Abstract
Background. Current guidelines suggest that statins may be beneficial in patients with myocardial infarction (MI) with non-obstructive coronary arteries (MINOCA). However, the available data are ambiguous.
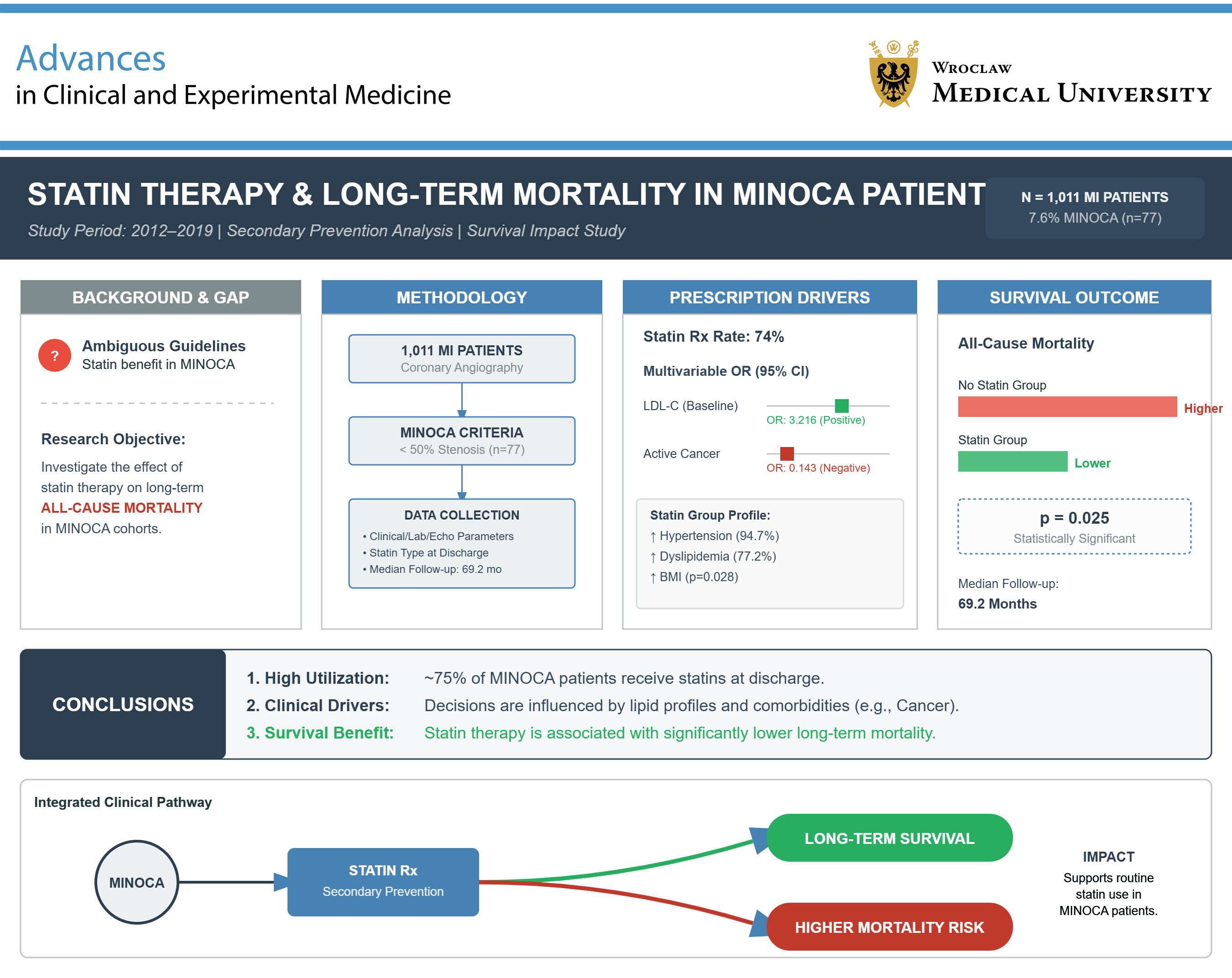
Objectives. We sought to investigate the effect of statin therapy on long-term mortality among MINOCA patients.
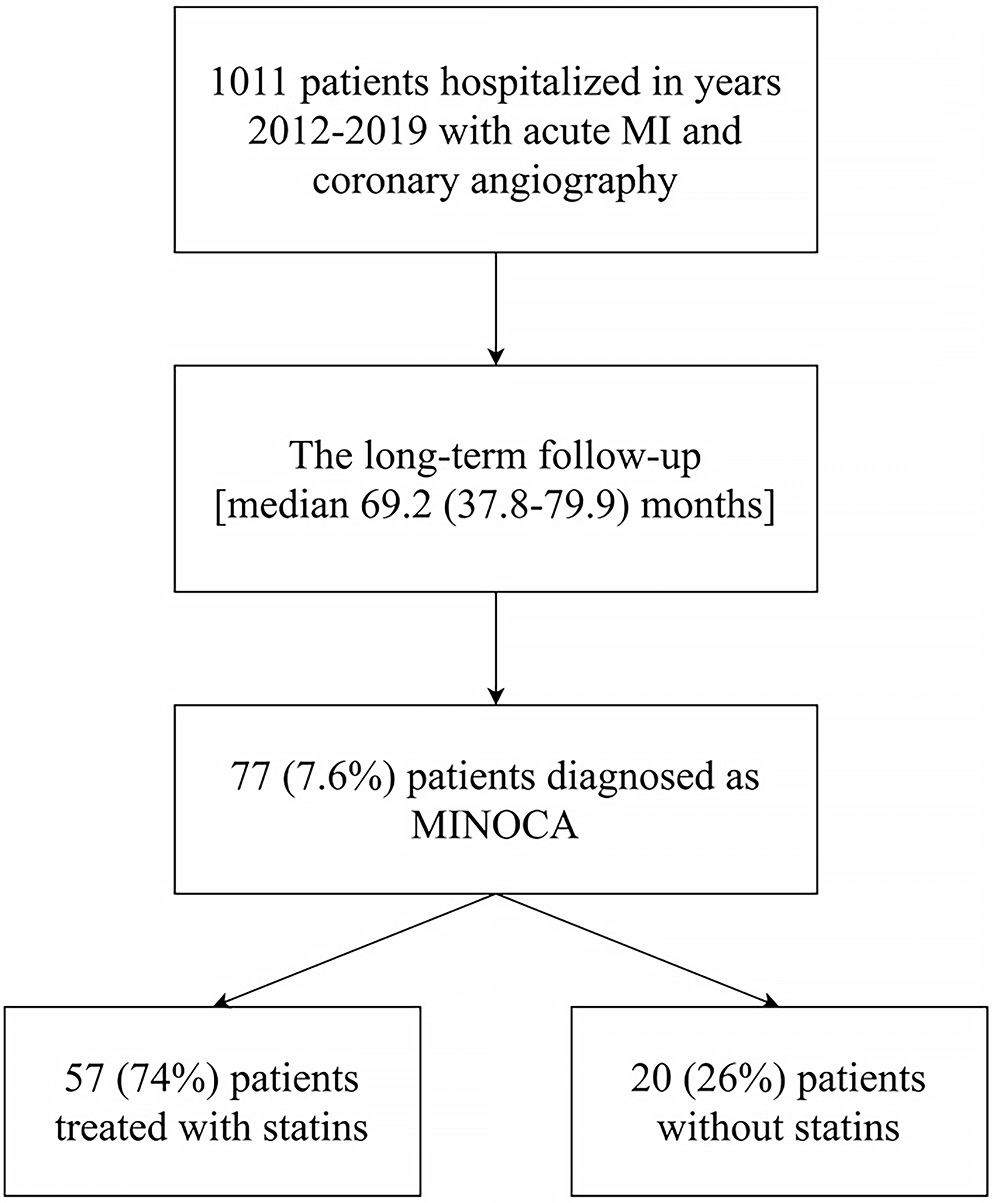
Materials and methods. Between 2012 and 2019, coronary angiography was performed in 1,011 patients hospitalized with a diagnosis of MI. Patients with lesions narrowing coronary arteries by less than 50% were classified as MINOCA. Baseline clinical characteristics, laboratory and echocardiographic parameters, statin type prescribed at discharge, as well as long-term all-cause mortality were collected in the MINOCA group.
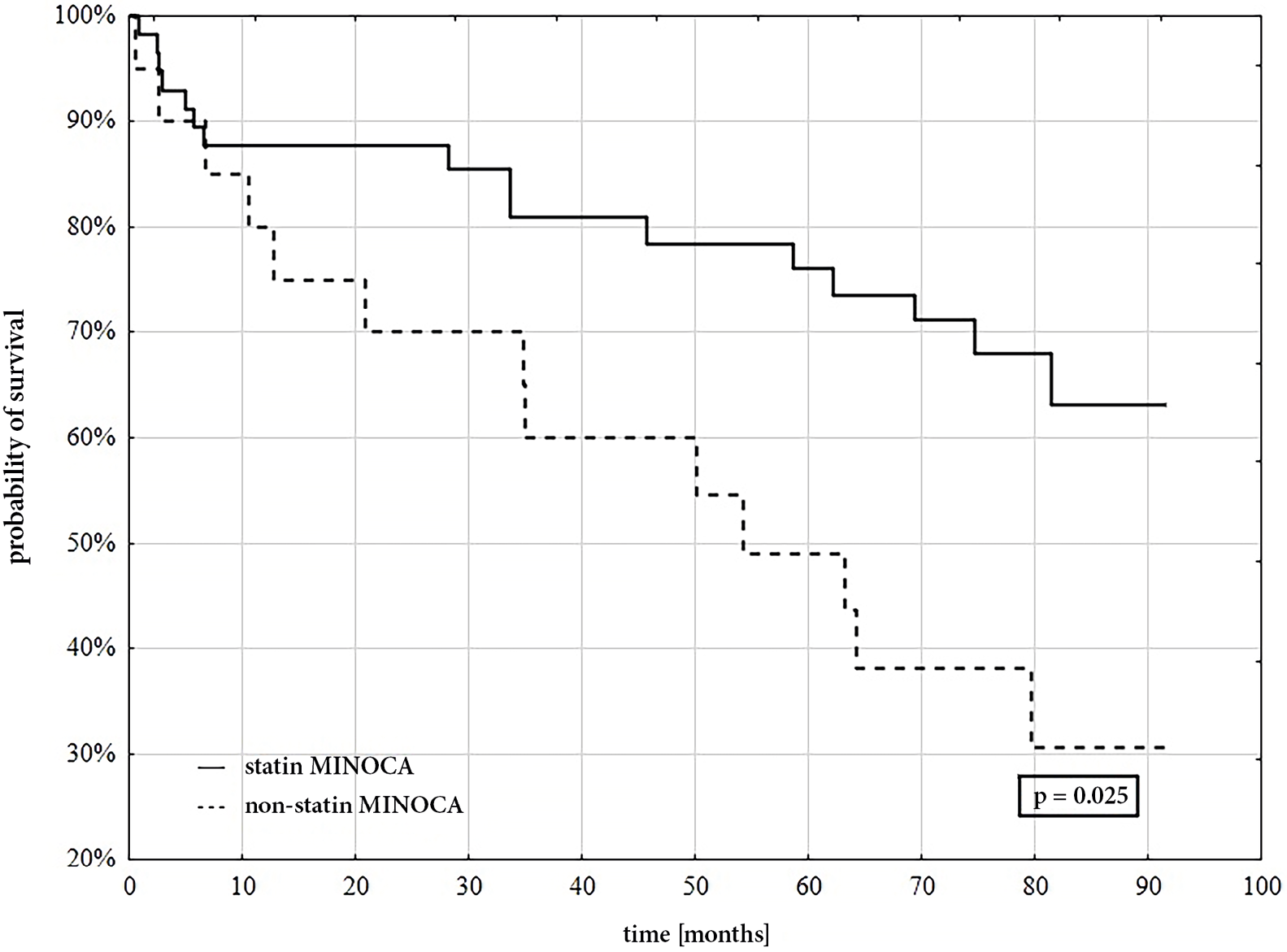
Results. MINOCA was diagnosed in 77 (7.6%) patients, while statins were prescribed in 57 (74%) of them at discharge. Subjects treated with statins had a higher prevalence of hypertension (94.7 vs 70%, p = 0.008) and dyslipidemia (77.2 vs 45%, p = 0.008), had a higher body mass index (BMI; p = 0.028), and were less likely to suffer from active cancer (17.5 vs 55.0%, p = 0.001). In multivariable logistic regression, active cancer independently reduced (odds ratio (OR) = 0.143, 95% confidence interval (95% CI): 0.027–0.748), whereas higher baseline low-density lipoorotein cholesterol (LDL-C) (OR = 3.216, 95% CI: 1.192–8.686) independently increased the likelihood of statin prescription at discharge. In the median follow-up of 69.2 [interquartile range (IQR): 37.8–79.9] months, all-cause mortality was significantly higher in MINOCA patients not treated with statins (p = 0.025).
Conclusions. Roughly 75% of MINOCA patients were treated with statins at discharge. This decision was independently influenced by clinical characteristics and lipid profile. Statin prescription was associated with lower long-term all-cause mortality.
Key words: statin therapy, myocardial infarction with non-obstructive coronary arteries (MINOCA), long-term mortality, secondary prevention, survival analysis
Background
The European Society of Cardiology (ESC) has proposed specific criteria for diagnosing myocardial infarction (MI) in patients with non-obstructive coronary arteries (MINOCA), which include an episode of acute MI with no lesions ≥50% in major epicardial arteries and the exclusion of other identifiable specific causes for the acute presentation.1, 2, 3 It is thought to be present in 5–7% of individuals with acute coronary syndrome and 3–15% of patients with acute MI.4 Atherosclerotic plaque disruption, supply-demand mismatch, coronary microvascular dysfunction, coronary thromboembolism, epicardial coronary vasospasm, and spontaneous coronary artery dissection are some of the possible causes of MINOCA, although the precise pathophysiology of the condition is still unclear.5, 6, 7, 8, 9 Even though the underlying mechanism is most likely complex, 8–25% of individuals still have an unknown reason for MINOCA.3, 6 Additionally, compared with obstructive acute MI, MINOCA patients are more likely to be younger, non-white women with a lower burden of classic cardiovascular risk factors.6, 8, 10
Given the absence of obstructive atherosclerosis, MINOCA patients are often misleadingly classified as “less severe” and as having a better prognosis than those with classic acute MI.5, 6, 8 Although some studies indeed suggest favorable outcomes in MINOCA compared with acute MI,4 others have shown a similar or even worse prognosis of MINOCA.11, 12, 13 Patients with MINOCA still lack comprehensive guidelines regarding the best medical therapy because of the disease’s complicated origin, lack of data, and frequently unclear underlying diagnosis.5, 6, 8 Previous studies have documented that the use of classical secondary prevention in MINOCA was lower compared with acute MI.6, 14 Several studies have shown beneficial effects of statin use in the MINOCA population. Statin-treated patients had a substantially decreased incidence of major adverse cardiac events (MACEs) throughout a mean follow-up of 4.1 years, according to an observational analysis of MINOCA patients from the SWEDEHEART registry.7 However, a prospective trial with an average follow-up of 19 months revealed that while statin therapy was linked to considerably longer survival in a Kaplan–Meier analysis, there was no correlation between statin use and MACE or death in a Cox regression analysis.7 Five observational studies, including those described above, were recently meta-analyzed, with follow-up ranging from 1.3 years to 7.5 years.5 However, a precise explanation of this improved survival, as well as the determinants of statin initiation in MINOCA patients, remains insufficiently elucidated.
Objectives
The aim of our study was to identify independent predictors of statin use and their effect on long-term mortality of MINOCA patients enrolled in the real-life acute MI registry.
Materials and methods
Between 2012 and 2019, coronary angiography was performed in a total of 1,011 patients hospitalized in the Department of Coronary Artery Disease and Heart Failure of the St. John Paul II Specialist Hospital (Cracow, Poland) with a diagnosis of MI (Figure 1). MINOCA was defined in accordance with current guidelines using classic MI criteria without epicardial artery stenosis ≥50%.1, 2, 3 According to ESC guidelines,15 patients with acute coronary syndrome were categorized as either non-ST-segment elevation MI (NSTEMI) or ST-segment elevation MI (STEMI). The included patients were divided into statin and non-statin MINOCA groups based on whether they were taking statins at the time of discharge (Figure 1). Then, long-term all-cause mortality was assessed in all patients (Figure 1). The long-term follow-up data were collected using the Polish National Death Registry.
The study design was approved by the Jagiellonian University Medical College Ethics Committee (approval No. 1072.6120.59.2018). The Declaration of Helsinki was followed when conducting the study. Every person who was enrolled in the study gave informed consent.
Study population
Data were collected on patients’ demographic, laboratory, and echocardiographic results. Previous medical history, cardiovascular risk factors, and medications at discharge were analyzed. Dyslipidemia was diagnosed as plasma lipoprotein concentration above the therapeutic target for low-density lipoprotein cholesterol (LDL-C) >1.8 mmol/L (>70 mg/dL).16 Hypertension was defined according to the ESC guidelines as repeated office systolic blood pressure (SBP) values ≥140 mm Hg and/or diastolic blood pressure (DBP) ≥90 mm Hg.17 Diabetes mellitus was determined by fasting plasma glucose values ≥7.0 mmol/L (126 mg/dL) on 2 separate days, 2-h post-load plasma glucose ≥11.1 mmol/L (200 mg/dL), HbA1c ≥6.5%, or random blood glucose ≥11.1 mmol/L (200 mg/dL) in the presence of signs and symptoms.18 Cancer diagnosed within the last 6 months, requiring treatment with antimitotic drugs at that time, recurring, metastatic, regionally progressed, or inoperable, was classified as active disease. All patients were assessed with the Killip classification on admission.
Laboratory tests performed on admission or within the first 24 h of hospitalization were assessed. Serum total cholesterol, LDL-C, high-density lipoprotein cholesterol (HDL-C), and triglycerides were measured using a Cobas 6000™ biochemical analyzer (Roche Diagnostics GmbH, Mannheim, Germany).
Angiography and echocardiography
Each of the coronary angiograms was analyzed offline. Unaware of the clinical information, an interventional cardiologist evaluated each artery in 2 contralateral projections. Quantitative coronary angiography (QCA Quantcor; Siemens, Munich, Germany) was used for a thorough assessment of borderline lesions between 40% and 70%. Epicardial artery lesions resulting in <50% coronary lumen stenosis were considered angiographically insignificant.1, 2, 3 All patients with insignificant stenosis were categorized into 2 groups: 1) those with normal coronary arteries or minimal intracoronary irregularities defined by stenosis <30%, or 2) those with mild to moderate lesions characterized by ≥30% and <50% lumen narrowing.
Between the 2nd and 4th day of hospitalization, a qualified medical professional performed 2-dimensional transthoracic echocardiography. Using a Vivid S5 ultrasound system (GE Healthcare, Solingen, Germany) equipped with a multi-frequency harmonic transducer (3Sc-RS, 1.3–4 MHz), the examination was conducted while the patient was at rest in the left decubitus position. Every measurement was performed in line with the guidelines provided by the European Association of Echocardiography (EAE)19 and the American Society of Echocardiography (ASE). To characterize distinct cardiac components and facilitate their functional evaluation, standard data were gathered.20
Statistical analyses
Continuous variables were first assessed for normal distribution using the Lilliefors test and expressed as mean ± standard deviation (SD) for normal distribution, and median (Q1–Q3) for non-normal distribution, whereas categorical variables were expressed as number (percentage). Continuous variables were compared using Student’s t-test with Levene’s test for variance homogeneity or the Mann–Whitney U test if the distribution was normal or non-normal, respectively. Categorical variables were analyzed using the χ2 test or Fisher’s exact test. Fisher’s exact test was performed if any of the expected counts were less than or equal to 5. All parameters based on theoretical knowledge associated with statin treatment were included in the multivariable logistic regression model. In the logistic regression model, the assumption of linearity of all continuous predictors was checked with the Box–Tidwell test, the assumption of lack of multicollinearity with the variance inflation factor (VIF), and the assumption of no influential outliers based on Cook’s distance. Kaplan–Meier curves for all-cause mortality in the studied groups were constructed and compared using the Gehan–Breslow–Wilcoxon test. Statistical significance was defined as a 2-sided p-value less than 0.05. All statistical analyses were conducted using Statistica v. 13.3 (StatSoft Poland, Cracow, Poland) or IBM SPSS v. 29.0 (IBM Corp., Armonk, USA).
Results
Baseline characteristics
Of 1,011 consecutive acute MI patients, 77 (7.6%) were diagnosed with MINOCA. Of these, 57 (74%) were treated with statins and 20 (26%) without statins at discharge (Figure 1). Women constituted the majority in both groups – 50.9% in the statin MINOCA group and 55% in the non-statin group (Table 1). MINOCA patients treated with statins had a higher body mass index (BMI; 26.8 [interquartile range (IQR): 24.2–32.0] kg/m2 vs 23.6 [21.8–29.3] kg/m2, p = 0.028), with a more frequent diagnosis of hypertension (94.7 vs 70%, p = 0.008) and dyslipidemia (77.2% vs 45%, p = 0.008), as well as a lower prevalence of active cancer (17.5% vs 55%, p = 0.001) (Table 1).
Subjects in both groups did not differ in clinical presentation (NSTEMI vs STEMI), but patients in the statin MINOCA group less frequently showed Killip class III/IV on admission (3.5% vs 20%, p = 0.037) (Table 1). The severity of nonsignificant atherosclerotic lesions was similar in both groups (Table 1). The use of the most important drug classes in both compared groups was similar (Table 1).
The analysis of laboratory parameters assessed on admission did not show significant divergence between the 2 analyzed populations, except for LDL-C, which was higher in MINOCA patients treated with statins compared to those without statins by 25.9% (2.7 [1.7–3.4] mmol/L vs 2 [1.5–2.6] mmol/L, p = 0.047) (Table 2).
In subjects receiving statin therapy, a tendency toward higher values of left ventricular ejection fraction (LVEF) (55% [45–60%] vs 47% [30–55%], p = 0.055) was observed, with no differences in other echocardiographic parameters (Table 3).
Statins regimen
As shown in Table 4, the most prescribed statin at discharge was atorvastatin (59.6%), with the most frequent doses of 20 mg (52.9%) and 40 mg (41.2%). The 2nd most common statin was simvastatin (21.1%), with 20 mg as the most common dose (75%). The least frequently prescribed statin, in only 19.3% of patients, was rosuvastatin. Notably, ezetimibe was not administered to any of the patients in the analyzed population (Table 4).
Independent predictors of statin use
The assumptions regarding linearity between predictors, lack of multicollinearity between explanatory variables, and absence of influential outliers were met for the model used to determine independent predictors of statin use. The multivariable logistic regression analysis revealed that active cancer (odds ratio (OR) = 0.143, 95% confidence interval (95% CI): 0.027–0.748) independently reduced, whereas higher baseline LDL-C (OR = 3.216, 95% CI: 1.192–8.686) independently increased the likelihood of statin prescription at discharge (Table 5).
Long-term mortality
In the median follow-up period of 69.2 [37.8–79.9] months, all-cause mortality was significantly higher in MINOCA patients who did not receive statin therapy (n = 13, 65%) compared with the others (n = 16, 28.1%) (p = 0.025) (Figure 2).
Discussion
The current study demonstrated that 75% of patients with MINOCA received statins at discharge. The decision to introduce statins was made based on clinical characteristics. Moreover, a large proportion of patients were not receiving the currently recommended high doses of statins, and none of the patients was receiving lipid-lowering combination therapy. Given the demonstrated positive association with long-term survival, our data may support the routine use of statins also in the MINOCA population.
As has been mentioned above, there is evidence on the positive impact of statins on survival and outcomes in MINOCA patients. In the largest synthesis of 5 observational studies, Masson et al. showed that statins were administered to 81.7% of patients with MINOCA, which led to a significant decrease in mortality and MACEs.5 The results of this investigation are consistent with both the better prognosis and the prevalence of statin use.5 However, there has not been any prior analysis of statin types, dosages, or predictors of their use.5 Randomized clinical studies assessing the role of statins in the MINOCA population have not yet been reported. Additionally, none of the research has discussed the individualized therapy that is now advised based on the etiology of MINOCA. It is known that statins can improve exercise duration and time to ST-segment depression in patients with syndrome X and are favorable in vasospastic angina by increasing endothelial-dependent vasodilation.21, 22 However, some studies suggest that the use of statins may not be appropriate for clinical conditions not directly related to cholesterol accumulation. Retrospective data from the Mayo Clinic group suggest a slightly increased incidence of spontaneous coronary artery dissection recurrence in patients using statins.23
The direct mechanism explaining the positive prognostic effect of statins in MINOCA remains unknown. Statins work primarily by inhibiting HMG-CoA reductase, an enzyme that regulates the process of cholesterol biosynthesis in the liver.24 However, the numerous pleiotropic benefits of statins, regardless of LDL-C levels, are even more substantial in the case of MINOCA, given its mostly non-atherosclerotic origin, which is characterized by the absence of major lesions in the coronary arteries.25 By increasing nitric oxide (NO) generation, statins may affect endothelial function through a variety of pleiotropic mechanisms.26, 27 Reduction of systemic and vascular inflammation is another pathway in the general population that is not dependent on LDL-C levels. Ridker et al. demonstrated that among a healthy population without high lipid levels but with elevated high-sensitivity C-reactive protein (CRP) levels, rosuvastatin, compared to placebo, significantly lowered the incidence of MACE as well as all-cause mortality.28 Furthermore, the MIRACL study found that individuals with acute coronary syndrome experienced a greater reduction in inflammation while taking high doses of atorvastatin.29 Additionally, it has been shown that statins have antithrombotic properties. According to reports, they promote protein C activation and reduce thrombin production while improving fibrin coagulation properties.25 The JUPITER study found that using 20 mg of rosuvastatin significantly decreased the incidence of venous thromboembolism in healthy individuals over a median follow-up of nearly 2 years.30 As we recently showed, individuals with stable coronary heart disease and venous thromboembolism had lower levels of coagulation factors while taking high doses of statins.31, 32 Considering the emerging reports on hypercoagulable states in MINOCA, the antithrombotic effect of statins may be crucial and could be a potential explanation for the observed mortality reduction.9
It has been shown that statins are less commonly prescribed in MINOCA compared with acute MI patients with obstructive coronary artery disease (CAD).8 However, it has not been determined why, since according to current guidelines, statins should be prescribed in all patients with acute MI.3 Our findings indicate that the lack of active cancer and higher LDL-C levels were the 2 independent determinants associated with the prescription of statins at discharge. The reasons for not prescribing statins in patients with cancer may be multifactorial and include a lack of potential benefits from treatment and worsening of health status due to neoplastic disease progression. Moreover, the absence of visible stenosis in the arteries could indicate less frequent prescription in this group than in MI patients with obstructed coronary arteries. In the study by Ishii et al., the only clinically relevant factor that influenced statin prescription in patients with coronary vasospastic angina was the presence of dyslipidemia.33 Our real-life results indicate that the decision to include statins in MINOCA is based on clinical characteristics and is not as strictly followed as in the case of classical acute MI. However, these results require verification in further studies.
Limitations of the study
It is important to note several limitations. First, we were able to obtain only data concerning all-cause mortality in the long-term observation without separation of cardiovascular and non-cardiovascular mortality, so a detailed analysis was not possible. Second, our study did not analyze adherence to the treatment regimen after hospitalization in patients with MINOCA, which may have affected the results. Moreover, it could also be speculated that the observed associations do not necessarily indicate a direct prognostic impact of statin use and should preferably be verified in further randomized studies. The current study relied on data from our center and comprised a relatively small number of patients. Finally, we refrained from using advanced imaging techniques, especially magnetic resonance imaging (MRI) and intracoronary imaging (ICI), as the diagnostic significance of these procedures was still uncertain at the time of patient enrollment in the registry.34 More research should be conducted to determine the relationship between statin administration and the exact MINOCA etiology.
Conclusions
This real-life study found that statins are administered in 3 out of 4 MINOCA patients after diagnosis. This decision was independently determined by clinical characteristics and lipid profile, but not by the MINOCA diagnosis itself. Moreover, a large proportion of MINOCA patients do not receive the currently recommended high doses of statins. As has been shown, in-hospital statin initiation was associated with lower all-cause mortality in the long-term observation. Further randomized controlled studies are required to determine the direct prognostic impact of statins and their appropriate doses in the MINOCA population.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.17600120. The package contains the following files:
Supplementary Table 1. The characteristics of statistical tests used for the assessment of baseline clinical data.
Supplementary Table 2. The characteristics of statistical tests used for the assessment of baseline laboratory data.
Supplementary Table 3. The characteristics of statistical tests used for the assessment of echocardiographic parameters.
Supplementary Table 4. The characteristics of statistical tests used for the assessment of the normality assumption (Lilliefors test) and the homogeneity of variances assumption (Levene’s test).
Data Availability Statement
The datasets supporting the findings of the current study are openly available in Zenodo at https://doi.org/10.5281/zenodo.17598275.
Consent for publication
Not applicable
Use of AI and AI-assisted technology
Not applicable