months after the completion of OXI administration.18, 19 Baek et al. defined chronic neuropathy as grade 2 neuropathy lasting seven days or longer, or grade 3 neuropathy of any duration.20 In contrast, Alejandro et al. categorized this adverse effect into acute neuropathy, lasting a maximum of 13 days, and chronic neuropathy, which persists for 14 days or longer.21
Objectives
This systematic review aimed to analyze the issues surrounding oxaliplatin-induced peripheral neuropathy (OIPN) in cancer patients. The mechanisms of onset, risk factors, clinical signs and symptoms, as well as treatment and prevention strategies, were examined.
Material and methods
The research was conducted using the PubMed database. The review included preclinical studies, clinical trials, and meta-analyses published between 1997 and 2024, with particular emphasis on studies from the past decade. Studies not published in English were excluded.
Pathomechanism of oxaliplatin-induced peripheral neuropathy
Chemotherapy-induced peripheral neuropathy is a common adverse effect of chemotherapy, with a variable, time-dependent incidence.22, 23 According to available data, approx. 50% of patients with peripheral neuropathy reported resolution of symptoms 6 months after completion of therapy.16 Treatment with OXI may cause myelotoxicity, fever, liver function abnormalities, as well as central and peripheral neurotoxicity, including OIPN.
Central neuropathy involves brain tissue influencing peripheral nerves; e.g., damage to the central nociceptive system can induce mechanical hyperalgesia, which patients report as pain during the course of acute OIPN. Platinum analogues are standard treatments for various gastrointestinal cancers, including advanced colorectal, esophageal, gastric, liver, and pancreatic tumors, with neuropathic symptoms persisting for up to 6 months after completion of the treatment cycle.23 The antitumor activity and toxicity of OXI are associated with ultrafiltrate platinum which is irreversibly bound to plasma proteins and red blood cells, transforming into a non-active pharmacological structure. Interestingly, the peak concentration of OXI is observed 2 h after administration.24, 25 An exceptional feature of OXI is its fast, nonenzymatic transformation into reactive platinum complexes and oxalate-free cytotoxic analogue. Both of these products induce peripheral sensory neuropathy, but it is the oxalate that primarily has an impact on the early and later decrease in mechanical threshold as well as acute cold-induced neuropathy, the most characteristic OIPN symptom.23 Total platinum content is taken out from the blood vessels by binding to plasma and/or blood constant components, tissue uptake, and urine elimination, which can explain the mechanisms underlying acute OIPN.24
Given the pharmacokinetics of OXI, OIPN manifests as 2 distinct temporal types of neuropathies: 1) the characteristic acute peripheral sensorimotor polyneuropathy, which typically occurs during or within hours after drug infusion and is usually reversible; and 2) sensory neuropathy, which is associated with cumulative drug dose.24
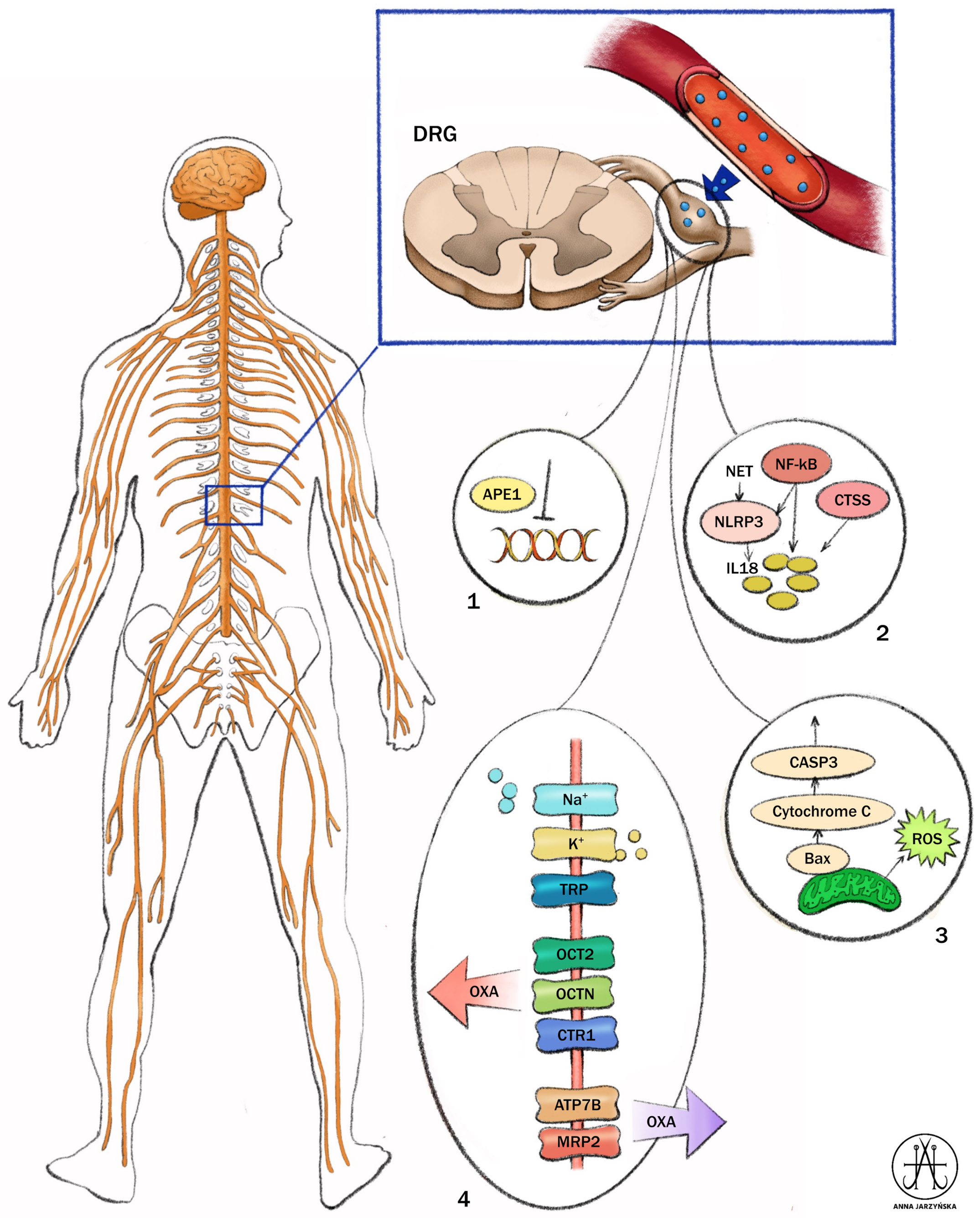
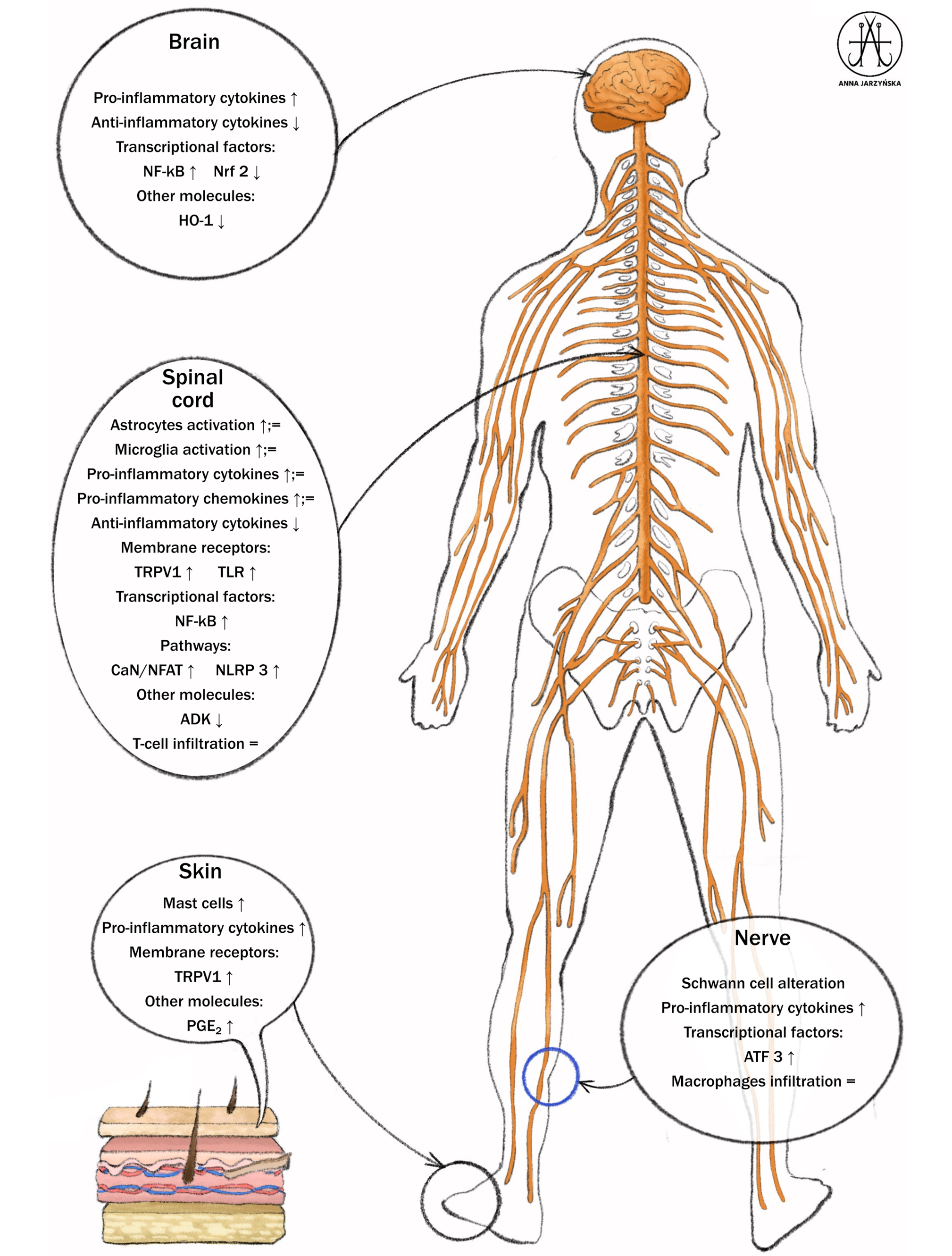
Oxaliplatin-induced peripheral neuropathy is characterized by complex, multifocal pathomechanisms involving several structures of the central and peripheral nervous systems (Figure 1, Figure 2).25, 26, 27 These mechanisms are associated with the aggregation of platinum products in dorsal root ganglion (DRG) and trigeminal ganglion (TG) neurons, with consequential functional impairment of the myelin sheath, neuronal cell body, and glial structures. The exact mechanism of OIPN remains ambiguous; however, it is believed that some aspects of ion channels abnormalities, disruption of normal axonal membrane potential, and typical antitumor mechanisms contribute to the toxic effect on neurons and glial cells.23, 28 The mechanisms of chemotherapy involve binding of the drug to tumor cell nuclear DNA followed by inhibition of DNA replication and RNA transcription, along with inhibition of mitochondrial DNA replication and transcription. This leads to altered mitochondrial function, disruption of the respiratory chain, and overproduction of reactive oxygen species (ROS). Additionally, chemotherapy activates the immune system, resulting in the production of pro-inflammatory cytokines and protein kinases. Ultimately, all of these processes contribute to apoptosis.
In acute OIPN, the pathomechanisms are connected with a dysfunction of voltage-gated ion channels (e.g., sodium, potassium, and calcium channels), transient receptor potential channels, organic cation transporters 1 and 2, the Mate-1 transporter, and glial cells. The chronic form of OIPN is primarily characterized by DNA and mitochondrial damage, ROS production, and inflammation, which are associated with immune activation of nervous system cells.29 The exact pathomechanisms of OIPN remain unclear and it is believed that several inducers with different mechanisms of action may affect the same targets.30
The onset of OIPN, occurring independently of chemotherapy agent mechanisms, is associated with microtubule disruption in neurons.23 The DRG (regions lacking the blood–brain barrier (BBB)), sensory neurons, satellite cells, Schwann cells, as well as neuronal and glial cells in the spinal cord are the most sensitive sites where the neurotoxicity-induced chemotherapy begins.28, 31 Alterations in channel activity leading to ectopic nociceptors activation, myelin sheath damage, and electrophysiological abnormalities are also observed as a result of OIPN.28
Studies conducted during OXI treatment have revealed the development of an acute sodium (Na+) channelopathy in sensory and motor axons. Slowing of sodium channel inactivation induces reversible cold allodynia. Motor nerve hyperexcitability may serve as a marker for the potential of more severe neurotoxicity.32, 33 Several studies have shown that although sensory symptoms predominate in the patients studied, the activity of voltage-gated sodium channels in motor axons are closely related to cold allodynia and exhibit a reversible nature.33, 34, 35
Membrane hyperexcitability may also be connected with chelation of intracellular calcium during activation of voltage-gated sodium channels.5 Gene ablation of Nav1.6 prevented the action potential burst upon electrical stimulation after OXI treatment at low temperatures, demonstrating that the induction of cold allodynia by OXI is linked to Nav1.6.36 Observations also suggest a role in the regulation of MAP kinase, which is transcriptionally regulated by the MAPK-ERK-CREB signaling pathway encoded by Scn8A.37 Transient axonal hyperexcitability, resulting from an imbalance in voltage-gated Na+ channels, leads to sustained depolarization and activation of the Na+/Ca2+ exchanger 2 (NCX2) reverse sodium-calcium channel, with subsequent increase in calcium cations concentration and axonal damage. In both in vitro and in vivo studies, the inhibition of NCX was observed to reduce axonal damage.31
Abnormalities in DRG
Most well-known mechanisms include processes in the DRG which are influenced by the specific structure of the first sensory pathway cell. Dorsal root ganglion is not protected by the BBB, making it more susceptible to the accumulation of OXI and other platinum derivatives. Abnormalities in DRG are associated with several dysfunctions including those related to the organic cation transporter 2 (OCT2), as well as potassium, chloride, and calcium voltage-gate channels which are also involved in pain-induced pathways.
Due to a local expression of OCT2, DRG is predisposed to the absorption of OXI especially in the early phase of treatment. In experimental models, the absence of OCT2 diminishes hypersensitivity to cold or allodynia during the acute phase of OXI treatment.38 Additionally, several transport enzymes including organic cation transporters, high affinity copper uptake protein 1 (CTR1), copper transporter ATPase alpha (ATP7A), and multidrug and toxin extrusion protein 1 (MATE1), can be responsible for OIPN.25
Research has shown that DRGs from OXI-treated mice exhibited a significant downregulation of mRNA expression of potassium voltage-gated channel Kv1.1, as well as K2P-TREK1 and TRAAK, which are thermosensitive and mechanosensitive 2-pore-domain potassium channels. Additionally, OXI-induced mechanical and cold allodynia was observed following co-ablation of TREK1 and TRAAK in mice.36
Elevated expression of the calcium channel subunit α2δ1 has also been detected in rat DRG neurons and spinal cord following paclitaxel or OXI treatment. Calcium channels are known to be crucial mediators of peripheral sensitization and are also the target of gabapentin, a recognized treatment for neuropathic pain.36, 39
The upregulation of gene and protein expression of voltage-gated Nav1.6 in rat DRGs is responsible for the generation of action potential peripheral nerve fibers, as well as repetitive action potential discharges in myelinated axons.36, 40
Although there are different expressions of NKCC1 and KCC2 in the cell bodies of DRG neurons and dorsal horn of the spinal cord, evidence supports the notion that the dysregulation of chloride balance activates disinhibition not only in DRG but also in the spinal cord. This, in turn, increases neuronal excitability, leading to hyperalgesia and allodynia. A new question has arisen regarding the role of other chloride channels in the pathogenesis of OIPN.36
The OXI treatment also induces an increase in intracellular hydrogen ion concentrations in the DRG during the acute phase by affecting the activity of the plasma membrane protein isoform 1 NHE1. This protein is responsible for Na+/H+ transport and is located on nociceptors, where it plays an important role in maintaining intracellular pH homeostasis.41 In preclinical rat models, the inhibition of FABP5, localized in DRG, attenuates pain in OIPN. FABP3, FABP5, and FABP7 are intracellular chaperones of endocannabinoids (eCB), which are expressed throughout the nervous system and activated in various pain models.29
Research using the cannabinoid receptor agonist CB2- β-caryophyllene (BCP) in animal models has shown that OXI therapy results in dysregulation of neuroinflammatory pathways, including an increased content of interleukin (IL)-1β, IL-6, and tumor necrosis factor alpha (TNF-α), along with reduced levels of IL-10 protein in the spinal cord. The use of BCP reduces the frequency and severity of nociceptive effects in these models. Additionally, studies on its beneficial effects have demonstrated that BCP restores TNF and IL-10 concentrations, as well as decreases ROS products and lipid peroxidation in the spinal cord.42
Wallerian degeneration of intraepidermal nerve fibers (IENFs), including unmyelinated C-fibers and, to a lesser extent, thinly myelinated axonal A-fibers responsible for transmitting touch, pain and thermal sensations, is also implicated in the mechanism of OIPN. Observations have shown ectopic firing – abnormal electrical activity in the distal ends of the IENFs. This leads to a phenomenon known as central sensitization, which refers to enhanced activation of pain pathways in the central nervous system, particularly in the spinal cord and brain. Central sensitization reinforces and perpetuates the activation of pain pathways thereby contributing to the persistence of chronic pain.36
Glial cells, including satellite and Schwann cells, might take part in the neuroinflammatory process in DRG and axons. Neuroinflammatory components have also been shown in the spinal cord, where microglia and astrocytes play a prominent role in OIPN development.43 Neuroinflammation, as indicated by the activation of glial cells and proinflammatory cytokines (e.g., NF-B), is detected not only at the peripheral nerve system structure but also in the spinal cord, contributing to the central sensitization. Oxaliplatin may also affect the activity of mast cells through the activation of proteinase-activated receptor 2 (PAR2), leading to intraepidermal nerve fibers degeneration and resulting in mechanical allodynia.36
The chemotherapy-related central effect of OXI administration have been explained by Branca et al.43 The administration of OXI induces modifications of the tight junction (TJs) proteins, including zonula occludens-1 (ZO-1) and of F-actin, which activate BBB disruption in the central nervous system. Additionally, intracellular OXI activates ROS and induces endoplasmic reticulum stress, as evidenced by increased expression levels of glucose-regulated protein 78 (GRP78). The induction of caspase-3 leads to extracellular ATP release.43, 44
Grading peripheral sensory neuropathy
It is well-established that the presence of peripheral neuropathy leads to a deterioration in a patient’s quality of life, which, in turn, negatively affects social functioning and work performance. Four grading systems, based on the patient’s self-reporting of symptoms, have been developed in order to standardize the assessment of peripheral sensory neuropathy. There is no significant difference between the Eastern Clinical Oncology Group (ECOG) and the National Cancer Information Center Common Toxicity Criteria (NCIC-CTC) scales. The grading for these systems is as follows:
− Grade 0: ‘none or no change’;
− Grade 1: ‘loss of deep tendon reflexes, mild paresthesias’;
− Grade 2: ‘mild or moderate objective sensory loss; moderate paresthesias’;
− Grade 3: ’mild or moderate objective sensory loss; moderate paresthesias’45;
According to World Health Organization (WHO):
− Grade 0 indicates ‘no symptoms’;
− Grade 1 denotes ‘presence of paresthesias and/or decreased deep tendon reflexes’;
− Grade 2 is characterized by ’severe paresthesias and/or light muscle weakness’;
− Grade 3 refers to ‘intolerable paresthesias and/or marked weakness’; and
− Grade 4 denotes ‘paralysis’.46
The National Cancer Institute (NCI) Common Terminology has introduced grade 5 which signifies ’death’ and has modified the definition of grade 4 to indicate ’life-threatening consequences: urgent intervention indicated.’47
Another valuable tool for assessing the grade of neuropathy is the questionnaires that evaluate the functional consequences of CIPN in cancer patients. One such tool is the Functional Assessment of Cancer Therapy/Gynecologic Oncology Group Neurotoxicity (FACT/GOG-Ntx-13), where a higher score correlates with a better quality of life. This validated 13-item questionnaire rates items on a 5-point Likert scale, ranging from “not at all” to “very much”, with a total possible score of 52.48
The Chemotherapy-Induced Peripheral Neuropathy Assessment Tool (CIPNAT), European Organization for Research & Treatment in Cancer Quality of Life Quest-CIPN 20 (EORTC QLQ-CIPN 20), Modified Total Neuropathy Score (mTNS), and Total Neuropathy Score clinical version (TNSc) and Total Neuropathy Score-reduced (TNSr) are all valuable patient-reported outcome measures used to evaluate the impact of neuropathy on patients’ lives.49 Oncologists using these scales should be aware that factors such as subjective misjudgment, poor anti-interference, and low sensitivity represent limitations of these questionnaires. A reduced version of the European Organization for Research and Treatment of Cancer (EORTC) Quality of Life Questionnaire Chemotherapy-Induced Peripheral Neuropathy scale (QLQ-CIPN20), called CIPIN15, has been shown to be a reliable and significant self-reporting tool for assessing CIPN.50
In a population of Chinese patients, a 20-item CIPN assessment tool, known as the Chemotherapy-Induced Peripheral Neuropathy Integrated Assessment–Oxaliplatin Subscale (CIPNIA-OS), was evaluated. The results were encouraging as the CIPNIA-OS questionnaire demonstrated higher sensitivity than QLQ-CIPN20 in the evaluation of OIPN. However, the small sample size remains the major limitation of this study.51
In an Australian study, patients receiving OXI completed a questionnaire containing standardized CIPN patient-reported outcome assessments (EORTC CIPN-20, PRO-CTCAE) using a tablet device. Patient-reported outcome assessment scores were correlated with the NCI-CTCAE grade and analyzed with consideration of cumulative dose of OXI. It is surprising that CIPN patient-reported outcomes provide additional information beyond the nurse-assessed NCI-CTCAE grade, particularly during the earlier stages of chemotherapy.52
Risk factors
A meta-analysis including nearly 11,000 participants from 20 studies identified factors associated with OIPN. Older age (above 60 years), female sex, a higher body mass index (BMI) (>25 km/m2), a larger body surface area (BSA) (≥2.0 m2), and those with low physical activity levels were associated with an increased risk of OIPN 53, 54, 55, 56, 57, 58
An analysis of 1,401 patients from 23 studies found that in patients aged 65 years and older at the time of cancer diagnosis age was an independent predictor of the development of CIPN. This finding was confirmed in a subsequent study of more than 3,500 patients aged ≥70 years.59, 60, 61, 62 In contrast, some investigators have reported that age is not associated with OIPN.8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 63, 64 One study recorded that female sex was associated with the incidence and severity of OIPN.65 Contrastingly, however, many published studies (including studies with multivariate analysis) reported no difference in the peripheral neuropathy rates between the sexes.18, 20, 64, 65, 66, 67
The latest study by Lee et al. noted that reduced physical activity increased the severity of peripheral neuropathy, as assessed using the CTCAE and the Functional Assessment of Cancer Therapy/Gynecologic Oncology Group-Neurotoxicity-13 (FACT/GOG-NTX-13). The FACT/GOG-NTX-13 subscale evaluates the degree and impact of sensory and motor neurotoxicity symptoms in the preceding 7 days.59
Comorbidities such as diabetes mellitus, alcoholism, anemia, and hypomagnesemia are also significantly associated with a higher risk of this kind of neuropathy.53, 58, 60, 61 Furthermore, lower hemoglobin before starting therapy with OXI correlated with more severe symptoms of persistent peripheral neuropathy after chemotherapy.56, 61 Additionally, peripheral neuropathy occurred twice as often in patients with autoimmune diseases.60
On the other hand, some reports did not show correlation between well-known risk factors such as sex, BMI, diabetes mellitus, alcohol consumption, and preexisting peripheral neuropathy.20, 56, 61, 66, 68, 69
The probability of OIPN increases after 6 cycles of OXI based chemotherapy.53 High BSA (>2.0 m2) and acute neuropathy after 3 cycles of OXI were identified as predictive factors for persistent neuropathy.20
There are discrepancies regarding the cumulative dose of OXI and peripheral neuropathy. In a meta-analysis by Wang et al., a cumulative OXI dose above 500 mg/m2 was associated with OIPN.53 In 2 studies, median cumulative doses of OXI were 810 mg/m2 and 766 mg/m2, respectively, and these doses were correlated with acute neuropathy. Moreover, persistent symptoms of neuropathy were described in 24% and 16% of patients, respectively.70, 71 A cumulative dose of OXI of 675 mg/m2 or higher, or treatment beyond the 8th cycle was correlated with grade 3 neuropathy and was an independent factor of persistent peripheral neuropathy.20 In Asians with colorectal cancer, OIPN of grade 2 or higher occurred with a total dose of 680 mg or more while grade 3 or higher neuropathy was observed with a total dose of 1,040 mg or more.72 According to Storey et al., a higher median cumulative OXI dose of 834 mg/m2 (range: 447–1040 mg/m2) compared to 702 mg/m2 (range: 130–1040 mg/m2) was associated with a statistically higher probability of chronic peripheral neuropathy, observed 12 months after completion of chemotherapy.10 In turn, a median cumulative dose >780 mg/m2 was identified as the main predictor of OIPN.73 Storey et al. confirmed that cumulative OXI dose is the only factor that induces chronic peripheral neuropathy, while other factors – including age, sex, grade ≥2 acute peripheral neuropathy, ‘legs like jelly’, or pseudolaryngospasm during OXI treatment – were not significant.10 On the other hand, Brouwers et al. showed that the dose of OXI is not related to OIPN.74
According to the aforementioned meta-analysis, smoking history and chemotherapy regimen (XELOX vs FOLFOX) did not result in an increased risk of OIPN.53 In contrast to the meta-analysis by Wang et al., the SCOT randomized controlled trial, which compared 3 months to 6 months of OXI-containing adjuvant chemotherapy in colorectal cancer patients, revealed that patients receiving the CAPOX regimen (OXI + capecitabine) had lower OIPN scores than those receiving the FOLFOX (OXI + 5-fluorouracil) regimen. Moreover, both acute and chronic neuropathy occurring after 3 months of chemotherapy were milder or moderate compared to those occurring after 6 months of therapy. The presence of neuropathy prior to the initiation of the OXI-based chemotherapy was identified as the 2nd factor associated with increased severity of peripheral neuropathy.75
Furthermore, based on the study by Tanishima et al., hyperacute peripheral neuropathy was identified as a predictor of persisent OIPN.76
Additionally, some reports have noted that serum levels of albumin, magnesium, vitamin D, and gamma-glutamyl transferase (GGT) play a role in the onset and severity of neuropathy symptoms.56, 61, 66
The histopathologic type, such as mucinous carcinoma of colorectal cancer, was found to be significantly correlated with grade 2 to grade 3 OIPN; however, this result requires confirmation in large studies.61
Housley et al. presented evidence that chronic sensory neuropathy depends on nonlinear interactions between chemotherapy and the cancer itself. The pathomechanism mentioned above involved the DRG, neuronal excitability, and inflammatory processes, and is additionally connected with inappropriate sensory encoding potassium ion channel Kv3.3.77
Environmental factors, such as lower temperatures during winter, did not contribute to an increased risk of acute neuropathy in California.20 In contrast, a study conducted in Denmark found the highest probability of dose modification and completion of OXI occurred during the period from December to February (mean temperature: 0.1–1.8°C).78 A prospective clinical trial showed that vitamin B6 and celecoxib intake did not influence the occurrence or intensification/exacerbation of OIPN.59
Genes such as CSHL1, GH1 (chorionic somatomammotropin hormone-like 1, growth hormone 1), KCMF1 (potassium channel modulatory factor 1, also known as FIGC (bFGF-induced in gastric cancer)), IL36G, EFCAB8 (EF-Hand Calcium Binding Domain 8), CSRP2 (also known as CRP2, codes for cysteine and glycine-rich protein 2), IQGAP1 (or p195, IQ motif containing the GTPase activating protein 1), GNRH2, SMIM1 (small Integral Membrane Protein 1), and C5orf17 are significantly up- or downregulated as a result of chemotherapy and are associated with the onset of OIPN.79 Understanding their expression in peripheral white blood cells could be useful in predicting the occurrence of neurotoxicity.
Another study revealed that single-nucleotide polymorphisms (SNPs) in the FARS2, ACYP2, and TAC1 genes contribute to a higher risk of OIPN. These outcomes should be explored further in cohort studies.80
The animal models have demonstrated a dependency between gut microbiota and the pathological condition of pain. Serum metabolomics correlated well with the impaired lipid metabolism, molecular transport, and augmented inflammatory responses. There are suggestions that changes in gut microbiota (dysbiosis) contributed to the development of OIPN.34
Neurophysiological examination
Oxaliplatin treatment induces 2 primary types of peripheral neuropathy: acute neurotoxicity and chronic neuropathy.20 Acute neurotoxicity is characterized by temporary sensations, such as tingling, typically triggered by cold, as well as muscle spasms, especially in the limbs and jaw. These symptoms typically appear during the infusion and resolve within several days, although they can intensify with repeated treatments. Nearly all patients experience these transient symptoms.5, 13, 19 Chronic neuropathy occurs with cumulative doses and is marked by sensory loss and tingling in a glove-and-stocking distribution, potentially progressing to sensory ataxia.20, 81 Conventional nerve conduction studies (NCS) are insufficient for early detection of nerve dysfunction, as they only reveal abnormalities once significant axonal or myelin damage has occurred. In some patients treated with OXI, features of axonal polyneuropathy were observed: sensory polyneuropathy, manifested by a reduction in the amplitude of the response from sensory fibers (SNAP), or, less frequently, sensorimotor polyneuropathy, where the amplitude of the response was reduced to stimulation of both sensory (SNAP) and motor fibers (CMAP). No conduction velocity slowing or conduction block was observed in these patients. Polyneuropathy was also observed in subsequent studies, 2 years after treatment completion.22 Nerve excitability testing offers additional insights by assessing ion channel function, energy-dependent pumps, and ion exchanges involved in nerve impulse conduction.7 Excitability assessment, in addition to the classic neurophysiological methods presented by Park et al., is a useful and rapid technique for axonal excitability assessment. It is recognized as a biomarker assessing potential predisposition to more severe axonopathy during OXI treatment.6
The precise mechanisms underlying acute OIPN remain unclear; however, studies suggest that abnormal functioning in axonal sodium channels in both motor and sensory axons may contribute to the acute phase of the condition following treatment.3, 6, 82, 83
High resolution ultrasound assessment of oxaliplatin-induced polyneuropathy
The high-resolution ultrasound (HRUS) examination is nowadays a widely accessible tool for assessing peripheral nervous system (PNS) lesions.84 It also complements electrophysiological tests and the combination of these diagnostic methods significantly enhances the sensitivity and specificity of the PNS assessment.84, 85, 86, 87, 88, 89 High-resolution ultrasound employs probes with frequencies greater than >15 MHz allowing for the accurate visualization of various elements of the PNS, including the smallest sensory nerves.
Multiple studies have demonstrated the utility and significance of HRUS in the diagnosis of inflammatory polyneuropathies (PNP).84, 85, 86, 87, 88, 89 The use of HRUS in PNP is primarily based on the observation of swelling and thickening of peripheral nerves associated with demyelinating damage. Reports also suggest potential deviations from the normal ultrasound (US) image of nerves in cases of axonal damage.85, 88 A decreased cross-section area (CSA) of the nerves is thought to occur in certain axonal neuropathies.85, 87, 88
Chemotherapy-induced peripheral neuropathy constitutes a distinct subgroup of polyneuropathies. To date, there have been only single studies on US imaging of the PNS in OIPN, primarily focusing on the effect of OXI on PNS. Briani et al. examined 15 patients before and after the administration of OXI in an attempt to demonstrate US and neurophysiological correlations in peripheral nerve imaging and function.90 Despite the presence of axonal lesions of the tested nerves, no decrease in the CSA of the nerves was observed. However, increased CSA at typical entrapment sites, such as median (wrist) and ulnar (elbow) nerves, was detected in 60% of the patients.90 These findings suggest that OXI may increase the susceptibility of peripheral nerves to compression damage.
Data on ultrasonographic imaging of peripheral nerves in OIPN ar still lacking. However, previous studies on HRUS and PNS evaluation reveal further horizons regarding aspects of structural assessment of nerves in multiple neuropathies. In the case of OIPN, the clinical evaluation should particularly emphasize US assessment of sensory nerves, with specific attention to prior reports of structural damage to PNS in small-fiber neuropathy (SFN).
Prevention and treatment
To date, no pharmacological agents have been recommended for the prevention of CIPN. Numerous preclinical rodent studies have evaluated the efficacy of various agents aimed at inhibiting OIPN, but without significant success.91 It is well established that oxidative stress plays a central role in the development of OIPN. Consequently, antioxidants, including vitamin C, vitamin E, acetyl-L-carnitine, alpha-lipoic acid, and glutathione, have been investigated for their potential protective effects against neuropathy.92, 93, 94, 95 Preclinical studies have confirmed the efficacy of these antioxidants. Additionally, several widely used and clinically approved drugs, including carvedilol, donepezil, dimethyl fumarate, and rosiglitazone, have demonstrated effectiveness in reducing OIPN in preclinical models.96, 97, 98, 99 However, some studies have reported inconsistent results, such as vitamin E failing to attenuate early symptoms of peripheral neuropathy.100, 101
Nonsteroidal anti-inflammatory drugs (NSAIDs), when administered concomitantly with OXI-based chemotherapy, were found to reduce the risk of peripheral neuropathy, although further studies are required to confirm these findings.72
Interleukin-1β, IL-6, and TNF-α are pro-inflammatory cytokines whose levels are elevated in the DRG spinal cord of animals treated with OXI.102, 103 Certain agents, including minocycline, and rapamycin, have been shown to attenuate the neurological side effects of oxaliplatin.104, 105 Oxaliplatin also modulates voltage-gated sodium (Na+) and potassium (K+) channels in neurons. Consequently, Na+ channel inhibitors such as lidocaine, mexiletine, and lamotrigine, as well as K+ channel inhibitors – including the glucosinolate glucoraphanin and isothiocyanates such as sulforaphane, allyl isothiocyanate, phenyl isothiocyanate, and carboxyphenyl isothiocyanate – have been reported to inhibit the development of peripheral neuropathy.106, 107
Gabapentin and pregabalin, which act on the α2δ subunit of voltage-gated calcium channels, have been shown to prevent OIPN.108, 109, 110, 111 Downregulation of transient receptor potential (TRP) channels – temperature-sensitive cation channels – may attenuate OIPN. Agents such as topiramate, acetazolamide, shakuyakukanzoto, goshajinkigan, eel calcitonin, nifedipine, diltiazem, and mexiletine exert their effects through modulation of these channels.112, 113, 114, 115 Modulators of the monoaminergic nervous system, including duloxetine, fluoxetine, vortioxetine, tandospirone, venlafaxine, xaliproden, clomipramine, and clonidine, have demonstrated analgesic effects in animal models of painful OIPN.116, 117, 118, 119, 120, 121, 122, 123, 124, 125, 126, 127, 128, 129 A clinical study was conducted to evaluate the efficacy and safety of duloxetine at a dose of 60 mg for the prevention of OIPN. One study arm received duloxetine, while the 2nd arm received a placebo. Duloxetine (or placebo) was administered during the first 14 days of each chemotherapy cycle. Patients receiving duloxetine experienced a lower incidence of distal paresthesia and throat discomfort compared with those receiving placebo.130
In a separate clinical study, patients with chemotherapy-induced pain following treatment with paclitaxel, another taxane, or OXI were treated with 30 mg of duloxetine or placebo for the 1st week, followed by 60 mg of duloxetine or placebo daily for the subsequent 4 weeks. Patients who received duloxetine reported a significantly greater reduction in pain compared with the placebo group, with 59% of duloxetine-treated patients reporting pain relief, compared with 38% in the placebo group.131 Currently, the European Society of Medical Oncology (ESMO) recommends duloxetine 30 mg daily for 1 week, followed by 60 mg daily, as a therapeutic intervention for CIPN.132 The American Society of Clinical Oncology (ASCO) currently endorses duloxetine as the only recommended agent for the treatment of CIPN.133
Recently published preclinical studies have shown disappointing results regarding the use of a duloxetine–prazosin (α-blocker) combination to prevent the development of allodynia and hyperalgesia during OXI therapy. This combination demonstrated no superiority over duloxetine alone, showing comparable efficacy in preventing OIPN.134 Other drug classes, such as riluzole, mirtazapine, ifenprodil, amitriptyline, trifluoperazine, and dimiracetam, act as modulators of the glutamatergic nervous system and reduce symptoms of OIPN.135, 136, 137, 138, 139, 140, 141, 142
It should be noted that early studies suggested potential benefits of administering intravenous calcium and magnesium before and after OXI treatment to prevent OIPN.143, 144, 145 However, subsequent larger studies failed to demonstrate a reduction in OIPN, indicating that calcium and magnesium are ineffective for the prevention of sensory neuropathy.146, 147, 148
An intriguing therapeutic approach involves the use of topical agents applied to the hands and feet to reduce or prevent symptoms of peripheral neuropathy. Wearing frozen gloves during chemotherapy to prevent CIPN in 180 patients with colorectal or breast cancer has been shown to alleviate certain neuropathic symptoms, such as reduced pain and muscle cramps in the hands and fingers, potentially leading to a modest improvement in quality of life.149
Additionally, a study by Arslan et al. demonstrated that applying henna to the palms, fingers, and soles of female patients after the 2nd, 3rd, and 4th cycles of OXI resulted in a reduction in symptoms of sensory neuropathy.150
The ongoing Danish OxaNeuro trial is a multicenter, double-blind, randomized study including 120 colorectal cancer patients receiving adjuvant OXI. Patients receive daily either fish oil capsules containing long-chain n-3 polyunsaturated fatty acids (n-3 LCPUFAs) or corn oil for a duration of 8 months. The results of this study are not yet available, as patient recruitment continued until the end of 2024.151
Mitochondrial therapy (mitotherapy) is a treatment strategy that relies on the transfer of functional exogenous mitochondria into mitochondria-defective cells to promote cellular recovery. This approach represents a novel therapeutic strategy for diseases caused by mitochondrial dysfunction, such as OIPN. In a preclinical study, mitotherapy was shown to prevent OIPN through anti-neuroinflammatory effects. Therefore, mitotherapy may offer potential as an analgesic adjunct during OXI-based chemotherapy.152
In the search for effective strategies to prevent and/or treat OIPN, the gold standard has been sought among traditional medicinal compounds used in various countries. Huangqi Guizhi Wuwu decoction (HGWD) is a traditional Chinese medicine (TCM) formula that has been investigated for its preventive potential against OIPN. Based on available study results, the preventive effects of HGWD are thought to arise from regulation of intestinal flora homeostasis and a reduction in serum lipopolysaccharide levels, including within the DRG.24
Similarly, Gosha-jinki-gan (GJG), a Japanese herbal medicine, with the expression of the transient receptor potential melastatin 8 (TRPM8) and transient receptor potential ankyrin 1 (TRPA1) channels is well-known for its effectiveness in minimizing symptoms of peripheral neuropathy by suppressing the overexpression of TRPM8 and TRPA1 mRNA.153, 154 Furthermore, a preclinical study demonstrated that 50% hydroalcoholic extract of Astragali radix exhibited analgesic effects and prevented neurological side effects induced by OXI.102
The positive effect of cannabis in oncologic patients treated with OXI was confirmed by Waissengrin et al. The effectiveness was significantly greater in patients who received cannabis prior to treatment with OXI compared with those who received it after the initiation of OXI therapy.155 In another study, daily administration of 300 mg of cannabidiol oil for 8 days, beginning 1 day before the initiation of chemotherapy with either OXI or paclitaxel, reduced the early symptoms of peripheral neuropathy.156 These findings, however, require confirmation in large randomized clinical trials.
Limitations of the study
The problem of neuropathy during OXI treatment remains under investigation and requires continued observation as well as further research into the pathophysiological mechanisms, diagnostic approaches, and therapeutic options. This paper discusses the relevant issues; however, one limitation should be acknowledged. As this is a narrative (literature) review rather than a systematic review, the systematic organization of the presented publications is limited.
Conclusions
Both the prevention and treatment of OIPN require further innovative and effective research. It is hoped that the outcomes of ongoing trials will address the issue of acute and OIPN in the near future, as this remains a persistent clinical concern for patients with gastrointestinal cancer.
Use of AI and AI-assisted technologies
Not applicable.