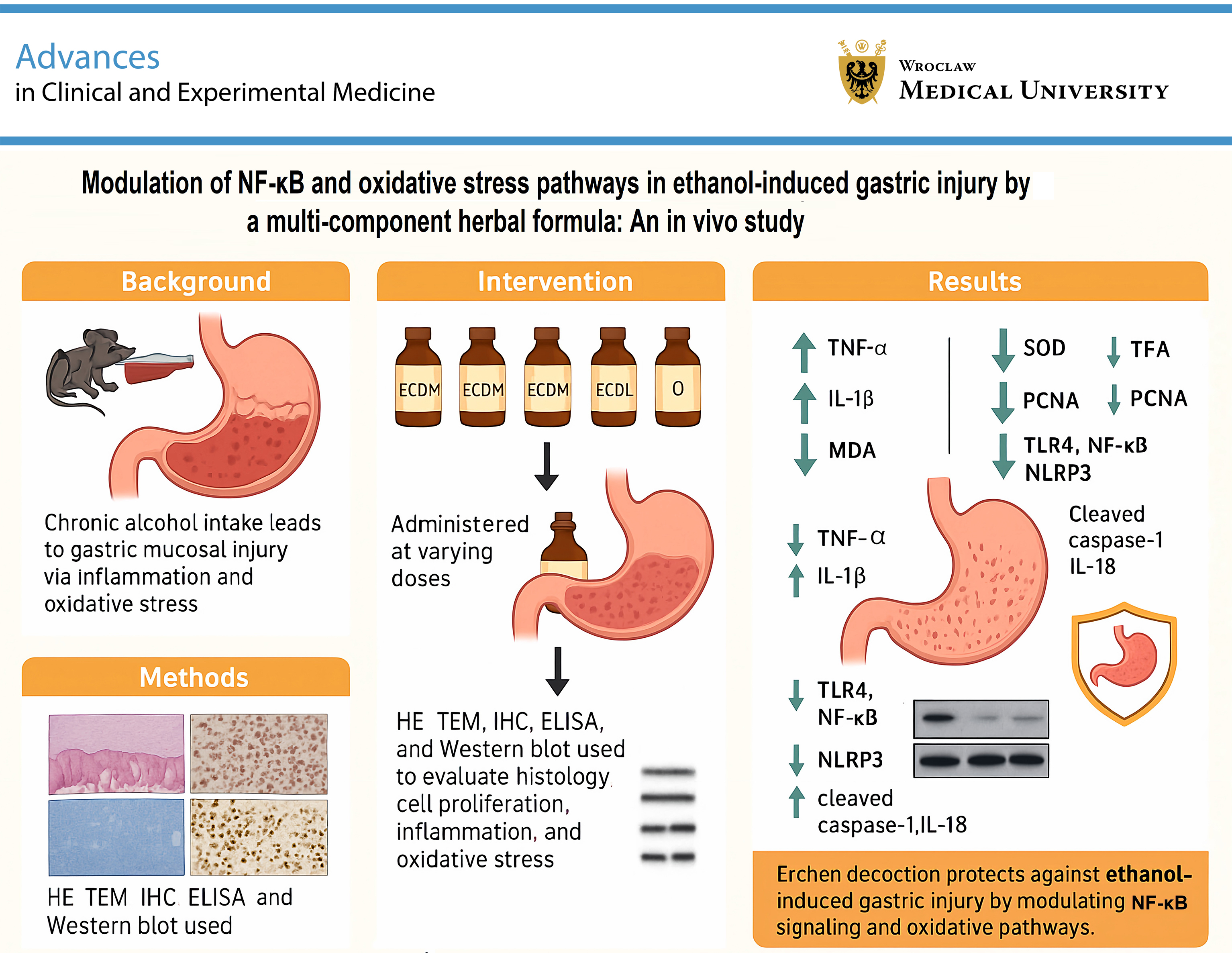
Abstract
Background. Chronic alcohol consumption can damage the gastric mucosa and increase the risk of severe gastrointestinal disorders, including gastritis, peptic ulcers, and potentially gastric cancer. Modern research has demonstrated that Erchen decoction possesses anti-inflammatory, antioxidant, and gastric mucosal protective properties and has been applied in the treatment of various inflammatory conditions.
Objectives. This study aimed to investigate the effects of Erchen decoction on chronic ethanol-induced gastric injury.
Materials and methods. Seventy-two male Sprague Dawley rats were randomly assigned to the following groups: Con group (control), Mod group (model), OXY group (oxymatrine, 5 mL/kg), ECDH group (high-dose Erchen decoction, 10 mL/kg), ECDM group (medium-dose Erchen decoction, 5 mL/kg), and ECDL group (low-dose Erchen decoction, 2.5 mL/kg). Gastric histopathological changes were evaluated using hematoxylin and eosin (H&E) staining and transmission electron microscopy (TEM). Proliferation of gastric mucosal cells was assessed using immunohistochemical detection of proliferating cell nuclear antigen (PCNA). Serum levels of tumor necrosis factor alpha (TNF-α), interleukin-1 beta (IL-1β), superoxide dismutase (SOD), and malondialdehyde (MDA) were measured using enzyme-linked immunosorbent assay (ELISA) and biochemical analysis. Western blot (WB) was used to analyze protein expression associated with inflammatory pathway activation.
Results. Compared with the Mod group, Erchen decoction significantly alleviated gastric injury and increased PCNA expression in the gastric mucosa. Its therapeutic efficacy decreased in a dose-dependent manner, with weaker effects observed at lower doses. Erchen decoction significantly reduced serum levels of TNF-α, IL-1β, and MDA while enhancing SOD activity; however, these effects were less pronounced in the ECDL group than in the ECDM and ECDH groups. Importantly, Erchen decoction markedly decreased the expression of toll-like receptor 4 (TLR4), nuclear factor kappa-B (NF-κB), NOD-like receptor protein 3 (NLRP3), apoptosis-associated speck-like protein (ASC), cleaved caspase-1, IL-1β, and interleukin-18 (IL-18).
Conclusions. Erchen decoction alleviates ethanol-induced gastric injury by reducing inflammation, attenuating oxidative stress, preserving cellular structural integrity, and regulating the NF-κB signaling pathway.
Key words: ethanol-induced gastric injury, gastric mucosal protection, NF-κB signaling pathway, oxidative stress, TLR4/NLRP3 inflammasome
Background
Chronic ethanol-induced gastric injury is a common digestive system disorder. Chronic alcohol consumption can damage the gastric mucosa and increase the risk of developing severe gastrointestinal conditions such as gastritis, peptic ulcers, and potentially gastric cancer.1, 2, 3 Alcohol exerts a direct effect on the gastric mucosa, leading to oxidative stress and inflammation, which can impair its protective function and contribute to tissue damage. In addition, the inflammatory process is mediated by interactions among multiple cytokines and signaling pathways.4, 5, 6 Among these, the nuclear factor kappa-B (NF-κB) pathway plays a central role in regulating the inflammatory response.7, 8, 9 Nuclear factor kappa-B is a transcription factor with a wide range of biological functions and regulates the expression of numerous inflammation-related genes. Under physiological conditions, NF-κB remains inactive in the cytoplasm.10, 11, 12 External stimuli, including bacterial or viral infections and inflammatory signals, can activate NF-κB, promoting its translocation to the nucleus and the subsequent initiation of the inflammatory response.13, 14, 15
Despite advancements in conventional therapies such as proton pump inhibitors (e.g., omeprazole) and histamine 2 (H2) receptor antagonists, their prolonged use is associated with adverse effects, including gastrointestinal disturbances, and incomplete efficacy in mitigating ethanol-related mucosal injury.16 Therefore, it is important to explore novel therapeutic approaches for alleviating chronic alcohol-induced gastric injury.
Erchen decoction, a classic traditional Chinese medicine (TCM) formula, consists of Pinelliae rhizoma, Citri exocarpium rubrum, Poria, and Zingiberis rhizoma recens, and contains 123 active components, including baicalein, stigmasterol, quercetin, and β-sitosterol.17 Erchen decoction has traditionally been used to treat headaches and chest tightness attributed to exogenous wind-cold, internal injury, and dampness retention. It has also been widely used in the management of nonalcoholic fatty liver disease (NAFLD) and chronic obstructive pulmonary disease.18, 19 The therapeutic application of Erchen decoction in various inflammatory diseases is largely attributed to its anti-inflammatory and antioxidant properties.17, 20 In recent years, with the increasing focus on the modernization of TCM, the potential therapeutic effects of Erchen decoction in chronic alcohol-induced gastric injury have attracted growing attention. Therefore, this study was undertaken to investigate the therapeutic effects of Erchen decoction in a rat model of chronic ethanol-induced gastric injury and to further explore its underlying mechanisms, thereby addressing existing mechanistic gaps in the current research on its effects in chronic gastric injury.
Advances in microscopy, particularly transmission electron microscopy (TEM), have revolutionized the morphological sciences by providing unprecedented levels of magnification and resolution for the examination of biological samples. Transmission electron microscopy enables detailed visualization of the ultrastructure and organization of cellular components, including the cytoskeleton, membrane systems, and organelles, thereby elucidating structural–functional relationships within cells and tissues.21 In the context of chronic alcohol-induced gastric injury, TEM has proven to be an invaluable tool for elucidating pathological alterations in gastric mucosal cells, including mitochondrial damage, endoplasmic reticulum stress, and disruption of cellular junctions. These ultrastructural changes are critical for understanding the underlying mechanisms of alcohol-induced gastric injury. Therefore, in this study, hematoxylin and eosin (H&E) staining, immunohistochemistry (IHC), TEM, biochemical assays, and western blot (WB) analysis were used to comprehensively evaluate and characterize the therapeutic effects and underlying mechanisms of Erchen decoction in alcohol-induced gastric injury.
Objectives
The study aims to investigate the effects of Erchen decoction on chronic ethanol-induced gastric injury.
Materials and methods
Animals
Seventy-two healthy male Sprague Dawley rats, aged 8 weeks and weighing approx. 170 g, were obtained from Henan Skbers Biotechnology Co., Ltd. (Zhengzhou, China). The rats were housed in a controlled environment at a room temperature of 20–24°C and relative humidity of 60–80%, with free access to food and water. The standard rat chow was purchased from Henan Huanyu Hekang Biotechnology Co., Ltd. (Zhengzhou, China). It consisted primarily of corn, soybean meal, bran meal, fish meal, macronutrients (vitamins), micronutrients (amino acids), mineral supplements (e.g., ferrous sulfate), edible oil, and salt (0.5% each). This study received ethical approval (No. GENINK-20230029; issued on October 8, 2023) from Tianjin Jinke Bona Biotechnology Co., Ltd. (Tianjin, China). All experiments were conducted in accordance with the Chinese Government’s Guidelines for the Care and Use of Laboratory Animals to minimize animal pain and discomfort.22
Laboratory reagent
Erchen decoction (Q/BJT 0002S-2020; Pinelliae rhizoma 15 g, Citri exocarpium rubrum 15 g, Poria 9 g, Glycyrrhizae radix et rhizoma praeparata cum melle 4.5 g, 7 pieces of Zingiberis rhizoma recens, 1 Mume fructus; Bozhou Bojiatang Biotechnology Co., Ltd., Bozhou, China); oxymatrine (OXY) solution (1ST40311; Tianjin Alta Technology Co., Ltd., Tianjin, China); H&E staining solution (BA4041–BA4024; Zhuhai BaSO Biotechnology Co., Ltd., Zhuhai, China); anti-proliferating cell nuclear antigen (PCNA) antibody (AF0261; Shanghai Beyotime Biotech Inc., Shanghai, China); serum biochemical factor enzyme-linked immunosorbent assay (ELISA) kits (A001-3-2, A003-1-2; Nanjing Jiancheng Bioengineering Institute, Nanjing, China); Bradford protein quantification kit (P0006; Shanghai Beyotime Biotech Inc.); anti-interleukin-18 (IL-18) antibody (1:2,000; 10663-1-AP; Proteintech, Rosemont, USA); anti-interleukin-1 beta (IL-1β) antibody (1:2,000; 16806-1-AP; Proteintech); anti-caspase-1 antibody (1:1,000; 24232S; Cell Signaling Technology (CST), Danvers, USA); anti-cleaved caspase-1 antibody (1:1,000; 89332S; CST); anti-toll-like receptor 4 (TLR4) antibody (1:1,000; 66350-1-Ig; Proteintech); anti-NF-κB antibody (1:1,000; 10745-1-AP; Proteintech); anti-NOD-like receptor pyrin domain-containing protein 3 (NLRP3) antibody (1:2,000; 68102-1-Ig; Proteintech); and anti-apoptosis-associated speck-like protein containing a CARD (ASC) monoclonal antibody (1:5,000; 10500-1-AP; Proteintech).
Grouping
As shown in Table 1, the study design involved randomly dividing 72 rats into 6 groups: a control group (Con group; n = 12), a model group (Mod group; n = 12), a high-dose Erchen decoction group (ECDH group; n = 12), a medium-dose Erchen decoction group (ECDM group; n = 12), a low-dose Erchen decoction group (ECDL group; n = 12), and an oxymatrine group (OXY group; n = 12). After a 1-week acclimatization period, the Con group received 8 g·kg–1 normal saline. Administration was performed 3 times daily for 12 weeks. Other groups received 8 g·kg–1 ethanol by gavage for 12 weeks.23
The Erchen decoction groups and the OXY group were treated daily during the final 4 weeks of ethanol exposure (weeks 9–12). The ECDH group received 10 mL·kg–1 Erchen decoction, the ECDM group received 5 mL·kg–1 Erchen decoction, and the ECDL group received 2.5 mL·kg–1 Erchen decoction. The OXY group received 5 mL·kg–1 OXY solution daily. The above dosages were calculated based on the adult daily dose (1 dose per day, 200 g per dose) and converted to the equivalent rat dose using a standard human body weight of 70 kg.24 No significant toxicity of Erchen decoction has been reported in previous studies.25, 26 According to common clinical dosages and relevant literature, the dosage used in this study complies with established safety standards for TCM.4 Animals were fasted overnight for 15 h on the final day of the experiment prior to anesthesia induction and blood sample collection.
Hematoxylin and eosin staining analysis
Rat gastric histopathology was assessed using H&E staining. To preserve tissue structure, gastric tissue samples were immersed in 4% paraformaldehyde for 24 h. After fixation, the samples were dehydrated through a graded ethanol series (70–100%) for approx. 1 h, followed by clearing in xylene. Following dehydration and clearing, the samples were embedded in paraffin and sectioned at a thickness of 4–5 μm. The sections were then dewaxed in xylene, rehydrated through decreasing concentrations of ethanol, and stained with H&E solutions for 3–5 min and 1–2 min, respectively. After staining, the sections were dehydrated again, cleared in xylene, and mounted with sealing medium and a coverslip. Pathological changes, including edema, hemorrhage, epithelial shedding, and inflammatory cell infiltration, were evaluated using light microscopy (model DP26; Olympus Corp., Tokyo, Japan).
Immunohistochemistry analysis
Gastric tissue samples were immersed in 4% formaldehyde for 24 h, dehydrated through a graded series of ethanol solutions, and subsequently embedded in paraffin. The paraffin blocks were sectioned at a thickness of 4–5 μm and mounted onto glass slides. The sections were dewaxed in xylene and rehydrated through decreasing concentrations of ethanol. For antigen retrieval, the sections were heated under high pressure in citrate buffer for 20 min.
The samples were subsequently incubated in a blocking solution containing 5% normal serum and 1% bovine serum albumin (BSA) for 30 min to reduce nonspecific binding. The sections were then incubated overnight at 4°C with a primary antibody against proliferating cell nuclear antigen (PCNA). After washing, the sections were treated with a secondary antibody for 1 h. The chromogenic reaction between the enzyme-labeled secondary antibody and its substrate (typically 5–10 min) was used to visualize antibody binding. Subsequently, hematoxylin was used for nuclear counterstaining, followed by clearing in xylene, and the sections were mounted with a coverslip. PCNA expression in the nuclei was observed and quantitatively analyzed under a light microscope (model DP26; Olympus Corp.) to assess cell proliferation in gastric tissue.
Transmission electron microscopy observation
Gastric tissue was excised and cut into 1 mm3 pieces, immediately fixed in 2.5% glutaraldehyde followed by 1% osmium tetroxide for 2 h, and then treated with 1% tannic acid for 30 min. The samples were subsequently dehydrated through a graded ethanol series (50–100%), with each step lasting 15 min.
After dehydration, the tissue was infiltrated with acrylic resin by incubation in a mixture of resin and ethanol for 2 h, followed by pure resin overnight, and then subjected to thermal polymerization and embedding in a mold. The cured resin blocks were subsequently sectioned into ultrathin sections (60–90 nm), mounted on copper grids, and stained with lead uranyl acetate for 10 min to enhance contrast. The sections were then placed in the TEM sample holder, and images were acquired for quantitative and qualitative analysis (model HT7800; Hitachi Ltd., Tokyo, Japan).
Serum biochemical detection
The manufacturer’s instructions were followed for reagent preparation and microplate handling using the premade ELISA kits. Diluted serum samples and standards were added to the microplate wells, incubated for 1–2 h, and then washed with wash buffer. An enzyme-conjugated secondary antibody was added and incubated, followed by washing and the addition of substrate solution. After 15–30 min, a stop solution was added to terminate the reaction. Absorbance was measured at 450 nm to determine the concentrations of inflammatory markers.
For the determination of superoxide dismutase (SOD) activity and malondialdehyde (MDA) levels, serum samples were added to the prepared reaction mixtures, and absorbance or fluorescence intensity was measured based on the ability of SOD to inhibit substrate oxidation and on the formation of the colored product generated by the reaction of MDA with thiobarbituric acid. Finally, SOD activity and MDA concentrations were calculated by comparison with the standard curve.
Western blot analysis
Sample proteins were extracted using radioimmunoprecipitation assay (RIPA) buffer supplemented with protease inhibitors, followed by centrifugation at 4°C for 10 min to remove insoluble material. A Bradford protein quantification kit (P0006; Shanghai Beyotime Biotech Inc.) was used to determine protein concentration and ensure equal loading onto the sodium dodecyl sulfate–polyacrylamide gel electrophoresis (SDS–PAGE) gel.
The samples were mixed with 5 volumes of loading buffer, boiled for 5 min to achieve denaturation, and then separated by electrophoresis on a 10% SDS–PAGE gel. The separated proteins were transferred onto a polyvinylidene difluoride (PVDF) or nitrocellulose membrane, followed by wet transfer at 100 V for 1–2 h. After blocking in Tris-buffered saline with Tween (TBST) containing 5% skim milk for 1 h, the membranes were incubated with specific primary antibodies, followed by horseradish peroxidase (HRP)-conjugated secondary antibodies.
The membranes were incubated with primary antibodies overnight and with secondary antibodies for 1 h. Finally, an enhanced chemiluminescence (ECL) detection system (GD50202; Monad Biotech. Co. Ltd., Wuhan, China) was used to visualize the signals, and the expression levels of each target protein were quantitatively analyzed based on band intensity.
Statistical analyses
We used IBM SPSS Statistics for Windows v. 22 (IBM Corp., Armonk, USA). Data (n = 3 or n = 9) are presented as the min–max range or as the median (1st and 3rd quartile (Q1, Q3)). The data were derived from 3 or 9 independent replications of a given experiment, and only the aggregate scores in Table 2, Table 3 represent the sum of scores for sub-indicators. For multiple comparisons involving variability across all groups, the nonparametric Kruskal–Wallis test (n = 3 or n = 9) was used for between-group comparisons, followed by Dunn’s test for planned pairwise comparisons.
When the global p-value in a Kruskal–Wallis test is non-significant, pairwise comparisons are not required. However, between-group comparison tests were not performed if any group had all values equal to a constant (n = 3). In cases where only 1 group had constant values (n = 3), 2 groups were directly compared using a one-sample nonparametric Wilcoxon signed-rank test. When both groups showed no variability (n = 3), standard statistical tests were not applied and the results were reported descriptively. In contrast, when both groups showed variation (n = 3), comparisons were performed using the Mann–Whitney U test. The p-values were corrected using the Bonferroni method for both multiple Kruskal–Wallis tests and multiple Dunn’s tests. Statistical significance was defined as p < 0.05.
Results
Erchen decoction regulates pathological injury of gastric tissue
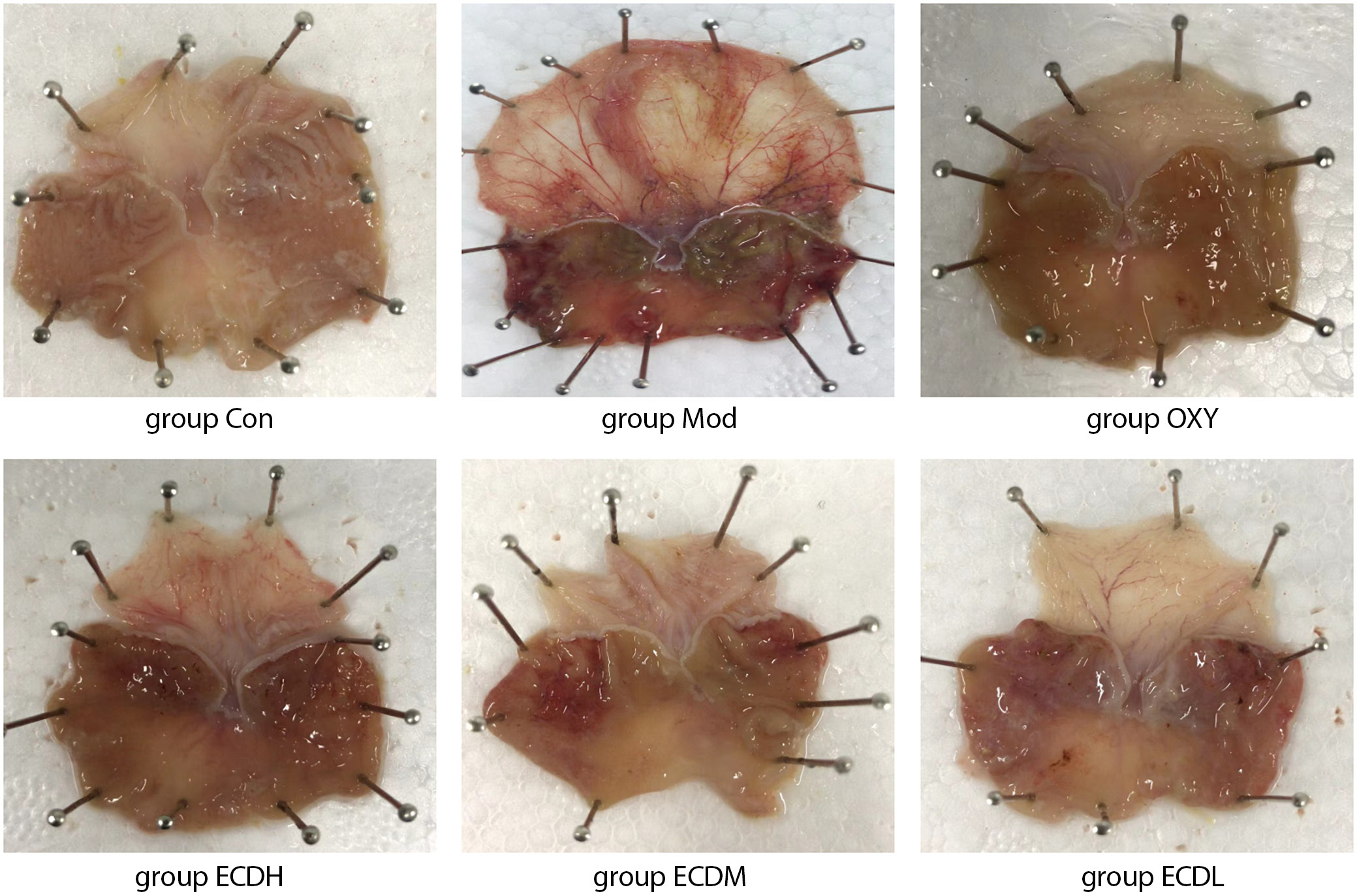
A comparison of the therapeutic effects of different doses of Erchen decoction based on macroscopic images of ethanol-induced gastric injury in rats showed that gastric tissue in the Con group exhibited no obvious lesions, with uniform coloration and intact structure. The scores for gastric epithelial erosion, ulceration, and hemorrhage were nearly 0, indicating no ethanol-induced damage. In contrast, gastric tissue in the Mod group displayed marked red hemorrhagic areas, including epithelial erosion, ulceration, and focal bleeding lesions. The Mod group had significantly higher aggregate scores than the Con group (Dunn’s test: ZMod vs Con = −13.667, adjusted pMod vs Con = 0.010), indicating severe damage induced by 50% ethanol. In the OXY group, gastric tissue damage was mild, hemorrhage was reduced, and the aggregate gastric injury score was lower than that in the Mod group, suggesting that OXY treatment may attenuate ethanol-induced injury. The severity of gastric tissue damage varied among the Erchen decoction dosage groups, with the ECDH group exhibiting the mildest damage, followed by the ECDM group. Compared with the ECDH and ECDM groups, the ECDL group exhibited more severe damage; however, relative to the Mod group, the ECDL group showed improvement, with lower scores for gastric mucosal and epithelial injury. These results indicate that Erchen decoction protects against ethanol-induced gastric injury in a dose-dependent manner (Figure 1, Table 2).
Erchen decoction regulates gastric injury in a dose-dependent manner
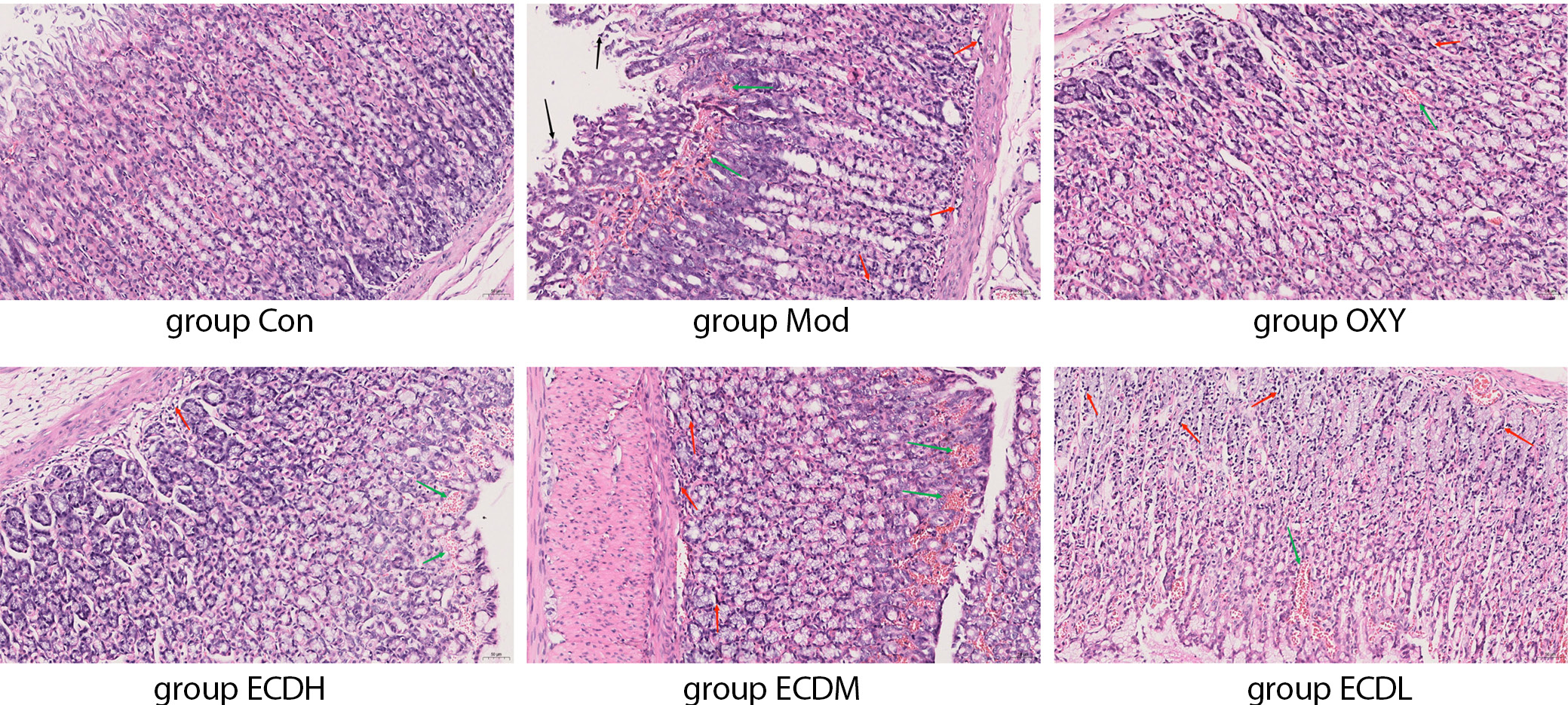
The results demonstrated the gastric tissue status across different treatment groups (Figure 2, Table 3). In the Con group, the overall gastric architecture was essentially normal: the mucosal layer was intact, the glands were neatly arranged, no epithelial shedding, atrophy, edema, or congestion was observed, and no abnormalities were detected in the submucosa or muscularis. Compared with the Con group, the Mod group exhibited an irregular mucosal layer, disorganized glandular arrangement, mucosal epithelial atrophy, edema and congestion, and submucosal edema accompanied by inflammatory cell infiltration.
The Mod group exhibited an abnormal overall gastric architecture, with a significantly higher aggregate score than the Con group (Dunn’s test: ZMod vs Con = −14.667, adjusted pMod vs Con = 0.005). Compared with the Mod group, both the OXY and ECDH groups showed improvement in ethanol-induced gastric injury. The general organization of the gastric tissue was largely preserved, with a slightly irregular mucosal layer, relatively well-organized glands, mild mucosal epithelial swelling and hyperplasia with congestion, and limited inflammatory cell infiltration in the submucosa. A reduction in the dose of Erchen decoction was associated with a gradual decrease in therapeutic efficacy against ethanol-induced gastric injury.
Erchen decoction promotes the proliferation of gastric injured cells
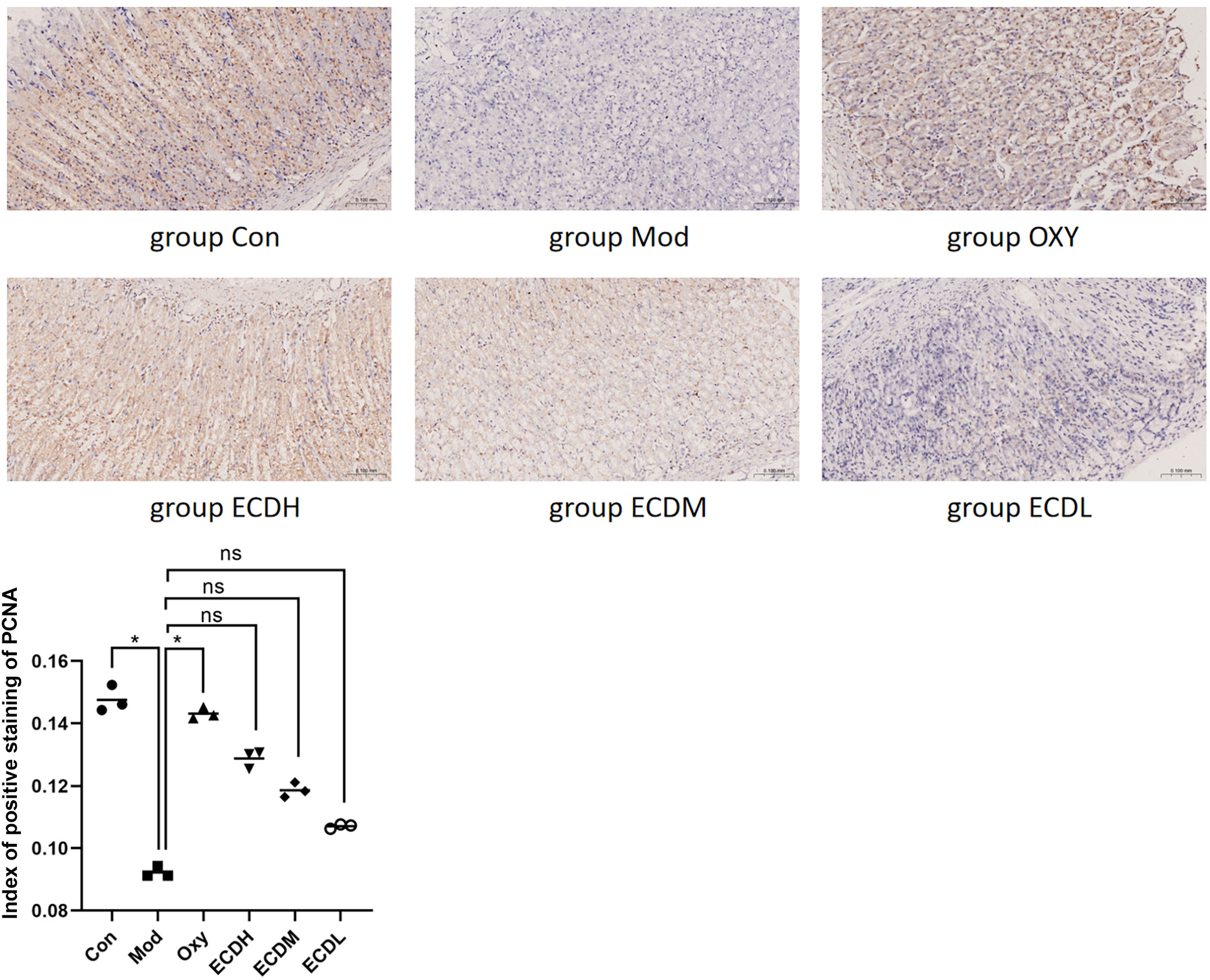
Cells in the Con group exhibited strong PCNA staining, indicating normal proliferative activity. In the Mod group, staining intensity was significantly reduced compared with the Con group (Dunn’s test: ZCon vs Mod = 14.667, adjusted pCon vs Mod = 0.005), suggesting that ethanol-induced gastric injury may suppress cell proliferation. In contrast to the Mod group, the OXY group demonstrated significantly stronger staining (Dunn’s test: ZOXY vs Mod = −12.333, adjusted pOXY vs Mod = 0.025), indicating that OXY treatment may promote the proliferation of damaged cells. Compared with the Mod group, PCNA staining in the Erchen decoction groups increased in a dose-dependent manner. The ECDH group exhibited the most intense staining and highest proliferative activity, whereas the ECDL group showed the lowest proliferative activity (Figure 3).
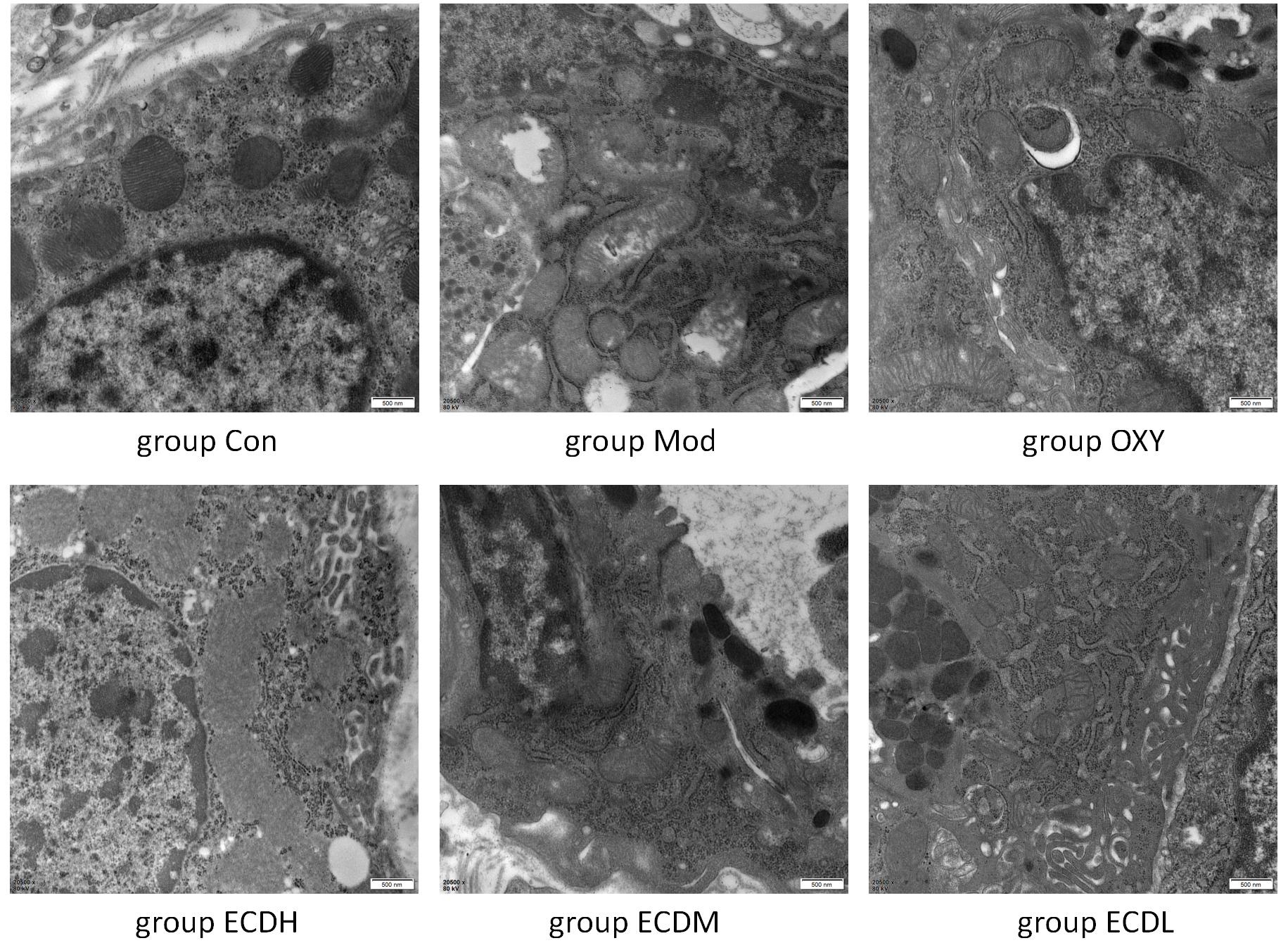
Erchen decoction improved the structure of gastric damaged cells
The Con group exhibited normal gastric ultrastructure, including well-defined mitochondria and intact cell membranes. Compared with the Con group, the Mod group showed marked mitochondrial swelling and cell membrane damage, indicating inflammation and cellular injury. The OXY and Erchen decoction groups demonstrated attenuation of these pathological changes, particularly in the ECDH group, where cellular architecture was substantially improved compared with the Mod group. Although the ECDM and ECDL groups showed partial structural improvement, these effects were less pronounced than those observed in the ECDH group (Figure 4).
Erchen decoction inhibited the expression of inflammatory factors
Compared with the Con group, the Mod group exhibited significantly higher levels of tumor necrosis factor alpha (TNF-α; Dunn’s test: ZMod vs Con = −44.500, adjusted pMod vs Con < 0.001) and IL-1β (IL-1β; Dunn’s test: ZMod vs Con = −42.778, adjusted pMod vs Con < 0.001), indicating that ethanol administration induced a pronounced inflammatory response. In the OXY group, the levels of these 2 inflammatory markers were comparable to those in the Con group but significantly lower than those in the Mod group (Dunn’s test for TNF-α: ZOXY vs Mod = 34.500, adjusted pOXY vs Mod < 0.001; Dunn’s test for IL-1β: ZOXY vs Mod = 34.944, adjusted pOXY vs Mod < 0.001), suggesting a significant anti-inflammatory effect of OXY treatment.
Compared with the Mod group, the ECDH group exhibited significantly reduced levels of TNF-α (Dunn’s test: ZECDH vs Mod = 28.000, adjusted pECDH vs Mod < 0.001) and IL-1β (Dunn’s test: ZECDH vs Mod = 27.111, adjusted pECDH vs Mod = 0.001), indicating that Erchen decoction decreased inflammation, with a more pronounced effect at higher doses. Compared with the Mod group, the ECDM group showed significantly lower IL-1β levels (Dunn’s test: ZMod vs ECDM = 21.167, adjusted pMod vs ECDM = 0.020) and intermediate TNF-α levels, whereas the ECDL group demonstrated intermediate levels of inflammatory markers. These findings suggest a mild anti-inflammatory effect at low and medium doses, which was less pronounced than that observed in the high-dose group (Table 4).
Erchen decoction enhanced the anti-oxidation of cells
Superoxide dismutase activity in the Mod group was significantly lower than that in the Con group (Dunn’s test: Z_Mod vs Con = 45.000, adjusted pMod vs Con < 0.001), whereas MDA levels were significantly higher (Dunn’s test: ZMod vs Con = −34.833, adjusted pMod vs Con < 0.001). These findings indicate that ethanol induced pronounced oxidative stress, reduced antioxidant capacity, and increased lipid peroxidation. The OXY, ECDH, and ECDM groups exhibited significantly higher SOD activity than the Mod group (Dunn’s test: ZOXY vs Mod = −30.111, adjusted pOXY vs Mod < 0.001; ZECDH vs Mod = −27.556, adjusted pECDH vs Mod = 0.001; ZMod vs ECDM = −23.333, adjusted pMod vs ECDM = 0.010), along with significantly lower MDA levels (Dunn’s test: ZOXY vs Mod = 31.222, adjusted pOXY vs Mod < 0.001; ZECDH vs Mod = 30.444, adjusted pECDH vs Mod < 0.001; ZMod vs ECDM = 28.611, adjusted pMod vs ECDM < 0.001). These results suggest that these interventions enhanced antioxidant defenses and attenuated oxidative damage. In the Erchen decoction treatment groups, SOD activity in the ECDH and ECDM groups was comparable to that in the Con group, and MDA levels were similarly low, indicating favorable antioxidant effects. Superoxide dismutase activity in the ECDL group also improved; however, the effect was less pronounced than that observed in the ECDH and ECDM groups (Table 5).
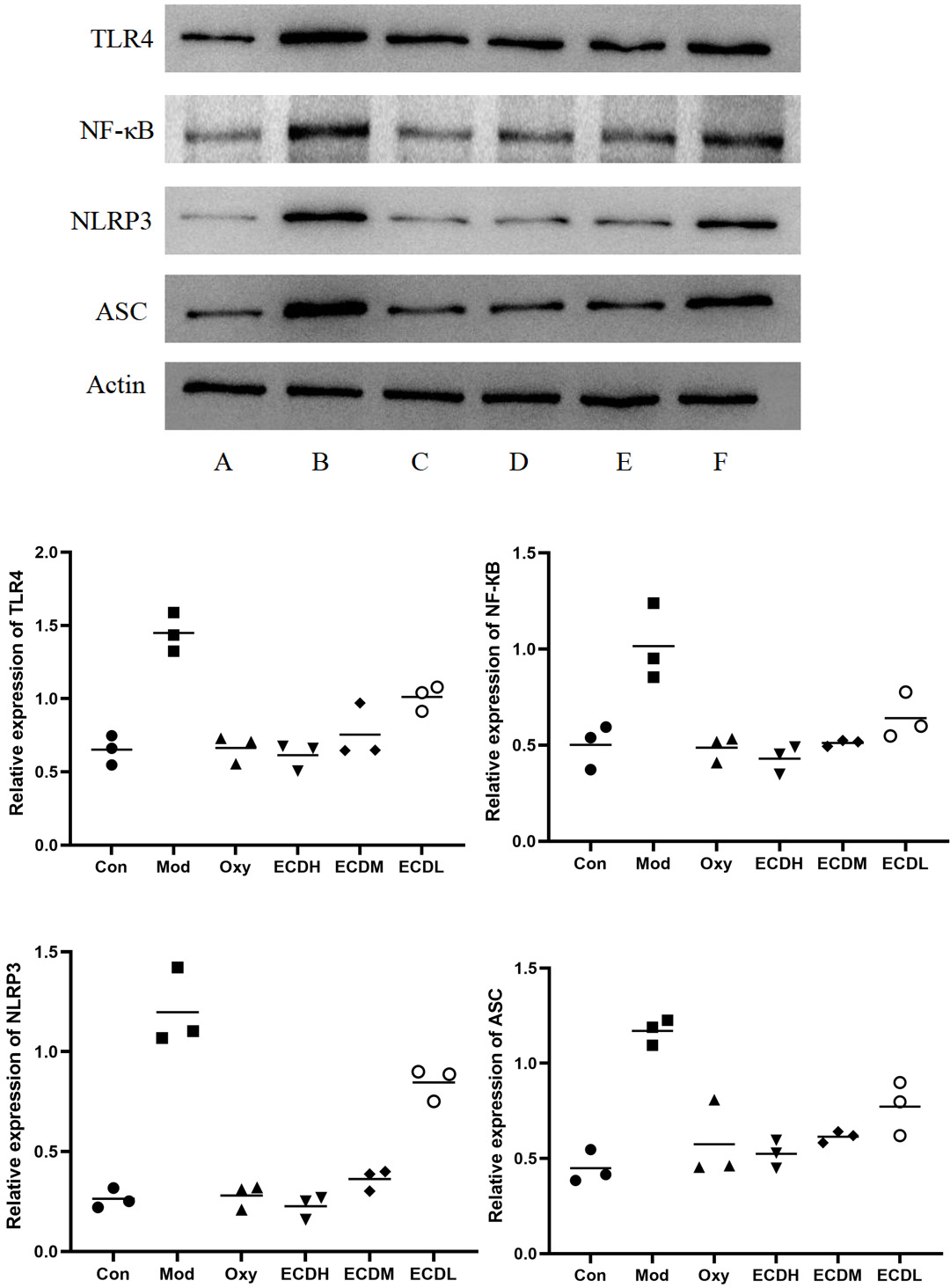
Erchen decoction inhibited the expression of inflammation-related proteins
The expression levels of the inflammation- and apoptosis-related proteins TLR4, NF-κB, NLRP3, and ASC were analyzed with WB. Compared with the Con group, the Mod group exhibited higher expression levels of these proteins. In contrast, protein expression in the OXY group was reduced relative to the Mod group, indicating that OXY treatment effectively alleviated ethanol-induced inflammatory activation. Similarly, protein levels in the 3 Erchen decoction-treated groups were lower than those in the Mod group, suggesting that Erchen decoction exerts anti-inflammatory effects (Figure 5).
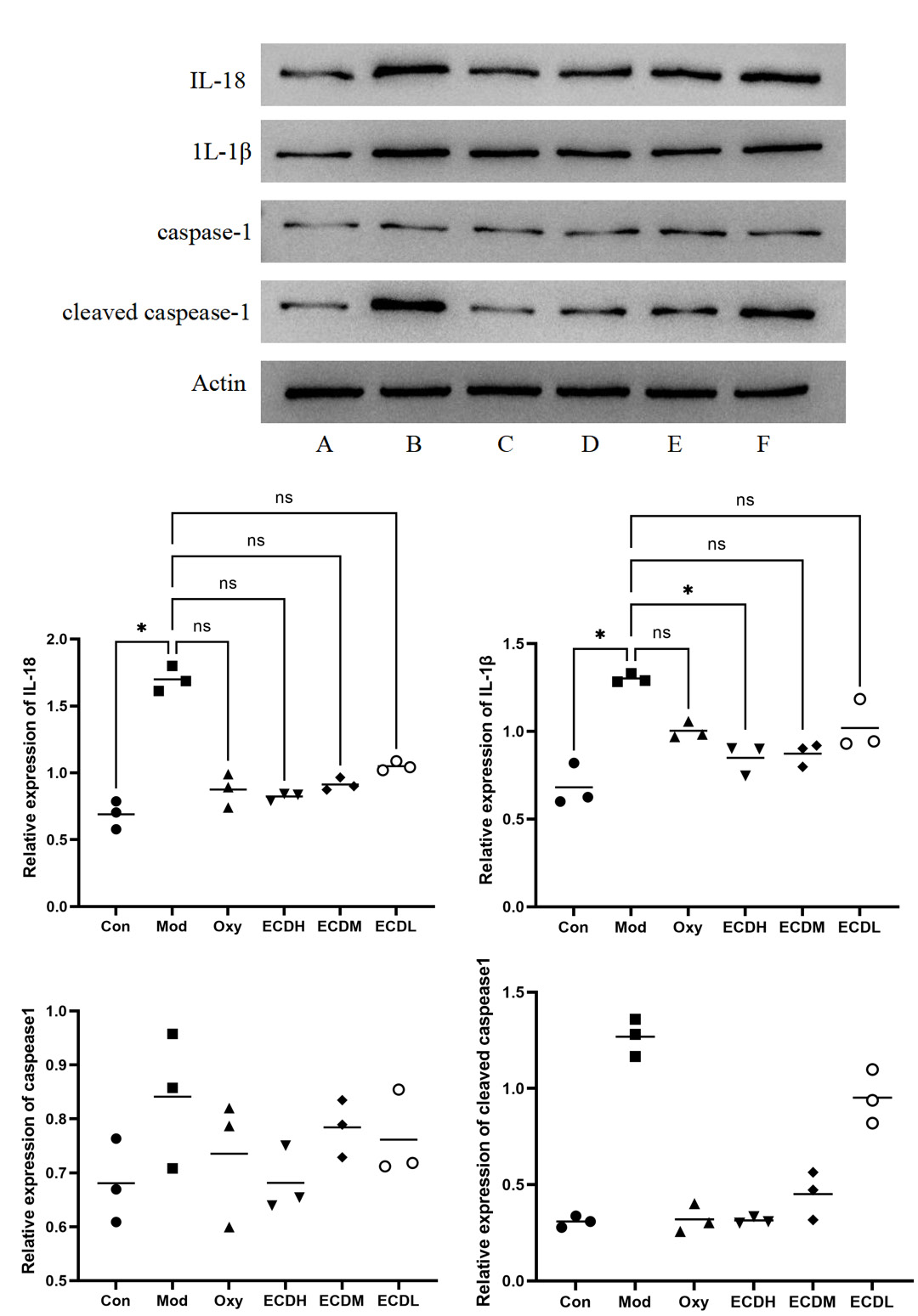
Erchen decoction inhibited the expression of proteins related to the activation pathway of inflammatory cells
Western blot analysis demonstrated higher expression of proteins involved in inflammatory pathway activation in the Mod group compared with the Con group, including significantly elevated levels of IL-18 (Dunn’s test: ZCon vs Mod = −14.667, adjusted pCon vs Mod = 0.005) and IL-1β (Dunn’s test: ZCon vs Mod = −14.333, adjusted pCon vs Mod = 0.005). These findings suggest that ethanol exposure induced a pronounced inflammatory response. Compared with the Mod group, the OXY group exhibited reduced levels of cleaved caspase-1, IL-1β, and IL-18, indicating that OXY treatment suppressed inflammatory activation and associated cell signaling. The levels of cleaved caspase-1, IL-1β, and IL-18 were lower in the ECDH, ECDM, and ECDL groups than in the Mod group. Notably, the reduction in IL-1β levels between the ECDH and Mod groups was statistically significant (Dunn’s test: ZECDH vs Mod = 11.333, adjusted pECDH vs Mod = 0.045). Although no statistically significant differences were observed among the other Erchen decoction-treated groups, the overall trend was consistent, suggesting that Erchen decoction exerted a dose-dependent suppressive effect on inflammation, with the highest dose (ECDH) showing the most pronounced effect (Figure 6).
Discussion
Gastric damage caused by excessive alcohol consumption is characterized by bleeding, gastric ulceration, erosion, and related lesions. Injured gastric tissue cells exhibited inflammatory infiltration, accompanied by decreased proliferative activity and increased apoptosis. Under ethanol stimulation, the expression of inflammation-related markers, including TNF-α and IL-1β, was significantly elevated, along with increased oxidative stress levels.
Erchen decoction was first recorded in the well-known Song Dynasty medical text Taiping Huimin Heji Ju Fang and has been widely used in TCM. Modern studies have demonstrated that Erchen decoction can reduce inflammatory mediators in tissues and is therefore frequently applied in conditions such as pneumonia, bronchitis, and hepatitis.27, 28, 29 In the present study, we confirmed that Erchen decoction effectively alleviates ethanol-induced gastric injury by regulating inflammatory factors and enhancing antioxidant capacity.
The gastric histopathological findings in the Erchen decoction-treated groups indicated a dose-dependent therapeutic effect, with higher doses producing greater histological improvement. Notably, the high-dose Erchen decoction achieved a therapeutic effect comparable to that of OXY. As a proliferation-associated nuclear antigen, PCNA expression is closely linked to cell proliferation.30, 31, 32 In the Mod group, ethanol exposure resulted in a significant reduction in PCNA expression, suggesting that ethanol not only disrupted the structural integrity of the gastric mucosa but also impaired cellular proliferative capacity. Compared with the Mod group, PCNA staining was more intense in the OXY group, indicating that OXY enhanced the proliferation of damaged cells and promoted gastric mucosal repair and regeneration. Similarly, PCNA staining intensity in the Erchen decoction groups increased in a dose-dependent manner.
Among the treatment groups, proliferative activity was greatest in the ECDH group, whereas the ECDL group also showed enhanced proliferation, albeit to a lesser extent. These findings indicate that Erchen decoction not only attenuates ethanol-induced gastric mucosal injury but also promotes the growth and repair of damaged gastric mucosal cells. Erchen decoction treatment reduced mitochondrial swelling and cell membrane disruption, thereby inhibiting cellular injury and the potential subsequent dysfunction or death.33 Following OXY treatment, the levels of inflammatory markers TNF-α and IL-1β were comparable to those in the Con group and significantly lower than those in the Mod group. Although the high-dose Erchen decoction did not achieve the same magnitude of effect as OXY, it still significantly reduced ethanol-induced inflammation, suggesting that high-dose Erchen decoction exerts a substantial anti-inflammatory effect.
Superoxide dismutase is a key antioxidant enzyme responsible for eliminating harmful superoxide radicals, whereas MDA is a byproduct of lipid peroxidation in cell membranes and is commonly used as a marker of oxidative damage.34 Compared with the Con group, the Mod group showed significantly reduced SOD activity and significantly elevated MDA levels. These changes indicate that ethanol exposure markedly impaired antioxidant defense capacity.11, 13, 14 Both the OXY group and the ECDH/ECDM/ECDL groups demonstrated varying degrees of improvement. Superoxide dismutase activity increased and MDA levels decreased in all treatment groups compared with the Mod group, suggesting that these interventions enhanced antioxidant defenses and attenuated oxidative damage. Overall, these findings indicate that Erchen decoction alleviates ethanol-induced gastric mucosal injury, at least in part, by increasing SOD activity and reducing MDA levels. Activation of the NF-κB pathway represents a central mechanism in ethanol-induced gastric injury. As a key regulator of the inflammatory response, the NF-κB pathway plays a pivotal role in promoting the expression of pro-inflammatory cytokines.35, 36 Alcohol exposure activates the NF-κB pathway, leading to elevated levels of inflammatory mediators such as TNF-α and IL-1β, thereby exacerbating gastric mucosal inflammation and injury. In the present study, ethanol exposure was associated with increased expression of NF-κB and related inflammatory signaling proteins, including TLR4, NLRP3, and ASC, further indicating activation of the NF-κB pathway in ethanol-induced gastric injury. Notably, this study further explored the regulatory effects of Erchen decoction on this pathway. Erchen decoction effectively suppressed NF-κB activation and the expression of downstream inflammatory mediators, particularly in the ECDH group. In addition, Erchen decoction reduced the levels of cleaved caspase-1, caspase-1, IL-1β, and IL-18, further suggesting that it alleviates ethanol-induced gastric mucosal injury, at least in part, through inhibition of the NF-κB pathway. These findings are important for understanding the mechanisms by which Erchen decoction mitigates chronic ethanol-induced gastric injury and suggest that its protective effects may be mediated by suppression of NF-κB signaling, thereby reducing inflammatory mediator release and inflammatory cell activation.
Limitations of the study
However, we acknowledge that our study has several limitations. Erchen decoction is a complex TCM formulation, and the specific active components and key target proteins responsible for regulating chronic ethanol-induced gastric injury remain unclear. We intend to address this important issue in future investigations.
Conclusions
This study confirmed that Erchen decoction alleviates ethanol-induced gastric injury by reducing inflammation, attenuating oxidative stress, preserving cellular structural integrity, and regulating the NF-κB signaling pathway. These findings not only suggest a potential TCM-based therapeutic approach for ethanol-induced gastric injury but also provide a scientific basis for further investigation of Erchen decoction in the treatment of other inflammation- and oxidative stress-related diseases.
Data Availability Statement
The datasets supporting the findings of the current study are openly available in the Zenodo repository at https://doi.org/10.5281/zenodo.16417628.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.