Abstract
Neuroimaging techniques such as magnetic resonance imaging (MRI) are routinely used in diagnostic radiology to evaluate brain changes associated with neurological and psychiatric conditions. Evidence suggests that imaging biomarkers predict clinical outcomes with varying accuracy across ethnic groups. Underrepresentation of ethnic diversity in neuroimaging research may result in generalization bias, whereby findings derived from one population are inaccurately applied to others. A scoping review methodology was employed to systematically identify and analyze relevant literature. Searches were conducted across EBSCOhost (including CINAHL and Medline Complete), Elsevier (Scopus), Clarivate (Web of Science), and PubMed. Eligible studies examined ethnicity-related differences in subcortical brain volumes and cortical thickness in healthy adults using neuroimaging. The search yielded 1,013 records, which were screened according to predefined inclusion and exclusion criteria. Fourteen studies met the eligibility criteria and were included in the final analysis. The reviewed studies demonstrated significant variations in cortical thickness and subcortical volumes across diverse populations and socioeconomic groups, underscoring the need for population-sensitive reference standards in neuroimaging to minimize generalization bias. These findings highlight the importance of incorporating ethnic variability into neuroimaging research and developing population-sensitive frameworks for MRI-based diagnostic applications. Additionally, the review identifies key areas for improvement, including the integration of ethnic and socioeconomic diversity in artificial intelligence (AI)-driven neuroimaging models to enhance diagnostic precision and equity.
Key words: brain, magnetic resonance imaging, cortex, artificial intelligence, ethnicity
Introduction
Alzheimer’s and Parkinson’s diseases are among the most prevalent neurodegenerative disorders, significantly impacting global health. Over 57 million people were living with dementia worldwide in 2019.1 This number is projected to rise to more than 153 million by 2050 due to population aging. Neuroimaging techniques, particularly magnetic resonance imaging (MRI) and positron emission tomography (PET), are invaluable for diagnosis. The integration of artificial intelligence (AI) into imaging enhances segmentation, lesion detection, and volumetric analysis, thereby facilitating the identification of subtle structural changes and improving early diagnosis of neurodegenerative and psychiatric disorders.2
Beyond neuroimaging, AI applications in health sciences continue to expand. For example, Razdan et al.3 explored the role of AI in pediatric dentistry by analyzing dentists’ perceptions of its diagnostic capabilities. Furthermore, AI models have been developed to assess pediatric frailty syndrome in heart failure4 and pediatric-onset multiple sclerosis.5 The cortical and subcortical brain regions are involved in higher-order cognitive processes, including decision-making, language, complex motor control, emotion, motivation, and basic survival functions, and play a fundamental role in behavior. Specific cognitive domains are associated with distinct brain structures. Memory is linked to hippocampal and entorhinal cortex volumes; attention and processing speed are related to parietal lobe volumes and white matter integrity6; executive functions involve the prefrontal cortex and caudate7, 8; emotional processing and social cognition are associated with amygdala volume8; and global cognitive and social functioning have been linked to thalamic volume.9 Additionally, the prefrontal cortex (PFC) plays a key role in executive functioning, emotional regulation, and decision-making,10 while the pre-supplementary motor cortex and the right inferior frontal gyrus are implicated in reactive action control.11 These brain structures serve as important biomarkers of neurological disorders; their morphology aids diagnosis, monitoring of disease progression, and prognostication. Alterations in cortical thickness and regional brain volumes – including total brain volume, parietal and temporal gray matter, and the hippocampus – are well-established biomarkers of neurodegenerative diseases such as Alzheimer’s disease (AD) and related dementias,12, 13, 14 Parkinson’s disease (PD),15 Huntington’s disease,16 amyotrophic lateral sclerosis (ALS),17 and global cognitive decline.6 Cortical structural abnormalities are likewise observed in psychiatric disorders, including major depressive disorder,18, 19 schizophrenia,20 and bipolar disorder.10, 21, 22
Furthermore, volumetric measures of subcortical structures – including the thalamus, basal ganglia, hippocampus, amygdala, and nucleus accumbens – serve as key diagnostic biomarkers. Subcortical structural alterations have been documented in a range of neurological and psychiatric disorders, including AD,23 PD,24 schizophrenia, bipolar disorder, major depressive disorder, and autism spectrum disorder.25, 26, 27
Ethnic differences in brain volumes in both health and disease are well documented in the literature and have important implications for clinical practice and research. For example, studies comparing Caucasian and African American populations,28 as well as Korean and other East Asian cohorts,29, 30 have reported significant differences in cortical and subcortical volumes. In a multiethnic study, Black participants demonstrated larger total brain volumes, while both Black and Hispanic participants exhibited higher white matter volumes compared with other groups.31 Differences in pathological markers have also been observed. Black participants showed a greater burden of white matter hyperintensities (WMH), whereas Japanese individuals demonstrated less functional disability despite comparable levels of brain atrophy.31
Understanding differences in these brain biomarkers is crucial for accurate diagnosis, particularly in the context of AI-driven neuroimaging. Although genetic ancestry may contribute to intergroup variation in brain morphology,30 such differences are more consistently associated with social determinants of health, including income, education, access to healthcare, and chronic stress exposure.31, 32 Recognizing race as a sociopolitical construct rather than a purely biological determinant33, 34 helps prevent the perpetuation of biological essentialism within AI-based diagnostic frameworks.35, 36 A significant challenge arises from the fact that commercially available AI-enabled neuroimaging software is primarily trained on data derived from geographically limited populations, particularly in the USA, Europe, and China,37, 38, 39, 40 thereby introducing potential bias. Consequently, the development of inclusive AI models with broader population representation – integrating biological, social, and geographic determinants – is essential to enhance diagnostic equity and generalizability.
This review critically examines evidence from the literature on how racial, ethnic, and social determinants, conceptualized as sociopolitical and structural factors, influence brain volumetric variability. Throughout the review, racial and ethnic groupings are used within a socio-structural context rather than as biologically fixed categories. Moreover, we adopt the term “population-sensitive” to describe approaches that consider both biological variation (e.g., ancestry and sex) and socio-structural determinants (e.g., race, ethnicity, and socioeconomic status).
Although the cited literature variably uses the terms “race,” “ethnicity,” and related constructs, our use of the term “population-sensitive” aims to provide a more inclusive and precise framework that avoids reinforcing essentialist or reductive interpretations. While acknowledging the diagnostic benefits of race-aware AI models and population-specific templates, we also highlight the associated ethical risks, including racial essentialism and algorithmic bias. This review emphasizes the importance of evidence-based, bias-reducing strategies that prioritize diagnostic accuracy, equity, and scientific rigor.
Objectives
This scoping review explores differences in cortical and subcortical volumes among diverse populations and addresses the following research questions:
– What is known about cerebral cortical and subcortical volumetric differences across ethnic groups, and how do these differences affect AI-based neuroimaging tools?
– What is the role of specific cortical and subcortical brain structures in health and in neurological and psychiatric conditions?
– How do normative brain volumes across different population groups influence diagnostic accuracy, AI model generalization, and population-sensitive standards?
– What are the potential challenges, biases, and ethical considerations involved in incorporating ethnicity and socioeconomic status (SES) when developing population-sensitive norms and AI models in neuroimaging?
Concept
This scoping review, based on MRI studies, examines population-specific differences in brain volumetry among healthy adults and their implications for ensuring accurate diagnoses and equitable AI-driven assessments. As described earlier, the term “population-sensitive” is used to highlight a comprehensive approach that integrates biological variation (e.g., ancestry and sex) with socio-structural influences (e.g., race, ethnicity, and socioeconomic status). A population-sensitive perspective emphasizes diagnostic precision and equity while preventing the reinforcement of racial essentialism in AI development.
Context
Given the increasing integration of AI into diagnostic processes, population-level diversity in brain volumetry is a key factor in interpreting MRI scans, necessitating population-sensitive reference standards. This review identifies key themes and evaluates the feasibility of personalized diagnostic standards to enhance the accuracy and fairness of AI-based neuroimaging.
Materials and methods
This scoping review follows the framework proposed by Arksey and O’Malley41 and later refined by Levac et al.42 to systematically explore ethnic differences in brain volumetry among healthy adults using MRI data. Scoping reviews are useful for mapping key concepts in emerging fields, identifying research gaps, and guiding future investigations.43 Although scoping reviews do not require formal quality appraisal,44 this study enhances transparency and reliability by acknowledging the methodological limitations of the included studies and presenting a structured summary table.
Search strategy
Initially, we conducted a preliminary search across the EBSCOhost databases (including CINAHL (Cumulative Index to Nursing and Allied Health Literature) and MEDLINE Complete), Elsevier databases (Scopus), Clarivate databases (Web of Science), and PubMed, which yielded no published reviews on the specified topic. Subsequently, a 2nd search was performed across the same databases to identify relevant articles published between 2014 and 2024, in order to capture the most recent data and refine the search output. The search terms were (“brain anatomy” OR “brain size” OR “brain structure” OR “brain morphology” OR “brain volume” OR “brain volumetric” OR “brain morphometry”) AND (“race” OR “racial” OR “ethnicity” OR “ethnic” OR “racioethnic”).
To enhance clarity and provide specific support for the need for population-sensitive diagnostic standards, the search specifically focused on studies examining ethnic variability in brain morphology. We included studies comparing cortical and subcortical brain volumes across different racial and ethnic groups to ensure that our review captured the full scope of morphological differences relevant to neuroimaging practice.
This review included studies that analyzed brain volumes using MRI across different ethnic groups, as well as studies that compared brain volumes among different ethnicities. Only studies involving healthy adult populations were included. The search encompassed peer-reviewed studies published in English and other languages.
Studies were excluded if they: focused primarily on adolescent or pediatric populations; did not compare volumetric outcomes across racial or ethnic groups; used imaging modalities other than MRI; or lacked full-text availability.
Selection of studies
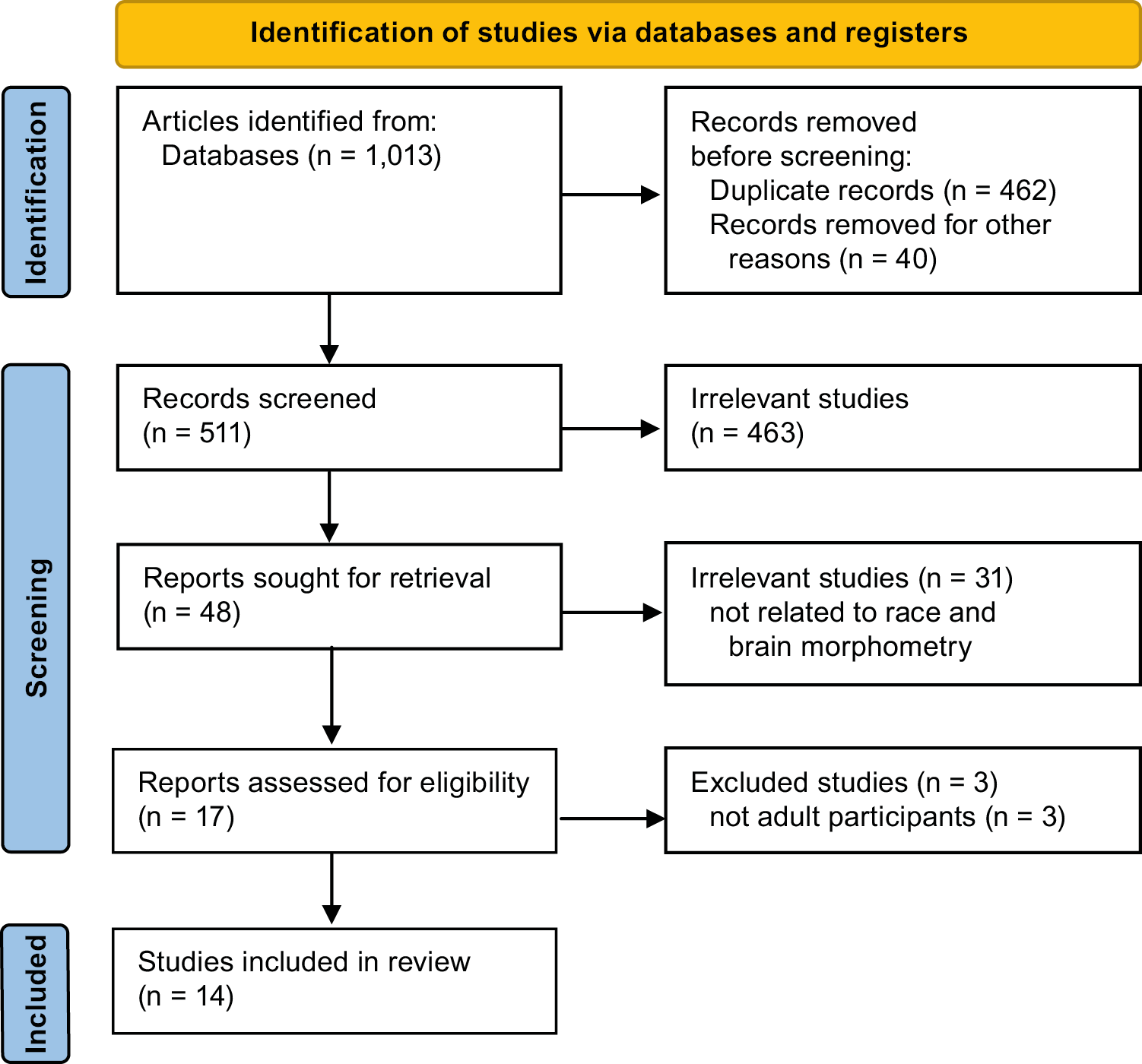
All citations retrieved through the database search were imported into the Zotero reference management software (https://www.zotero.org). Two independent reviewers (A.J. and S.R.) assessed the titles and abstracts to determine their relevance according to the inclusion criteria. Disagreements regarding study inclusion were resolved through discussion to ensure consistency in the selection process. Following the initial screening phase, full-text versions of the selected articles were reviewed, and the exclusion criteria were applied. Excluded studies were documented, with justifications provided for their removal. The final selection process followed the Preferred Reporting Items for Systematic Reviews and Meta-Analyses Extension for Scoping Reviews (PRISMA-ScR) flowchart (Figure 1). A standardized data extraction form was used to chart study design, population characteristics, MRI modalities, segmentation software, and key findings. Two reviewers (A.J. and S.R.) independently extracted the data, and discrepancies were resolved by consensus.
Study heterogeneity
Studies included in this review varied in MRI acquisition protocols, analytical approaches, and population demographics, which may introduce inconsistencies in the findings. To manage this heterogeneity, we: 1) identified common study designs (predominantly cross-sectional); 2) highlighted differences in MRI modalities and processing methods; and 3) evaluated key limitations, such as scanner variability, small sample sizes, and confounder adjustment (e.g., for age, sex, and SES).
Quality assessment and methodological limitations
Although a formal risk-of-bias assessment was not mandated, we conducted a structured methodological appraisal using the Joanna Briggs Institute (JBI) Checklist for Analytical Cross-Sectional and Cohort Studies.44 The identified limitations are summarized in Table 128, 29, 30, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55 and include: variability in MRI protocols; differences in volumetric measurement software; limited ethnic diversity and generalizability; and inconsistent adjustment for confounding variables. To complement Table 228, 29, 30, 45, 46, 47, 48, 49, 51, 52 and Table 350, 54, a domain-based summary of the potential risk of bias across the included studies is presented, based on the JBI critical appraisal tool for cross-sectional and cohort studies. Each study was evaluated across key domains, including clarity of inclusion criteria, validity of outcome measurement, and management of confounders. This visual overview provides a structured understanding of the distribution of bias risk across the evidence base.
Data analysis
Thematic analysis was performed following Braun and Clarke’s six-step framework.56 This process included familiarization with the findings, coding, and synthesis of themes related to racial differences in brain structure, diagnostic implications, and challenges associated with AI-based models. Given the high methodological heterogeneity, a meta-analysis was not feasible.57 Instead, a qualitative synthesis was conducted to explore nuanced patterns in population-specific volumetric variability.
Results
Search outcome
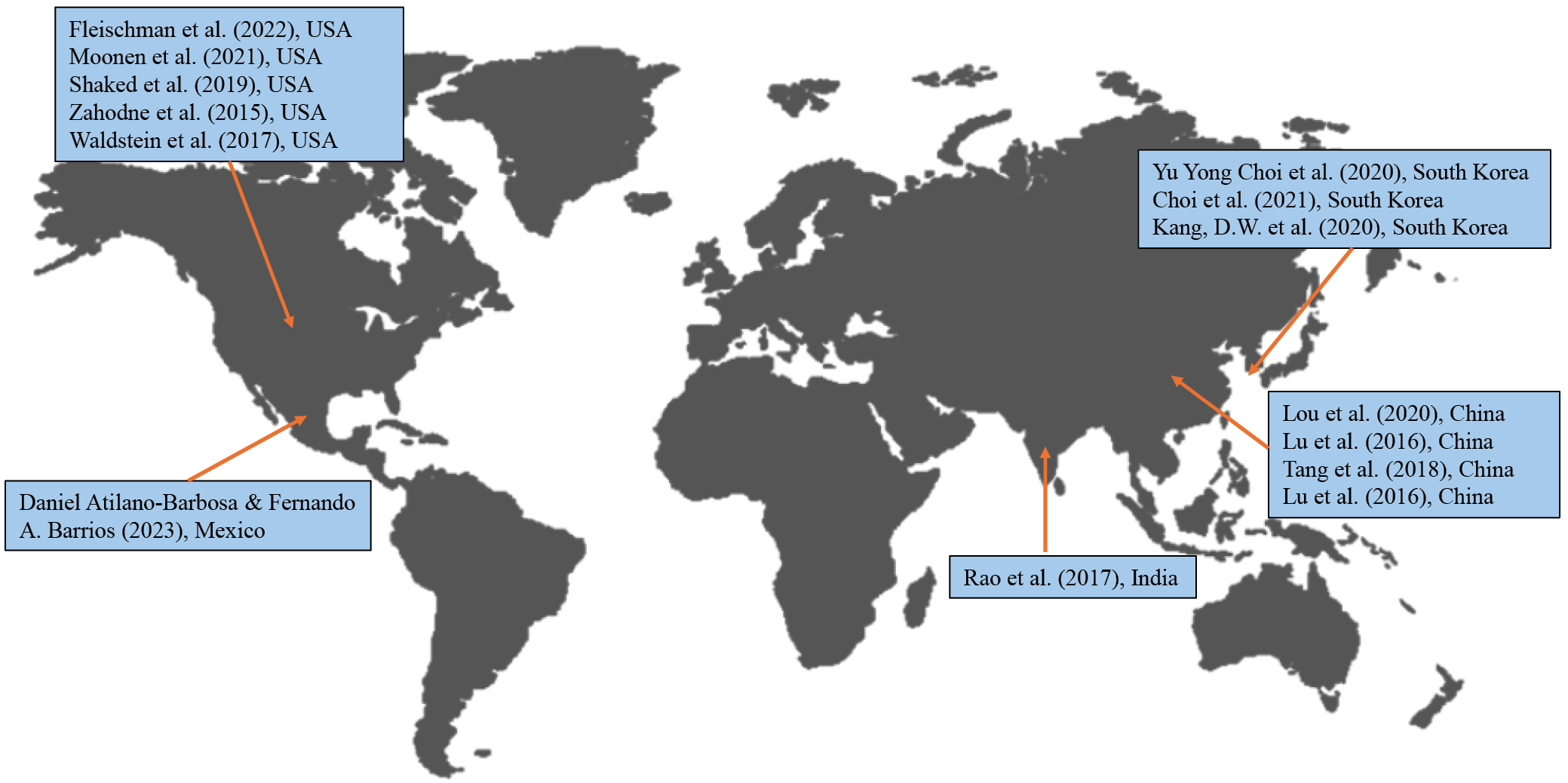
Initially, 1,013 research articles, including publications in English and other languages, were identified across 4 databases (EBSCOhost, Elsevier, Clarivate, and PubMed). During the identification stage, 462 duplicate records were removed, and an additional 40 articles were excluded due to lack of full-text availability. The remaining 511 articles were screened based on titles and abstracts, and 463 were excluded as they were not relevant to the review topic. The remaining 48 articles underwent full-text review, and 31 were excluded because they did not focus on racial differences in brain volumetric outcomes. Thus, the remaining 17 relevant articles were assessed against the exclusion criteria, and 3 were excluded because the primary study population consisted of adolescents. Consequently, this scoping review included 14 articles with comparative and cross-sectional observational study designs that provided evidence relevant to the research questions. The shortlisted studies were conducted in the USA, Mexico, South Korea, China, and India (Figure 2, Table 1).
Thematic analysis of the shortlisted studies
This scoping review synthesizes findings from 14 multinational studies published between 2014 and 2024, encompassing diverse populations, including Caucasian, East Asian, South Asian, Latinx, and African American cohorts. Specifically, findings from the shortlisted studies were categorized into 3 major themes: 1) race/ethnicity-specific differences in brain morphometry; 2) the impact of SES and race/ethnicity on brain health; and 3) the role of racial identity in cognitive aging and brain health trajectories. These themes collectively emphasize the interplay between genetic, environmental, and socioeconomic factors in shaping brain aging and disease susceptibility.
Theme 1: Race/ethnicity-specific differences in brain morphometry
Studies consistently reported structural differences across ethnic groups in cortical thickness, surface area, and subcortical volumes. The most frequently cited regions included the frontal, temporal, parietal, and occipital lobes, as well as subcortical structures such as the hippocampus and choroid plexus.28, 29, 30, 45, 46, 47, 48, 49, 50 For instance, Chinese participants generally exhibited larger temporal and cingulate cortices but smaller frontal and parietal regions compared with Caucasian participants.49 Similarly, significant morphometric differences were observed between Chinese and Indian groups, particularly in gray matter distribution within the frontal, temporal, and occipital lobes.47 Studies comparing East Asian groups (Koreans and Uyghurs) with White participants reported widespread differences in cortical thickness and surface area, especially in language-associated regions such as the frontoparietal operculum.29, 30, 46 In U.S.-based samples, African American participants exhibited distinct patterns of white matter and subcortical volume compared with White participants,28 with larger hippocampal volumes associated with better memory outcomes among non-Hispanic White individuals.50 These group-level differences in brain volume should be understood as arising from interacting factors such as ancestry, environmental exposures, education, and systemic inequalities, rather than as evidence of inherent biological distinctions. This perspective supports the view that population-level differences are shaped by environmental influences over time rather than by innate biological traits.
Theme 2: Impact of socioeconomic status and race/ethnicity on brain health
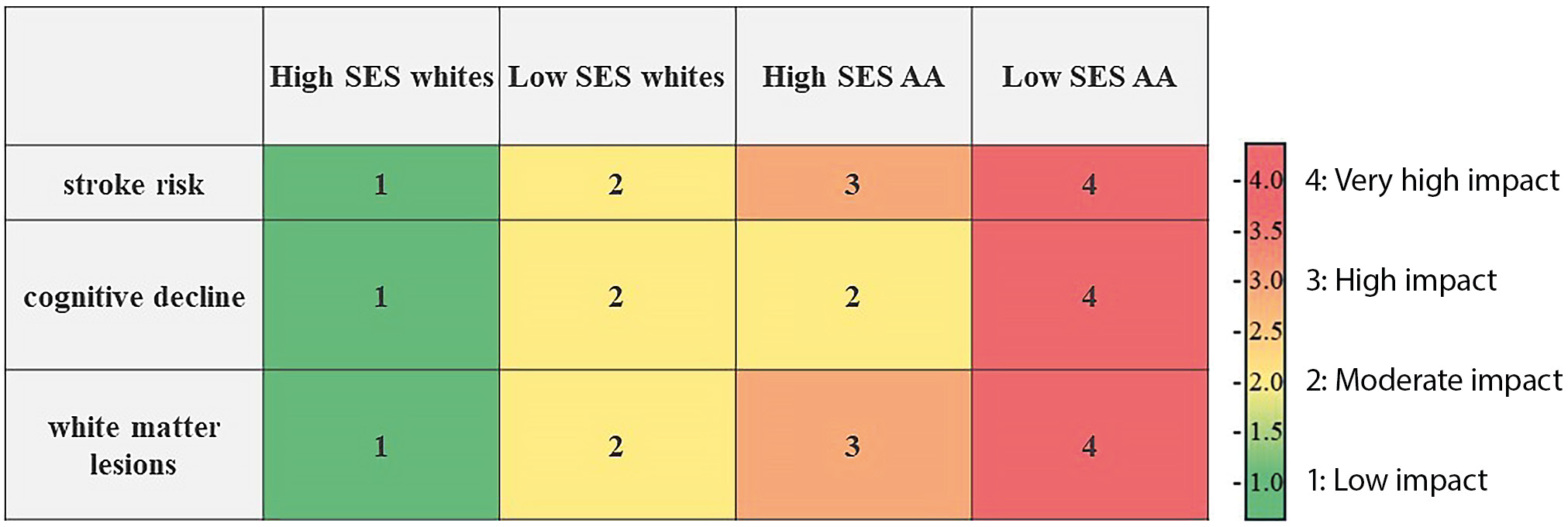
The interplay between race/ethnicity, SES, and brain health is a significant factor in understanding neurodevelopment and cognitive aging. Disparities in education, income, access to healthcare, and employment stability influence brain structure and function, with cumulative exposure to stressors exacerbating racial disparities in neurological health (Figure 3).51, 52 Waldstein et al.52 found that higher SES was associated with neuroprotection against cognitive decline and cerebrovascular damage, predominantly among White populations, whereas African American participants derived fewer protective benefits from SES advantages. This finding suggests that economic gains alone may not counteract the effects of chronic stressors that disproportionately affect racial minority groups. Zahodne et al.50 identified racial disparities in WMH, an MRI marker of cerebrovascular health. African American participants exhibited larger WMH volumes, which were strongly associated with poorer executive function, whereas this pattern was not observed in non-Hispanic White participants.50 Increased WMH burden was associated with lower psychomotor speed and poorer executive function, reflecting overall cognitive status and brain health.58 Fleischman et al.53 found that lower hippocampal volume was associated with episodic memory deficits in aging Black adults. Despite these well-documented disparities, current AI-driven diagnostic models in neuroimaging often fail to incorporate SES-related variables, potentially exacerbating healthcare inequities.
Theme 3: The impact of racial identity on cognitive aging and brain health trajectories
Racial identity influences not only baseline brain structure but also the trajectory of cortical atrophy during aging. Studies suggest that cortical thinning accelerates with age, typically worsening after 70 years of age,59 although these patterns vary significantly across racial and gender groups. Choi et al.29 observed that Caucasian women had steeper rates of cortical atrophy compared with Korean women, suggesting the influence of potential cultural or environmental protective factors. Moonen et al.54 reported faster gray matter decline in Black men than in White individuals or Black women, likely reflecting chronic stress exposure and systemic inequalities. These findings are supported by studies indicating that brain regions involved in language and cognition, such as the frontal and temporal lobes, are particularly susceptible to social determinants of health.60, 61
Discussion
This scoping review examines the need for population-sensitive neuroimaging standards by synthesizing evidence on differences in brain morphology and their implications for AI-based diagnostics, while critically addressing the concept of biological essentialism.
Influence of biological and SES factors and associated risk of bias
While biological distinctions in brain morphology may support the development of population-sensitive neuroimaging reference standards, these differences are shaped by evolutionary adaptations30, 62 and broader factors, such as SES and systemic discrimination, which accelerate brain aging in marginalized groups, as evidenced by accelerated gray matter decline among Black adults.34, 54 This suggests that the observed differences in brain morphology are more accurately attributed to socioeconomic and environmental determinants than to innate biological variation.34, 63 Similarly, African American participants demonstrate a stronger association between larger WMH volumes and poorer cognitive function compared with non-Hispanic White individuals.50 Documented evidence of ethnic disparities in biomarkers of AD and PD highlights the risk of misclassification if AI models are trained on homogeneous datasets, potentially leading to inaccurate diagnoses in diverse populations.29, 50
Advantages and limitations of population-sensitive templates
Developing population-sensitive MRI templates and normative brain volumes (NBVs) offers potential advantages for diagnostic accuracy. Studies by Choi et al.29, 55 demonstrated that population-specific templates significantly enhance diagnostic precision and reduce prediction errors in neurodegenerative disease models. For example, an MRI template developed for Indian individuals was tested on MRI scans from an Indian population, yielding better alignment compared with the standard MRI template based on a Caucasian population.48
In addition, Caucasian normative brain volume references were unable to accurately classify Korean patients with AD, as AD-vulnerable regions are larger in cognitively normal older Korean adults.29
Subsequent studies found that ethnicity-adjusted norms for brain volumes improved the diagnostic accuracy of AD.55 Nathoo et al.64 similarly highlighted substantial disparities in multiple sclerosis lesion burden and clinical outcomes, with poorer outcomes observed among African American and Latin American populations compared with non-Hispanic White individuals. The same study also reported increased lesion load and worse clinical outcomes among Latin American patients, further emphasizing health disparities across underrepresented populations. In contrast, Hedderich et al.65 found that normative brain volumes have a limited impact on diagnostic accuracy in neurodegenerative diseases. These variations underscore the need for population-sensitive neuroimaging models that account for biological and socioeconomic influences to improve diagnostic precision.
Integrating race/ethnicity and SES in AI models and their ethical considerations
Although integrating race and SES into AI models offers potential advantages, addressing issues of transparency and ethical governance is essential. Reinforcing racial essentialism and embedding bias in clinical AI systems – particularly through misinterpreting race as a biological determinant – may result in the reinforcement of racial stereotypes, breaches of privacy, and the exacerbation of healthcare inequities. It is crucial to distinguish biologically based variability from disparities arising from systemic inequities, such as structural racism, limited access to healthcare, and environmental exposures, in order to prevent the reinforcement of biological essentialism. Therefore, population-sensitive standards must be developed alongside appropriate safeguards, including the incorporation of socioeconomic variables, robust informed consent procedures, and continuous bias monitoring, to ensure equitable implementation in clinical practice. Given the critical role of SES in brain health, its integration as a core variable in neuroimaging datasets – using tools such as the HOUSES index, which measures individual socioeconomic status based on housing characteristics such as cost and crowding – is essential.66
To mitigate these risks, researchers and clinicians must ensure transparency in data collection methodologies, obtain explicit informed consent from participants, and implement robust ethical oversight mechanisms. Providing clear information about the populations included and their demographic characteristics can help clinicians make better-informed decisions when using AI-based neuroimaging tools. Artificial intelligence-based diagnostic tools should serve as supportive aids rather than substitutes for clinical judgment, ensuring that radiologists and neurologists retain primary responsibility for interpreting neuroimaging results within the broader clinical context (Figure 4).
Artificial intelligence-based neuroimaging platforms should incorporate a structured clinician feedback loop to ensure continuous improvement and ethical oversight. Clinicians should be able to flag suspected diagnostic discrepancies through a secure, anonymized interface. Expert reviewers should then validate these flagged cases, and confirmed discrepancies should inform model retraining pipelines. This iterative process would enhance model calibration, facilitate bias detection, and promote accountability in the deployment of clinical AI systems. Developers can implement broader ethical safeguards by conducting subgroup-specific performance audits, applying bias quantification metrics, and validating models using diverse population datasets. Explainable AI (XAI) techniques, such as SHAP (Shapley Additive Explanations) and LIME (Local Interpretable Model-Agnostic Explanations), can help clarify model decision-making processes, thereby reducing the “black box” effect and highlighting the influence of input features such as SES and race/ethnicity. Consent frameworks should clearly define data provenance, usage parameters, and the implications of algorithmic outputs to ensure transparency and participant autonomy. Finally, clinicians must receive targeted training to interpret AI predictions in the context of population metadata and social determinants of health, reinforcing their essential role in context-aware, multidisciplinary diagnostic workflows.
Limitations of the study
This scoping review, by design, does not include a critical appraisal of bias or a meta-analysis, which would provide the statistical precision and pooled effect estimates typically reported in systematic reviews. Nevertheless, adherence to the PRISMA-ScR framework mitigated selection bias by ensuring a structured approach to study identification, screening, and inclusion.42 As noted in the “Study heterogeneity” section, methodological inconsistencies in MRI protocols, software, and adjustments for confounding variables pose challenges to generalizability. Furthermore, a substantial proportion of the included studies employed cross-sectional designs, limiting the ability to establish causal relationships or assess longitudinal changes. Variations in sample size (ranging from 30 to 2,537 participants) also constrain generalizability. The present review primarily emphasized structural and socio-structural dimensions without extensively addressing genetic predispositions or environmental exposures, including air pollution, nutrition, and early-life stress.
Conclusions
This scoping review synthesizes evidence of population-sensitive variability in regional brain morphology across racial, ethnic, and socioeconomic groups in both health and disease. These differences have important implications for the interpretation of neuroimaging findings and the development of AI-based diagnostic models. The review highlights persistent gaps in demographic diversity within current neuroimaging datasets, particularly in commercial and research-based AI tools. Addressing these gaps requires studies that incorporate larger, socio-demographically diverse populations and account for structural determinants of health. Developing population-sensitive neuroimaging templates is critical for improving diagnostic accuracy and advancing healthcare equity; however, this effort must also consider practical challenges, including data harmonization, potential misclassification, and ethical risks such as the inadvertent reinforcement of racial bias. To navigate these complexities, coordinated efforts at national and international levels are necessary, alongside the rigorous implementation of ethical safeguards. These should include transparency in data provenance, the inclusion of socioeconomic variables, and interpretability mechanisms within AI tools. Explainable AI techniques can be incorporated to visualize how input features – including race, ethnicity, and socioeconomic indicators – contribute to diagnostic predictions. These methods help quantify feature attribution, promote transparency in model behavior, and enable clinicians to assess whether outputs are influenced by spurious or biased correlations. The proposed implementation approaches are consistent with emerging recommendations to integrate structural and functional neuroimaging features into personalized, context-aware diagnostic frameworks.7, 65 Population-sensitive standards and inclusive models may help reduce bias, misclassification, and inequities in AI-driven neuroimaging. Interpretation of intergroup brain differences must remain grounded in a socio-structural framework to prevent the misrepresentation of race as a biological essence. Crucially, group-level morphological differences should be interpreted within the broader context of social determinants of health rather than as biologically inherent traits, in order to avoid reinforcing essentialist narratives.
Recommendations
To address the clinical, ethical, and research challenges identified in this review, the following strategies are recommended:
– Fostering international partnerships and data-sharing initiatives to create large, representative neuroimaging datasets that capture population diversity. These datasets could be stratified by race/ethnicity, age, and sex to improve diagnostic precision and minimize bias in AI applications.
– Incorporating validated socioeconomic indicators (e.g., HOUSES index, education, housing quality) and ensuring subgroup performance auditing during model development.
– Employing explainability (XAI) methods, such as SHAP and LIME, to enhance transparency in algorithmic decision-making.
– Integrating population-sensitive neuroanatomical features (e.g., ethnicity-adjusted volumetrics, SES-modulated patterns) into AI models while contextualizing them as proxies for lived structural and social experiences rather than fixed biological traits. This approach safeguards against the misinterpretation of race as a biological essence and aligns diagnostic models with equity-focused precision medicine.
– Employing federated learning approaches to preserve data privacy while improving inclusivity across geographic and demographic populations.39
– Implementing ethical AI toolkits to detect and address algorithmic bias at all stages of model deployment.
– Establishing a structured feedback mechanism through which clinicians can report AI-generated diagnostic discrepancies, allowing for systematic monitoring, auditing, and model retraining. This mechanism should include a secure, anonymized reporting interface and a validation pipeline for incorporating clinical corrections into future model updates.
– Developing future models that include metadata fields capturing key social determinants of health.
Use of AI and AI-assisted technologies
Not applicable.