Abstract
Background. The “equilibrium theory” posits that the tongue and perioral muscles, including the orbicularis oris (OO) muscle, function synergistically to maintain balanced tooth positioning. Surface electromyography (sEMG) is a valuable, nonivasive method for assessing muscle activity. However, previous electromyographic (EMG) studies comparing lip muscle activity in children with and without lip competence (LC) have yielded inconsistent results. Therefore, further research is needed to clarify OO muscle activity patterns in this population.
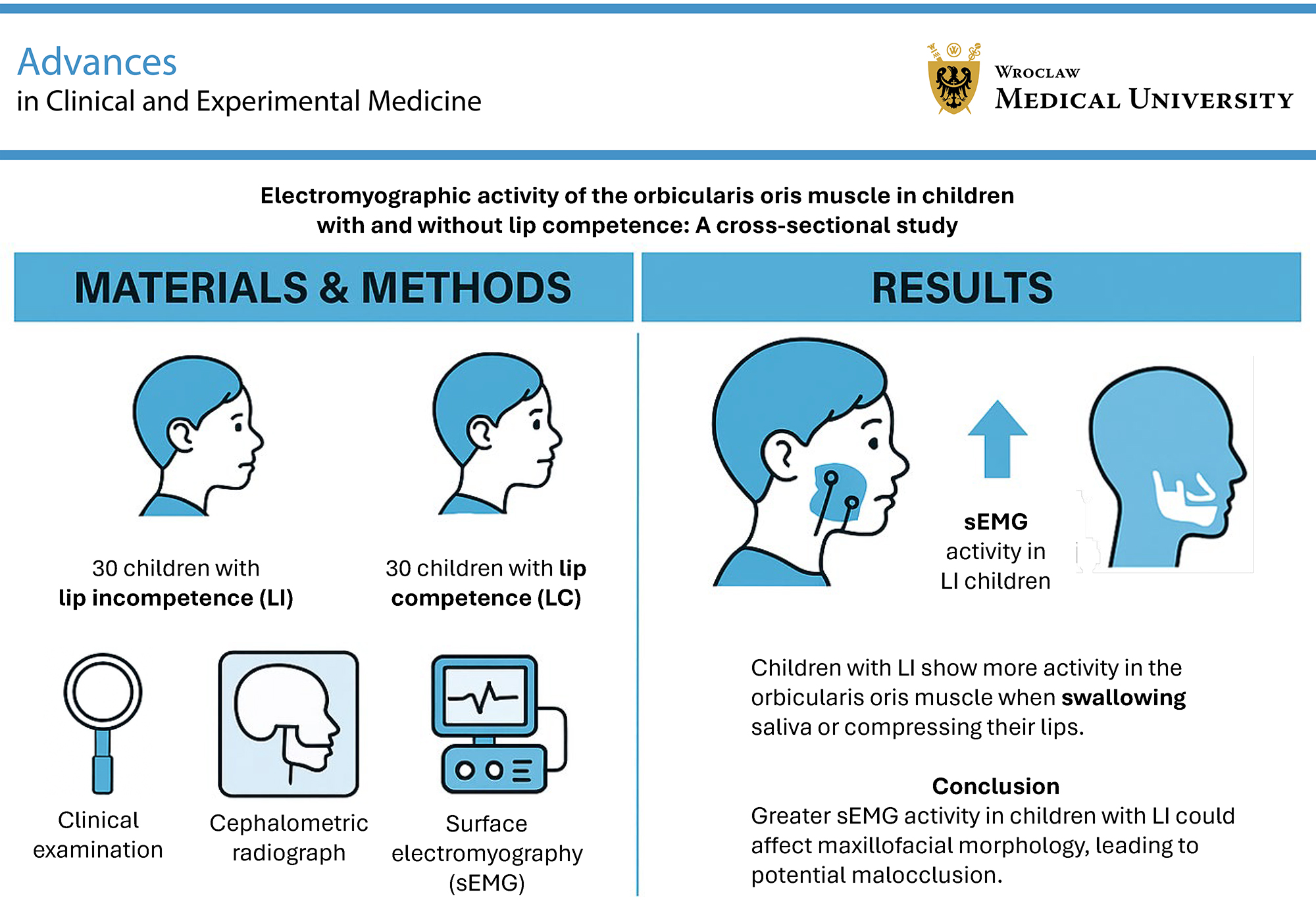
Objectives. The aim of the study was to evaluate the EMG activity of the superior (SOO) and inferior orbicularis oris (IOO) muscles in children with and without LC.
Materials and methods. The sample comprised 30 children with lip incompetence (LI) (mean age 9.46 ± 1.76 years) and 30 children with LC (mean age 8.85 ± 1.52 years). Electromyographic recordings of the SOO and IOO muscles were obtained using a DAB Bluetooth Instrument (Zebris Medical GmbH, Isny im Allgäu, Germany) at clinical rest, during saliva swallowing, lip protrusion (“kissing” position), lip compression, and while articulating the syllables /pa/, /ba/, and /ma/. Statistical analyses were performed using Stata v. 11.0 (StataCorp, College Station, USA). The level of significance was set at p < 0.05.
Results. Electromyographic activity of the SOO and IOO muscles during saliva swallowing (p < 0.001, adjusted p (padj) = 0.002) and lip compression (p = 0.001, padj = 0.013 for SOO; p < 0.001, padj = 0.005 for IOO) was significantly greater in children with LI compared to those with LC. Similar EMG activity at rest and during speech production was observed in children with and without LC.
Conclusions. Children with LI demonstrate increased SOO and IOO muscle activity during saliva swallowing and lip compression, suggesting greater muscular effort is required to achieve lip seal. This increased activity may disturb the muscular force balance essential for proper maxillofacial growth and could contribute to the development of malocclusion.
Key words: surface electromyography, craniofacial growth, lip competence, lip incompetence, incompetent lips
Background
Competent lips are essential for maintaining a balance between the muscles of the cheeks and the tongue. Lip competence (LC) is defined as the ability to maintain slight contact between the lips in a state of clinical rest (relaxed musculature). In contrast, the term incompetent lips (IL) describes anatomically short lips that are unable to maintain contact in a state of relaxed musculature. In such cases, lip seal requires active contraction of the orbicularis oris (OO) and mentalis muscles, clinically observed as chin wrinkling.1, 2, 3, 4, 5 Incompetent lips may result from anteroposterior and vertical disharmony of the dentofacial complex or may develop as an adaptation to mouth breathing due to nasal obstruction, respiratory difficulties, or other forms of altered oral function.5
The “equilibrium theory” proposes that the tongue and perioral muscles, including the OO, work together to maintain balanced tooth positioning.6, 7 Proper growth and development of the maxillofacial structures and stable occlusion depend on a balance of muscular forces. Maintaining healthy perioral soft tissue function is essential, as lip and tongue dysfunction may lead to malocclusion or orthodontic relapse.8, 9, 10, 11, 12, 13
Surface electromyography (sEMG) is a valuable tool for assessing muscle activity. This noninvasive technique measures electrical signals on the skin above superficial muscles, providing an objective, quantitative representation of neuromuscular balance in the stomatognathic system. Therefore, it is a highly useful instrument for studying the relationship between morphology and function in the oral-maxillofacial system.14, 15, 16, 17, 18, 19 The sEMG’s simplicity, nonivasiveness, and accessibility are important factors for studies involving children.20, 21, 22, 23
Currently, there is limited research on OO muscle EMG activity in patients with lip incompetence (LI). Moreover, existing studies vary with respect to study populations, methodologies (e.g., tasks assessed using sEMG), and results. Some studies have focused on EMG activity of the OO in children and adolescents.1, 2, 10, 24, 25, 26, 27 Some authors have reported EMG findings in patients with LI and malocclusion,1, 24, 25, 26, 27 while others did not assess occlusal characteristics in their study populations.2, 10 The results of previous studies vary considerably.
Some studies reported higher EMG activity of the OO during tasks such as swallowing,1, 2, 24, 25, 26 lip compression,24, 25, 26, 27 speech,2 chewing,2 puffing out the cheeks,10 and at rest5 in individuals with LI. Other investigations reported lower EMG activity of the inferior OO at rest and during swallowing in individuals with LI, or similar activity during speech and swallowing in the superior OO in children with and without IL.10 Because EMG studies of lip activity comparing children with CL and IL have reported contradictory results, and EMG patterns of the OO, particularly in children with LI, remain unclear, further research on lip EMG activity in this population is necessary.
Objectives
The objective of this study was to evaluate the EMG activity of the superior (SOO) and inferior (IOO) orbicularis oris muscles in children with and without IL. We hypothesized that there would be no difference in the EMG activity of the SOO and IOO muscles between children with and without LC during the studied tasks.
Materials and methods
This cross-sectional study was reported in accordance with the Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) Statement. The study adhered to the Declaration of Helsinki and protocol was approved by the Local Bioethics Committee of the Pomeranian Medical University in Szczecin, Poland (approval No. KB-0012/08/15). Written informed consent was obtained from the parents before the procedures.
Study population
The study included 30 children with IL (mean age: 9.46 ±1.76 years) and 30 children with CL (mean age: 8.85 ±1.52 years). The children were recruited over 2 months from a consecutive sample at an orthodontic clinic. They were selected in the order of arrival for screening, forming a convenience sample. During the clinical examination, each child sat upright without head support, looking straight ahead with the jaw relaxed. An orthodontist classified each child into either the competent lip group (CLG), defined as lips lightly touching without mentalis muscle contraction, or the incompetent lip group (ILG), defined as lips apart at rest or lips touching with visible mentalis muscle activity (chin shrinkage).2
The study included both boys and girls with mixed dentition, Class I occlusion, and positive overjet and overbite. Children had to be free of congenital disorders, behavioral problems, disabilities, environmental allergies, common colds, nasal obstruction, oral parafunctional habits (e.g., thumb or finger sucking, nail biting), restrictions of the labial or lingual frenulum (e.g., ankyloglossia), and atypical swallowing (AS). They must not have undergone previous orthodontic treatment, myofunctional or speech therapy, or trauma or surgery in the orofacial region. Children with systemic diseases affecting muscles, muscle disorders such as temporomandibular dysfunction or bruxism, or a history of neuromuscular disease were excluded. Participants must not have been taking any medications that might affect muscle activity. Only children for whom voluntary parental consent was obtained were included. Children who did not meet these criteria were excluded from the study.
Sample size
Each study group was planned to include 30 participants. The sample size was determined using a two-sided t-test with a significance level of 0.05 and a power of 80% (1 − β = 0.20). The effect size, defined as large (Cohen’s d = 0.80), was determined based on pilot data and prior research.2, 10, 25 The calculation was performed using the pwr v. 1.3.0 and report v. 0.5.7 packages in R v. 4.3.1 (R Foundation for Statistical Computing, Vienna, Austria), running on Windows 10 x64 (Microsoft Corp., Redmond, USA).28, 29 An additional 15% correction was applied to account for deviations from a normal distribution.
Experimental procedures
An experienced orthodontist determined whether each participant had CL or IL during the clinical examination. The examination also included an assessment of breathing mode (via a parental questionnaire), mouth posture (observed by the examiner), ear, nose and throat conditions, and speech and language. Perioral muscles were visually assessed to diagnose AS, defined as the tongue pressing against the dental arches or being positioned anteriorly or laterally during swallowing and accompanied by myofunctional alterations, such as hyperactivation of the lips, facial muscle tension, or abnormal head and mandible movements. An intraoral analysis evaluated occlusal features, including vertical overlap, overjet, Angle Class, crossbite, and open bite. Body mass index (BMI) was calculated for each participant using weight [kg] and height [m].
To assess the correlation between LC and dentofacial morphology, cephalometric radiographs routinely obtained during the diagnostic process were analyzed. All cephalometric radiographs were acquired using a digital X-ray device (Cranex Tom; Soredex, Tuusula, Finland). The measurements were evaluated by the same orthodontist using cephalometric analysis software (Orthodontics 7.0; Orto-Bajt, Wrocław, Poland) and were based on the Segner and Hasund method.30
Surface electromyography
The EMG activity of the SOO and IOO muscles was simultaneously measured using a DAB Bluetooth electromyography device (Zebris Medical GmbH, Isny im Allgäu, Germany) with a gain of 1000, a high-pass filter of 7–5 kHz, a sampling rate of 1 kHz, and a 12-bit analog-to-digital converter. The system was calibrated before each recording. Bipolar Ag/AgCl surface electrodes (Noraxon Dual Electrode; Noraxon, Scottsdale, USA) with a 20-mm inter-electrode distance were placed on the SOO and IOO muscles. Electromyographic procedures followed established protocols.24, 25, 26 During the recording, participants sat in a dental chair with their heads in a natural position. Electrodes were positioned as follows: SOO muscle – along the line from the lip corner to the subnasal point; IOO muscle – along the line from the lip corner to the chin midline; reference electrode – behind the right ear. A single trained and experienced examiner, blinded to the clinical findings, positioned the electrodes to ensure consistent placement relative to muscle fiber orientation.
To prepare for EMG recordings, each participant’s skin was gently cleaned with a 70% ethyl alcohol solution and dried using disposable cotton to reduce impedance. Slight skin reddening following cleaning was considered an indicator of adequate preparation. After electrode placement, an impedance test was conducted using a Metex P-10 measuring device (Metex Instruments Corporation, Seoul, South Korea). Surface EMG recordings of the SOO and IOO muscles commenced 5 min later.
The EMG recordings of the SOO and IOO muscles were obtained while participants performed a series of tasks: 1) resting with relaxed lips; 2) swallowing saliva; 3) pursing the lips into a “kissing” position; 4) compressing the lips together; and 5) pronouncing the syllables /pa/, /ba/, and /ma/. Before the EMG recordings, the examiner explained each task to ensure proper execution. Participants were provided with instructions and asked to practice the tasks by imitating the examiner. They were instructed to: 1) relax the jaw; 2) swallow saliva naturally (as visually confirmed by hyoid bone movement); 3) pucker the lips into a “kissing” position; 4) press the lips together firmly; and 5) articulate the syllables /pa/, /ba/, and /ma/.
Following established protocols, each movement was repeated at least 3 times.24, 25, 26 For each participant, the final EMG value represented the average of the last 2 recordings, as the initial measurement often deviated substantially and was discarded as a “learning” trial.31 To prevent muscle fatigue, participants rested for approx. 1 min between tasks. Electrode signals were amplified, digitized, and stored on a computer. Surface EMG data were subsequently normalized to the peak EMG value for each task. Among various normalization protocols, some authors recommend using the peak EMG value obtained during dynamic contraction as the reference value, as this approach helps reduce inter-subject variability.32
The task with the highest potential served as the maximum reference point (taken as 100% OO muscle activity). Lip protrusion (pursing lips into a “kissing” position) showed significantly higher EMG activity than all other tasks and was used as the reference for normalization. Measured values were expressed as a percentage of the lip protrusion reference value using the formula: mean task activity (µV)/mean lip protrusion activity (µV) × 100%. This resulted in normalized EMG values [µV/µV%] for all measurements.24, 25, 26, 33, 34
The repeatability of the EMG protocol was assessed by performing duplicate measurements in 20 randomly selected children. The same examiner repeated the recordings after a 15-min interval under identical experimental conditions. The comparison of the 1st and 2nd measurements demonstrated good intra-examiner repeatability.
Statistical analyses
All statistical analyses were performed using Stata v. 11.0 (StataCorp, College Station, USA). The level of statistical significance was set at p < 0.05. To assess differences in EMG activity of the SOO and IOO muscles between the ILG and CLG across 6 functional tasks (rest, lip compression, swallowing saliva, lip protrusion, and articulation of /pa/, /ba/, and /ma/), a total of 12 primary comparisons were conducted (6 per muscle). To reduce the risk of type I error due to multiple testing, the Holm–Bonferroni correction was applied, maintaining the family-wise error rate at α = 0.05.
This method ranks the unadjusted p-values from smallest (rank 1) to largest (rank 12) and compares each to a sequentially adjusted threshold according to the following equation:
P-value (unadjusted) ≤ Thresholdk (1),
where
Thresholdk = α/(m − k + 1) (1);
m – total number of comparisons (12);
k – rank of the unadjusted p-value (1 to 12).
An unadjusted p-value is considered statistically significant if it is less than or equal to its corresponding threshold. Because only 2 groups were compared, omnibus tests were not required. The Holm–Bonferroni correction was therefore applied directly to the 12 predefined pairwise comparisons constituting the primary endpoints. Exploratory subgroup analyses (by sex) were performed without p-value adjustment to preserve statistical power for hypothesis generation. However, these secondary analyses should be interpreted with caution due to the increased risk of type I error.35
Normality of distribution was assessed separately for each group using the Shapiro–Wilk test. For variables demonstrating normal distribution in both groups, homogeneity of variances was evaluated using Levene’s test (Supplementary Tables 1–3 present the tests of distributional assumptions for variables included in Table 1, Table 2, Table 3, respectively). Continuous variables meeting both normality and homogeneity of variance assumptions were analyzed using the independent-samples Student’s t-test. In cases of unequal variances, Welch’s t-test was applied. When normality was violated in at least 1 group, continuous variables were analyzed using the Mann–Whitney U test.
Categorical variables were compared using Pearson’s χ2 test (Supplementary Table 4 presents the observed and expected frequencies, along with the results of Pearson’s χ2 test for the association between gender and LC). The paired Student’s t-test was used to analyze the repeatability of the EMG procedure, as the derived data were normally distributed. Continuous variables were reported as mean ± standard deviation (SD) for normally distributed data or as median with interquartile range (IQR, 25th–75th percentiles) for non-normally distributed data. Categorical variables were presented as counts and percentages (%).
Results
A total of 60 individuals participated in the study. The ILG comprised 30 children (15 girls (50%) and 15 boys (50%) mean age: 9.46 ±1.76 years). The CLG included 30 subjects (17 girls (56.7%) and 13 boys (43.3%), mean age: 8.85 ±1.52 years). Table 1 presents the characteristics of the participants. The groups showed statistically significant differences in average overjet, ANB angle, Sp’–Gn distance, and Index (p < 0.001); however, no significant differences were found for the other analyzed variables.
The 1st and 2nd EMG examinations revealed no statistically significant differences in SOO and IOO activity. Children with IL exhibited significantly higher SOO and IOO muscle activity during swallowing compared to those with CL (p < 0.001, Holm–Bonferroni-adjusted padj = 0.002) (Table 2, Table 3; Figure 1). Similarly, EMG activity of both the SOO and IOO muscles was significantly higher in the ILG during lip compression (p = 0.001, padj = 0.013 for SOO and p < 0.001, padj = 0.005 for IOO) (Table 2, Table 3; Figure 2).
Resting SOO and IOO activity levels were similar in both groups (p = 0.606, padj > 0.999 for SOO and p = 0.670, padj > 0.999 for IOO) (Table 2, Table 3; Figure 3). No statistically significant differences were found in SOO and IOO muscle activity during the production of bilabial sounds (/pa/, /ba/, /ma/) between the ILG and CLG (/pa/ p = 0.706, /ba/ p = 0.464, /ma/ p = 0.900, padj > 0.999 for SOO and /pa/ p = 0.412, /ba/ p = 0.442, /ma/ p = 0.882, padj > 0.999 for IOO) (Table 2, Table 3; Figure 4, Figure 5, Figure 6). No statistically significant differences in SOO and IOO muscle activity during any of the tasks were observed between boys and girls in either group (Table 2, Table 3).
Discussion
This study compared the electrical activity of the OO muscle in children with IL to that of children with CL using surface electrodes. Repetition of the procedure confirmed the reliability of electrode placement and the study protocol. Higher activity of the SOO and IOO muscles was observed in children with IL during swallowing and lip compression, suggesting increased effort required to achieve lip seal. The significantly higher activity during swallowing may be particularly important due to the frequency of this action (600–3,000 times per day), potentially affecting maxillofacial morphology and occlusal stability. Previous research has demonstrated increased EMG activity of the OO muscle in individuals with LI compared to those with normal lip seal.1, 2, 24, 25, 26, 27 However, the findings remain inconsistent, likely due to heterogeneity in study populations and methodological differences. Our results, showing higher OO muscle activity during swallowing in children with LI, are consistent with the observations of Gustafsson and Ahlgren1 and Gamboa et al.,2 although the latter study included an older cohort (17–27 years of age).
In contrast, Lipari et al.10 reported similar SOO muscle activity in both groups and lower IOO activity during swallowing in children with IL aged 7–13 years. They proposed that children with IL have hypotonic IOO muscles and require activation of the mentalis muscle to achieve lip seal.10 Consistent with our findings, Tosello et al.27 observed significantly higher activity in the SOO, IOO, and mentalis muscles during lip compression in children with IL, suggesting muscle hyperactivity.
In the present study, similar OO muscle activity at rest was observed in children with and without IL, consistent with the findings of Gamboa et al.2 However, this result contrasts with the findings of Tomiyama et al.,5 who reported higher resting EMG activity in adults with IL. Lipari et al.10 observed lower resting activity in children with IL. Orbicularis oris muscle activity during speech was similar in both groups, aligning with the results of Lipari et al.10 This suggests that speech function in the OO muscle is not affected by LC, potentially due to compensatory mentalis muscle activity.36 However, this differs from Gamboa et al.,2 who found higher SOO activity during speech in individuals with IL.
When interpreting the study results, it is important to consider that various factors may influence the findings. Previous research has suggested that EMG activity in the SOO muscle is elevated in individuals with LI, as well as in those with anterior open bite and increased overjet.37 In the present study, no study participants with open bite were included, and no significant difference in mean overbite was observed between the groups. Although overjet was significantly greater in the ILG than in the CLG, no significant differences in OO muscle EMG activity at rest were observed between the groups, suggesting that overjet does not significantly affect this parameter.
Moreover, it should be noted that the study sample was controlled for malocclusion. Only participants with Class I occlusion and positive overjet and overbite were included. However, significant differences in cephalometric characteristics were observed, including a larger ANB angle, increased Sp’–Gn distance, and a reduced Index (indicating greater lower anterior facial height) in the ILG. These findings suggest that, independent of occlusal conditions, dentofacial morphology may be associated with LI, which is consistent with previous studies.38 Another factor that could affect OO muscle activity is AS. Some authors39, 40 have reported higher EMG activity of the OO muscle in patients with AS and LI compared to subjects with LC and controls, suggesting greater effort during swallowing.
In our study, however, patients with clearly identifiable AS were excluded. Nevertheless, a few borderline cases were observed in the ILG, as these children exhibited some features of an infantile swallowing pattern. Many other factors, such as disabilities, oral parafunctional habits, restrictions of the labial or lingual frenulum, as well as a history of frenotomy or speech and myofunctional therapy, may also affect muscle activity.25, 41, 42, 43, 44, 45 It has been reported that higher resting activity of the perioral muscles is observed among thumb and pacifier suckers,41 as well as in individuals with disabilities (e.g., Down syndrome and orally educated deaf individuals), during swallowing or speech.25, 42 Similarly, the presence of ankyloglossia may affect the resting position of the tongue and lips.43 In light of the above, children with these characteristics were excluded from the study sample. It should also be noted that individuals with nasal obstruction or other airway-related conditions were excluded. However, some of these conditions may have been underreported, as the assessment was based on clinical examination and a parent-completed questionnaire.
The present study utilized sEMG to measure muscle activity. This method is particularly suitable for use in children, as it does not involve needles or other invasive procedures. However, sEMG recordings may be influenced by factors such as inconsistent electrical conductivity and difficulty in distinguishing signals from different muscle fibers.15 Crosstalk, defined as the recording of signals over a muscle that originate from adjacent muscles, represents a significant source of error in sEMG and may lead to overestimation of muscle activity. Several factors influence the extent of crosstalk, including the inter-electrode distance of the EMG sensor and the accuracy of sensor placement on the muscle surface.46, 47
The accuracy of sEMG recordings also depends on factors related to the psychological and physiological state of the participants, as well as the equipment used.14, 15 To address these challenges, a consistent inter-electrode distance was maintained, electrodes were carefully placed at the center of the muscle belly, and the data were analyzed using appropriate quantitative methods, including a normalization procedure, to enhance accuracy. Another limitation of sEMG is its sensitivity to impedance imbalance.
To minimize impedance, the skin was thoroughly cleaned with 70% ethyl alcohol, a method commonly employed in sEMG studies involving children.24, 25, 26, 45 The children included in this study were well accustomed to the procedure, and the use of alcohol did not cause discomfort or elicit abnormal muscle activity.
In summary, it is important for clinicians to recognize that increased muscle activity in patients with LI may eventually influence dental arch form and facial morphology. Furthermore, stable tooth positioning relies on a balance among the oral, maxillofacial, and respiratory systems. Therefore, the therapeutic goal should be to establish normal jaw posture, nasal breathing, and proper lip closure.2, 48, 49
The diagnosis of LC is a critical element of orthodontic treatment planning. In this context, sEMG may serve as a valuable noninvasive tool to support accurate diagnosis. A precise analysis of sEMG recordings of the OO muscles is essential for refining treatment protocols aimed at achieving orofacial muscular balance. In this regard, the present study highlights the importance of early diagnosis of LC and timely intervention strategies, as functional lip alterations may occur even in the absence of overt malocclusion.
If left untreated, these functional disturbances may lead to more complex orthodontic, periodontal, or esthetic complications over time. Therefore, knowledge of OO muscle activity in growing individuals with LI may be particularly valuable for clinicians involved in the multidisciplinary management of such cases.10, 25, 50, 51, 52, 53
For growing patients diagnosed with LI, the combined use of myofunctional therapy and interceptive orthodontics – alongside conventional orthodontic treatment – is fundamental to establishing new neuromuscular patterns and restoring harmony within the stomatognathic system.45, 49, 54, 55 Surface electromyography may serve as a valuable tool for monitoring the progression and therapeutic outcomes of these interventions.45
Limitations of the study
This study has several limitations that should be acknowledged. We recognize that regulation of OO muscle activity is multifactorial and may be differentially influenced by occlusal conditions, oral habits, soft tissue morphology, and vertical and sagittal facial dimensions.13, 56 One limitation of the present study is the lack of detailed analysis of soft tissue morphology, such as upper lip length. In addition, the diagnosis of LI was based on clinical observation – specifically, the presence of lip separation at clinical rest or lip contact achieved through visible contraction of the mentalis muscle in the mandibular rest position (chin shrinkage). It should be noted that subtle contraction of the orofacial muscles may go unnoticed during visual assessment.4 Another limitation of this study is the lack of subdivision of the ILG into more specific subgroups, which could have allowed for a more detailed characterization of functional differences within this population. Furthermore, the cross-sectional design provides data collected at a single point in time and does not allow for conclusions regarding causality or longitudinal changes. Therefore, considering the aforementioned limitations, further well-designed, long-term studies conducted on larger sample sizes are required to confirm these findings and to better understand their potential impact on maxillofacial morphology.
Conclusions
Despite its limitations, the findings of this study indicate that children with LI exhibit increased EMG activity of the SOO and IOO muscles during saliva swallowing and lip compression. This may reflect the need for greater muscular effort to achieve adequate lip seal. Such increased muscular activity could potentially disturb the equilibrium of perioral forces required for proper maxillofacial growth and development, thereby contributing to the risk of malocclusion. However, longitudinal studies are necessary to confirm these observations and to clarify their long-term clinical implications.
Supplementary files
The supplementary files are available at https://doi.org/10.5281/zenodo.15807698. The package contains the following files:
Supplementary Table 1. Tests of distributional assumptions for variables in Table 1.
Supplementary Table 2. Tests of distributional assumptions for variables in Table 2.
Supplementary Table 3. Tests of distributional assumptions for variables in Table 3.
Supplementary Table 4. Observed and expected frequencies with Pearson’s χ2 test results for gender and LC association.
Data Availability Statement
The datasets supporting the findings of the current study are openly available in Zenodo repository at https://doi.org/10.5281/zenodo.16093588.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.

















.jpg)
.jpg)
.jpg)
.jpg)
.jpg)
.jpg)