Abstract
Background. Wake-up stroke (WUS), an ischemic stroke occurring during sleep, accounts for 15–25% of acute ischemic strokes (AIS) cases and poses unique therapeutic challenges due to an unknown onset time. Circadian rhythms, regulated by the suprachiasmatic nucleus (SCN), influence various cardiovascular and metabolic processes, and disruptions of these rhythms have been implicated in stroke pathogenesis.
Objectives. This study explored whether WUS patients exhibit distinct circadian abnormalities compared with non-WUS patients, focusing on markers such as melatonin, cortisol, circadian clock gene expression, blood pressure (BP), and heart rate (HR).
Materials and methods. This exploratory, cross-sectional study included 28 participants (WUS: 8; non-WUS; 9, controls: 11). Blood samples were collected every 6 h over 24-h period, with melatonin and cortisol levels assessed via ultra-high-performance liquid chromatography–tandem mass spectrometry (UHPLC–MS/MS) and chemiluminescence, respectively. Circadian gene expression (CLOCK, CRY1, CRY2, PER1, PER2, and BMAL1) was analyzed using quantitative real-time reverse transcription polymerase chain reaction (RT-qPCR). Blood pressure and HR were recorded at 2-h intervals, and circadian rhythmicity was determined using MetaCycle analysis.
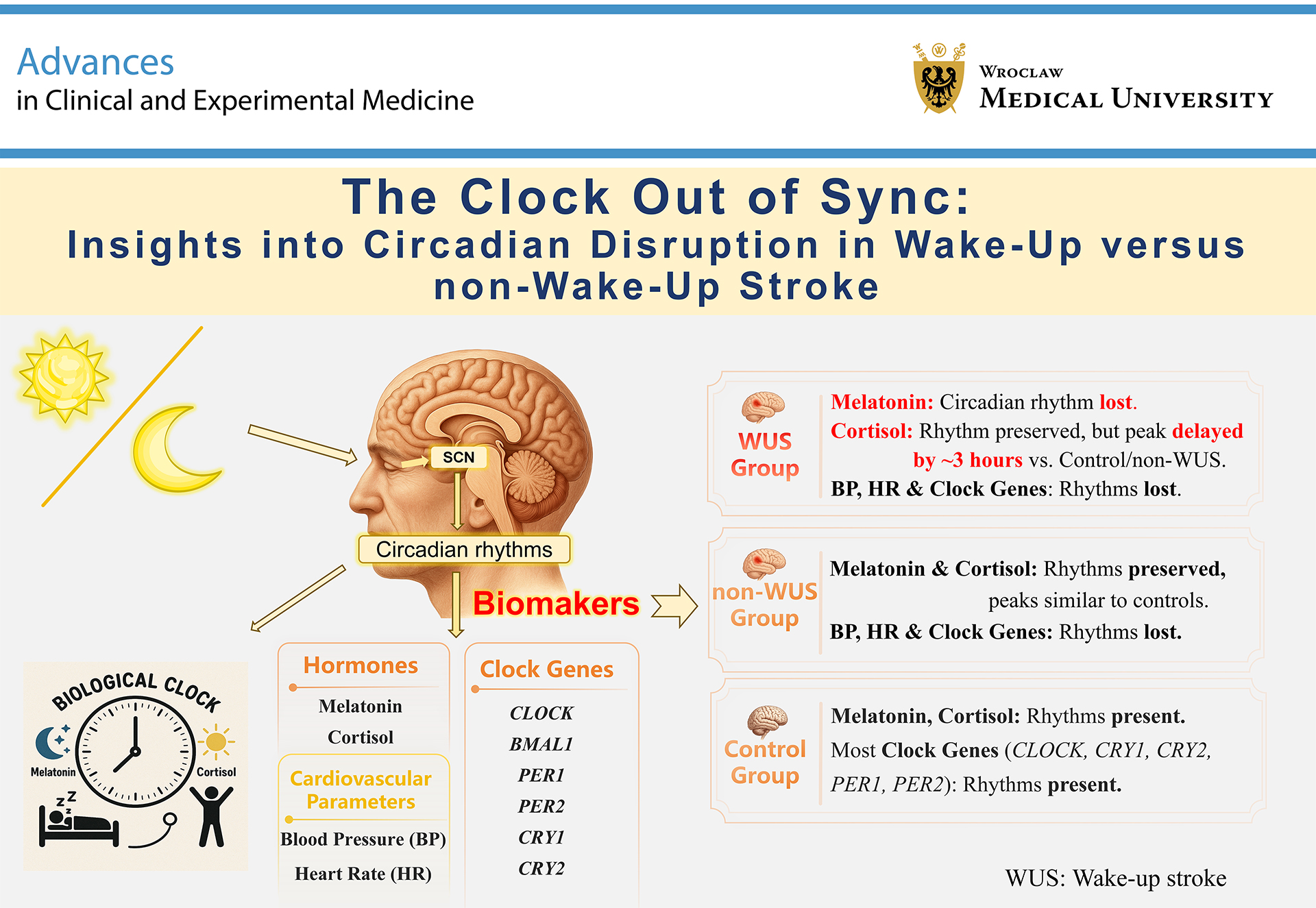
Results. The results revealed significant circadian rhythms in melatonin and cortisol in the non-WUS and control groups, with WUS patients showing a complete loss of melatonin rhythm and a 3-h phase delay in cortisol. Blood pressure and HR circadian variations were absent in both stroke groups, and none of the 6 clock genes exhibited rhythmicity in either the WUS or non-WUS group.
Conclusions. This study highlights the potential role of disrupted circadian rhythms in WUS pathogenesis, providing insights into targeted interventions such as light therapy. Future studies with larger cohorts are essential to confirm these findings and assess their clinical implications for stroke prevention and recovery.
Key words: circadian rhythm, melatonin, cortisol, clock genes, wake-up stroke
Background
Wake-up stroke (WUS) is an ischemic stroke that occurs during sleep and accounts for approx. 15–25% of acute ischemic strokes (AIS) cases.1, 2, 3 It is well known that stroke management emphasizes the critical importance of the therapeutic time window.4, 5, 6 Specifically, if the delay from symptom onset to discovery is prolonged, the optimal treatment window may be missed or fall into a suboptimal range, resulting in worse patient outcomes. Since WUS occurs during sleep, the therapeutic time window may have already elapsed by the time the patient wakes up. To better understand WUS, researchers have conducted various observational studies to analyze and compare its differences with non-WUS, aiming to identify distinct risk factors and develop targeted interventions to reduce its incidence.7, 8, 9, 10 Studies have shown that WUS is associated with a significantly higher prevalence of severe obstructive sleep apnea and an increased likelihood of newly diagnosed atrial fibrillation.11, 12, 13 However, no significant differences have been observed between WUS and non-WUS regarding other traditional risk factors, such as hypertension, diabetes, and smoking.14 Whether WUS is driven by pathophysiological mechanism distinct from those of non-WUS remains unclear. Some researchers have proposed that circadian factors may play a role in the pathogenesis of WUS.14, 15
Epidemiological studies have demonstrated a circadian distribution of AIS, with a higher incidence in the morning and a lower incidence during sleep.16, 17 This variation may be explained by the circadian rhythmicity of certain cardiovascular markers. For example, previous studies have shown that circadian variations in blood pressure (BP) mirror the temporal bimodal pattern of AIS.18, 19 Similarly, circadian rhythms within the hemostatic system warrant consideration, including morning increases in fibrinolytic activity, platelet aggregation, and endothelial dysfunction.20, 21 Given these observations, a key question arises: is WUS merely a random event, or does it involve pathophysiological mechanisms distinct from those of non-WUS? This study aims to explore this question from the perspective of circadian rhythms.
The circadian rhythmicity of the aforementioned cardiovascular factors, including endothelial function, thrombosis, and BP, is regulated by the circadian clock.22, 23 The circadian clock refers to an internal timekeeping mechanism in living organisms that regulates physiological and behavioral rhythms, aligning them with the day–night cycles of the external environment.24 In mammals, the master control center of this clock is located in the suprachiasmatic nucleus (SCN) of the hypothalamus.25 The SCN’s regulation of physiological processes related to circadian rhythms is extensive, encompassing the secretion of melatonin and cortisol, the expression of clock genes within the SCN itself, and the modulation of clock gene expression in peripheral tissues, as well as exerting strict control over numerous other bodily functions and the secretion of various fluids.26, 27, 28 Adamczak-Ratajczak et al. investigated the circadian rhythms of AIS through studies on melatonin and cortisol, finding that melatonin rhythm was preserved without phase shifts, while cortisol rhythm was advanced with higher median levels.29 However, circadian rhythmicity in WUS patients and whether it remains consistent with that of non-WUS patients have yet to be investigated.
Objectives
Based on these considerations, we hypothesized that WUS patients exhibit distinct circadian rhythmicity compared with non-WUS patients, potentially contributing to differences in pathogenesis. To test this hypothesis, we comprehensively evaluated and compared several circadian markers, including melatonin and cortisol levels, the expression patterns of circadian clock genes, and key cardiovascular parameters such as BP and heart rate (HR). These markers were analyzed across 3 groups: WUS patients, non-WUS patients, and healthy controls. This approach aimed to provide deeper insights into potential circadian abnormalities in WUS patients.
Materials and methods
Study design and participants
The study protocol has been described previously.30 This was an exploratory, single-center, prospective, cross-sectional, observational study.31, 32 All AIS patients who attended the Emergency Department of the First Affiliated Hospital of Soochow University (Suzhou, China) were evaluated for eligibility. Precise inclusion and exclusion criteria were established to reduce the number of confounding variables that could alter circadian rhythms. The inclusion criteria were as follows: 1) age between 40 and 80 years; 2) lesions of the anterior circulation caused by infarction (for patients); 3) mental clarity (for patients); 4) hospital admission between 6:00 and 18:00; and 5) willingness to participate in this study. The exclusion criteria were as follows: 1) daytime unwitnessed stroke; 2) infarction lesions involving the thalamus (for patients); 3) patients who underwent mechanical thrombectomy; 4) caffeine intake within 24 h prior to the study; 5) diagnosed depression or other psychiatric illnesses; 6) history of stroke, cancer, or other neurological disorders; 7) a history of insomnia, visual disorders, severe liver or renal dysfunction, or malignancy; 8) change in workplace or travel across 2 time zones within the previous 6 weeks; and 9) use of certain medications, such as β-receptor blockers (e.g., Betaloc) that suppress melatonin release, steroids, benzodiazepines, opioids, and immunomodulatory drugs. According to whether the symptom onset occurred during sleep or while awake, eligible patients were divided into 2 groups: WUS and non-WUS. Age-matched healthy subjects were recruited as a control group.
Ethics statement
The study was approved by the Medical Ethics Committee of the First Affiliated Hospital of Soochow University (approval No. 2019053) and was registered with the Chinese Clinical Trial Registry (ChiCTR1900024381). All participants provided written informed consent.
Initial assessment
Within 24 h of admission, all recruited patients were asked to undergo the initial evaluation, which consisted of clinical and neurological examinations, laboratory analyses, electrocardiography (ECG), cerebral computed tomography (CT), CT angiography, and CT perfusion imaging.
Circadian rhythm analysis
The participants were instructed to adhere to their regular nighttime sleep habits throughout the study period. No daytime naps were permitted. During the awake period, patients and healthy volunteers were exposed to standard fluorescent lighting, and the lights were switched off between 20:00 and 22:00 according to individual sleeping patterns. To ensure undisturbed sleep, all participants were accommodated in private rooms. During sleep, all subjects wore activity bracelet monitors (HUAWEI Band 4; Huawei Technologies Co., Ltd., Shenzhen, China) and eye masks. To minimize light interference, especially from corridor lighting, participants were required to keep their eye masks on throughout the sleep period, including during blood collection. Blood samples were drawn every 6 h over a 24-h period, specifically at 2:00, 8:00, 14:00, and 20:00. A dim, pen-sized yellow lamp was used during nighttime sampling to prevent light-induced suppression of melatonin secretion.
Blood samples were drawn from the antecubital vein into ethylenediaminetetraacetic acid (EDTA) tubes for the extraction of RNA to analyze circadian clock gene expression, and into coagulant tubes for serum separation. Serum was subsequently separated from blood samples collected in coagulant tubes and stored at −80°C until melatonin and cortisol levels were assessed.
Using a Philips patient monitor (model IntelliVue MX400; Koninklijke Philips N.V., Amsterdam, the Netherlands), HR and BP were measured hourly in all patients throughout the 24-h study period. In contrast, the volunteers’ HR and BP were measured using a 24-h ambulatory BP monitoring device. Data were extracted at 2-h intervals from the initiation of monitoring for each participant. After the participants awoke, sleep parameters such as total sleep duration, frequency of awakenings, and the proportions of rapid eye movement (REM) sleep, light sleep, and deep sleep were recorded. These parameters were transmitted from the activity bracelet monitor to a smartphone via a Bluetooth module.
Melatonin and cortisol assay
Melatonin levels were determined using ultra-high-performance liquid chromatography coupled with tandem mass spectrometry (UHPLC–MS/MS), a method developed to measure neurotransmitter concentrations with higher speed, greater resolution, and increased peak capacity.15 Cortisol levels were measured using chemiluminescent immunoassays with test kits provided by Mindray (Shenzhen, China). These immunoassays were performed in the Department of Nuclear Medicine at the First Affiliated Hospital of Soochow University.
Determination of clock gene expression
The expression levels of 6 circadian clock genes were analyzed, including CLOCK, BMAL1, 2 period genes (PER1 and PER2), and 2 cryptochrome genes (CRY1 and CRY2). The expression levels of the 6 target genes and the β-actin gene (used as an internal reference control) were examined using quantitative real-time reverse transcription polymerase chain reaction (RT-qPCR). Total RNA was extracted from blood samples using TRIzol reagent, and RNA integrity and quantity were assessed with 1% agarose gel electrophoresis. RNA was reverse transcribed into cDNA using a reverse transcription kit (Takara, Tokyo, Japan). The reverse transcription reaction volume was 10 μL, and the reaction conditions were 37°C for 15 min followed by 85°C for 5 s. The primers were designed using Primer Premier 5.0 software (PREMIER Biosoft International, Palo Alto, USA). RT-qPCR was performed in accordance with the manufacturer’s instructions using a qTower3G real-time PCR thermal cycler (Analytik Jena, Jena, Germany). The amplification was performed in a reaction volume of 20 μL, and the program consisted of an initial denaturation at 95°C for 3 min, followed by 45 cycles of 95°C for 5 s, 60°C for 5 s, and 72°C for 15 s. Relative gene expression levels were quantified using the comparative threshold cycle (2–ΔΔCt) method. The relative expression levels of the clock genes were determined by normalizing their expression values to those of β-actin in each sample.
Statistical analyses
Statistical analyses were conducted using R v. 4.4.1 (R Foundation for Statistical Computing, Vienna, Austria) and IBM SPSS Statistics v. 27 for Windows (IBM Corp., Armonk, USA). GraphPad Prism v. 8 (GraphPad Software, San Diego, USA) was used to generate graphs. A two-sided p < 0.05 was considered statistically significant.
Baseline data analysis
The study cohort included 8 participants in the WUS group, 9 in the non-WUS group, and 11 in the control group. Given the small sample sizes in each group and the resulting low power of normality tests, we opted for nonparametric statistical analyses. Categorical variables are presented as frequencies and percentages (n, %), and group differences were assessed using Fisher’s exact test for 2 groups or the Fisher–Freeman–Halton test for 3 groups. Continuous variables are presented as the median and range (min–max) for sample sizes ≤8 and as the median with interquartile range (IQR; Q1–Q3, where the 1st quartile (Q1) and the 3rd quartile (Q3) represent the 25th and 75th percentiles) for sample sizes >8. Group differences were assessed using the Wilcoxon rank-sum test for 2 groups or the Kruskal–Wallis test for 3 groups.
Circadian marker analysis
Group comparisons
The mean value for each participant, based on circadian marker data collected at multiple time points, was used for intergroup comparisons. Due to the distribution of the data, nonparametric tests were applied. Two-group comparisons were conducted using the Wilcoxon rank-sum test, whereas 3-group comparisons were performed using the Kruskal–Wallis test, followed by a post hoc pairwise Wilcoxon rank-sum tests with Bonferroni correction.
Repeated measures analysis
To account for repeated measures and within-subject correlations in the data, statistical analyses employed generalized estimating equations (GEE), a marginal modeling approach suitable for small sample sizes and non-normally distributed repeated-measures data.33 For both melatonin and cortisol levels, separate GEE models were fitted with the group category as a fixed effect. The models used an identity link function and assumed an exchangeable correlation structure, as indicated by the alpha and gamma parameters (for melatonin: alpha = 0.319, gamma = 10,934; for cortisol: alpha = 0.348, gamma = 20.1). Model specifications included residual degrees of freedom (df) = 100, a number of clusters (n.clusters) = 28, and a maximum cluster size of 4. Post hoc pairwise comparisons of marginal means for cortisol were performed at each time point, with p-values adjusted using the Bonferroni correction to control for multiple testing. All analyses were conducted at a significance level of α = 0.05.
Circadian rhythmicity analysis
Circadian rhythm parameters were analyzed using the MetaCycle package in R to obtain rhythmicity indices, including amplitude, period, phase, and p-value.34 Amplitude reflects the magnitude of oscillation, the period represents the length of the 24-h cycle, the phase indicates the timing of the rhythm peak, and a p < 0.05 indicates the presence of circadian periodicity for that parameter.
Results
Baseline characteristics of the WUS, non-WUS, and control groups
A total of 28 participants were included, with 8 in the WUS group, 9 in the non-WUS group, and 11 in the control group. The mean age of the participants was 59.4 ±9.5 years, and 18 (64.3%) were male. There were no significant differences in baseline characteristics, including age, sex, and sleep metrics (total sleep duration, frequency of awakenings, and the proportions of REM sleep, light sleep, and deep sleep), among the 3 groups. The prevalence of comorbidities (such as diabetes, hypertension, and atrial fibrillation) showed no significant differences between the WUS and non-WUS groups (Table 1).
Melatonin and cortisol
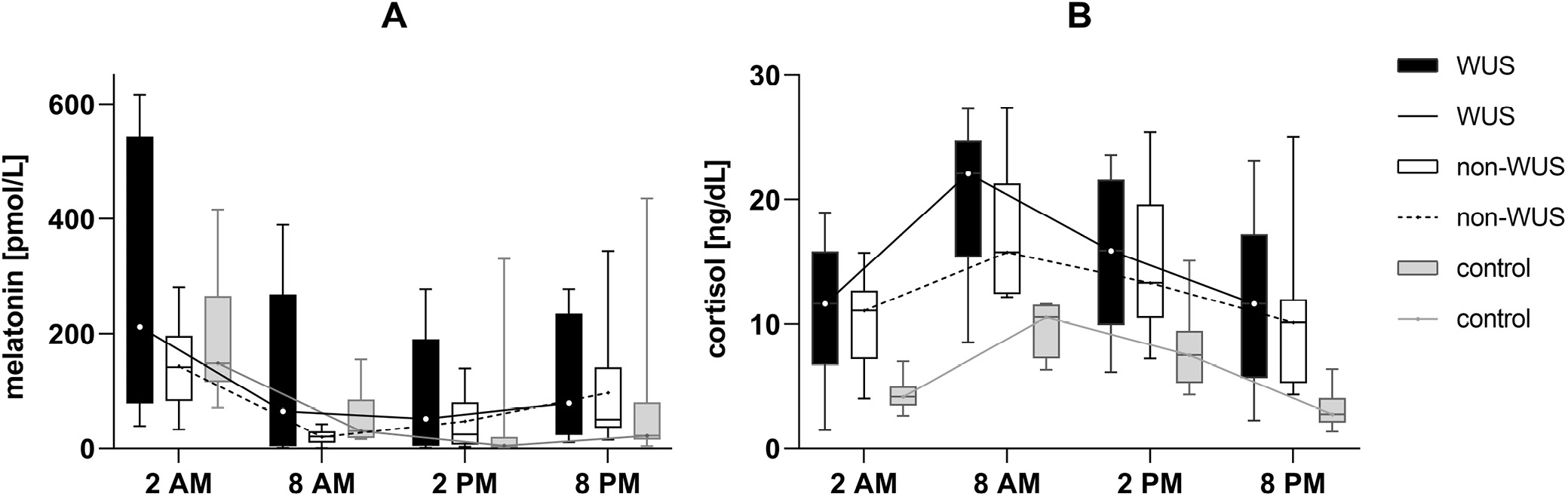
As shown in Table 2, melatonin and cortisol concentrations in the control group exhibited significant circadian rhythmicity across different time points. Rhythmicity analysis identified an acrophase at 02:30 for melatonin and at 08:22 for cortisol. Similarly, in the non-WUS group, both melatonin and cortisol concentrations also displayed a circadian rhythm. Rhythmicity analysis revealed an acrophase at 02:14 for melatonin and at 08:56 for cortisol, timing that was similar to that observed in the control group. However, in the WUS group, the circadian variation of melatonin was absent, whereas cortisol maintained its rhythmicity. Rhythmicity analysis showed an acrophase at 11:59 for cortisol in the WUS group, indicating a phase delay of 3 h 37 min compared with the control group. Additionally, overall cortisol concentrations differed significantly among the groups (H = 18.98, p = 0.001). Post hoc Bonferroni tests showed significantly higher cortisol levels in both the WUS (W = 1384, adjusted p < 0.001) and non-WUS (W = 1584, adjusted p < 0.001) groups compared with controls. No significant difference was found between the WUS and non-WUS groups. Descriptive statistics are presented in Table 1, and the full post hoc comparisons are provided in Supplementary Table 1.
The GEE analysis assessed group differences in melatonin and cortisol levels, adjusting for repeated measures and using the WUS group as the reference. Melatonin levels did not differ significantly between the WUS group and either the non-WUS group or control group. Regarding cortisol, no significant difference was found between the WUS and non-WUS groups. However, WUS patients exhibited significantly higher cortisol levels compared with the controls (β = −6.710, p < 0.001). Post hoc Bonferroni-adjusted pairwise comparisons further revealed that, relative to controls, both the WUS group (estimate = 8.472, standard error (SE) = 1.804, adjusted p < 0.001) and the non-WUS group (estimate = 6.836, SE = 1.014, adjusted p < 0.001) had significantly elevated cortisol levels (Table 3; pairwise comparisons are shown in Supplementary Table 2). Figure 1 displays the melatonin and cortisol concentrations at the 4 time points for the 3 groups, with circadian profiles plotted based on their median values.
Circadian clock genes expression
MetaCycle analyses indicated that in the control group, the expression profiles of C LOCK, CRY1, CRY2, PER1, and PER2 exhibited significant circadian rhythmicity, with the exception of BMAL1. However, none of the 6 assessed clock genes showed circadian rhythmicity in the WUS or non-WUS groups (Table 2).
Blood pressure and heart rate
In the control group, systolic and diastolic blood pressure (SBP and DBP) and HR exhibited regular circadian rhythmicity (p < 0.001); however, this rhythmicity was absent in both the WUS and non-WUS groups (Table 2).
Group comparisons revealed significant differences in SBP (H = 10.11, p = 0.006) and DBP (H = 9.353, p = 0.009) among the 3 groups. Post hoc analyses with Bonferroni correction showed that the WUS group had significantly higher SBP than controls (W = 80.5, adjusted p = 0.009), and the non-WUS group had significantly higher DBP than the controls (W = 85, adjusted p = 0.023). All other pairwise comparisons for SBP and DBP yielded nonsignificant results. Descriptive data are presented in Table 1, and the full post hoc results are provided in Supplementary Table 1.
Discussion
In mammals, circadian rhythms are essential for maintaining physiological homeostasis. Disrupted circadian rhythm can contribute to the loss of homeostasis and the development of disease.35, 36 Circadian rhythms are controlled by the SCN in the hypothalamus, which is influenced by a variety of factors. In this study, we explored circadian rhythm indicators in WUS patients to investigate potential associations between WUS and abnormal circadian rhythms. The results revealed disrupted circadian rhythmicity in the WUS group compared with the non-WUS and control groups.
Melatonin is an endogenous neurohormone primarily produced by the pineal gland. It is currently regarded as the most accurate marker of human circadian rhythmicity and SCN function.37 Its rhythm is minimally, if at all, influenced by environmental factors such as the sleep–wake state, exercise, or emotions. However, known external factors that can affect the melatonin phase include light exposure, posture, and ambient temperature.38, 39, 40 With the onset of darkness, melatonin levels gradually increase throughout the night, peaking in the early morning (between 02:00 and 04:00).
In this study, MetaCycle analyses revealed that melatonin secretion in the non-WUS and control groups exhibited significant circadian rhythmicity, with peak secretion times at 02:14 and 02:30, respectively. These findings suggest that the 2 groups share a similar circadian rhythm for melatonin. The results align with a previous study by Adamczak-Ratajczak et al., which analyzed the circadian phase of AIS based on plasma melatonin and found that the circadian rhythm of melatonin was sustained without any phase shift.29 However, the WUS group showed a complete loss of melatonin circadian rhythmicity.
Previous studies have established that the majority of humans exhibit a robust and persistent daily melatonin rhythm.37, 41 Therefore, the findings of this study may indicate that WUS patients experience disrupted circadian rhythms, and this discrepancy compared with non-WUS patients suggests that circadian rhythm abnormalities in WUS patients may be present even before stroke onset.
Cortisol, the major end product of the hypothalamic–pituitary–adrenal (HPA) axis,42 is another important marker for investigating circadian rhythms. Under normal, stress-free conditions, the adrenal cortex produces cortisol, which is then released into the bloodstream in a circadian pattern. Peak cortisol secretion in adults typically occurs between 08:00 and 10:00.43 In this study, cortisol levels exhibited circadian rhythmicity across all 3 groups. However, in the WUS group, the cortisol rhythm showed an acrophase shift, with an approximate 3-h delay compared with the control and non-WUS groups (the acrophase occurred at 11:59 in the WUS group, compared with 08:22 in the control group and 08:56 in the non-WUS group). This finding supports the conclusion that WUS patients exhibit disrupted circadian rhythms, specifically manifested as a phase delay. In addition, serum cortisol levels in the WUS and non-WUS groups were significantly higher than those in the control group, consistent with previous studies.44, 45 One prior study reported that cortisol levels remain elevated for at least 7 days following stroke in most patients.45 As a stressor, AIS can stimulate the HPA axis, leading to increased cortisol secretion.46
In mammals, circadian rhythms are believed to be governed by a fundamental molecular autoregulatory feedback loop involving key clock genes such as Period (PER1 and PER2), Cryptochrome (CRY1 and CRY2), BMAL1, and CLOCK, along with their protein products.47 Beyond the central SCN circadian pacemaker, clock genes are expressed in most mammalian cells and organs, including peripheral blood cells.48, 49 In this study, we investigated the circadian rhythmicity in the expression of 6 clock genes in the peripheral blood of the WUS, non-WUS, and control groups. In the control group, all assessed clock genes, except BMAL1, exhibited significant rhythmic expression. This observation aligns with previous research suggesting a highly variable daily pattern of BMAL1 expression across individuals.50, 51 Although BMAL1 is essential for the regulation of 24-h behavioral cycles, it is not critical for 24-h molecular rhythms at the transcriptional, translational, or post-translational levels, as demonstrated by Ray et al.52
In contrast, neither the WUS group nor the non-WUS group showed rhythmic expression of any of the 6 clock genes. Previous studies have shown that the disruption of circadian rhythms increases the risk of stroke by affecting physiological processes such as BP, platelet function, coagulation, and fibrinolysis.53, 54 However, an acute stroke itself can also disrupt circadian rhythms and impair the neuronal clock mechanism. Research has demonstrated that neurons in the SCN have their own clock gene cycles, while astrocytes help synchronize SCN timing by recycling gamma-aminobutyric acid and glutamate.55, 56, 57 During the occurrence of AIS, astrocyte function is altered, potentially disrupting this recycling process and, consequently, impairing the normal rhythmicity of the SCN.58 Therefore, based solely on these gene expression results, it remains challenging to determine whether circadian rhythm precedes or merely follows stroke onset in WUS patients.
Several physiological processes, including BP and HR, exhibit circadian rhythmicity in humans.59, 60 Analysis of our control group confirmed this phenomenon. However, our findings revealed a complete loss of circadian variation in BP and HR in both the WUS and non-WUS groups. Previous research has shown that irregular BP rhythms can increase the risk of stroke,61 and that stroke events themselves can pathologically reduce or abolish circadian BP variation.62 In a study using spontaneously hypertensive rats prone to malignant stroke, Masaki et al. observed a sudden change in HR, followed by a progressive loss of circadian BP variation occurring just as a stroke began.63 Consequently, this aspect of the data provided limited insight into the specific circadian rhythms of WUS patients. Additionally, participants in the stroke groups exhibited significantly higher SBP or DBP compared with the control group in our study. This finding may be attributed to the higher prevalence of hypertension among stroke patients, combined with the acute physiological stress induced by the stroke event.
Although an acute stroke itself may cause circadian rhythm disruptions, this study revealed several noteworthy findings. The rhythmicity of the most prominent circadian rhythm marker, melatonin, was completely absent in the WUS group but preserved in the non-WUS group. Additionally, the cortisol rhythm persisted in the WUS group but exhibited a phase delay compared with the non-WUS group. Although other markers showed no differences between the 2 groups, we hypothesize that WUS patients may have preexisting circadian rhythm disruptions before disease onset. For these patients, as well as others with clearly identified circadian rhythm disorders, targeted interventions aimed at regulating circadian rhythms, such as light therapy or pharmacological treatments, could be implemented alongside strategies to prevent stroke recurrence.64
These measures may help reduce the occurrence of strokes, particularly WUS. Moreover, research suggests that disruptions in the circadian rhythm system may impair the ability of neural tissue to repair itself, thereby affecting the recovery process.65 Restoring circadian rhythms after a stroke may also contribute to improved neurological function and overall health outcomes in patients.65, 66
Limitations of the study
This study has several limitations that should be acknowledged. First, given the exploratory nature of this study, the sample size was relatively small; therefore, the findings should be interpreted with caution, and replication with a larger sample size is needed in future research. Second, due to the limitations inherent to human studies, obtaining repeated blood samples over 2 or 3 consecutive days is challenging, which limits the reproducibility of circadian rhythm analyses and may weaken the robustness of the results. Third, as this was a cross-sectional study, causal relationships cannot be determined. Fourth, the strict exclusion criteria were primarily designed to exclude conditions, dietary factors, medications, or work–rest patterns (e.g., shift work or night work) known to affect circadian rhythms.67, 68, 69
Such external factors that interfere with circadian rhythms would likely have influenced the results of this study. The comorbidities and medications that were excluded were not specific to stroke and therefore would not bias the study toward a profile unrepresentative of the broader stroke patient population. Nevertheless, patients engaged in shift work or night work were excluded, and examining the influence of circadian rhythms on WUS in these patients would be of interest in future studies. Finally, in this study, sleep was monitored using a smart device. Participants were accommodated in single-occupancy rooms, lights were not permitted, and they were required to wear eye masks during sleep. The only disturbance occurred during nighttime specimen collection. To avoid turning on the room lights, only a dim yellow light from a penlight was used. Efforts were made to minimize exogenous interference, but some confounding factors may still have been present. Despite these limitations, we believe that our work may provide additional insights into the pathogenesis of WUS and serve as inspiration for future studies exploring its potential mechanisms. For example, in future research, when establishing databases for long-term follow-up of high-risk stroke populations, dynamic electrocardiographic and blood pressure data could be included, as these may serve as feasible indicators of circadian rhythms. If a stroke event is observed during follow-up, we would also record the time of stroke onset to further examine the correlation between circadian rhythms and stroke onset time. More research with an adequate sample size is crucial to validate our outcomes.
Conclusions
This study represents the first exploratory investigation of circadian rhythms in WUS patients. Our findings reveal that WUS patients exhibit significant disruptions in circadian rhythms, including the loss of melatonin rhythmicity and a phase delay in cortisol secretion. These alterations differ from those observed in non-WUS patients, in whom some circadian markers, such as melatonin and cortisol, remained intact. Our results suggest that disrupted circadian rhythms may be an intrinsic feature of WUS, possibly predating stroke onset. From a theoretical perspective, this study provides evidence supporting the role of circadian rhythms in stroke pathogenesis, particularly in WUS. For example, light therapy could be optimized for WUS patients by delivering 2,000–10,000 lux of blue-enriched light in the morning (06:00–08:00) to phase-advance delayed rhythms, while minimizing evening light exposure to prevent further disruption.70, 71 Such strategies, combined with pharmacological treatments or lifestyle adjustments, may help regulate circadian rhythms and reduce the risk of stroke recurrence. These findings underscore the need for larger-scale, longitudinal studies to validate and expand upon the observed associations and to explore the causal mechanisms underlying circadian rhythm disruptions in WUS. By deepening our understanding of the interplay between circadian biology and stroke, this line of research holds promise for the development of novel preventive and therapeutic strategies tailored to the unique characteristics of WUS.
Supplementary data
The supplementary data are available at https://doi.org/10.5281/zenodo.15745360. The package contains the following files:
Supplementary Table 1. Detailed post hoc test results for group comparisons of cortisol, SBP, and DBP using the Kruskal–Wallis test in Table 1.
Supplementary Table 2. Bonferroni-adjusted pairwise comparisons of GEE-estimated marginal means of cortisol concentrations between groups across 4 time points.
Use of AI and AI-assisted technologies
In the preparation of this manuscript, we used ChatGPT, solely for language refinement. The AI tool was employed to assist with language editing and enhancement. However, the author carefully reviewed and ensured that all language revisions accurately conveyed the intended meaning. The authors take full responsibility for the content of the manuscript, including any portions enhanced by AI tools.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.