Abstract
Background. Procedural sedation reduces pain, anxiety, and discomfort during emergency department (ED) procedures. Analgesics, sedatives and anxiolytics ensure patient comfort but can cause respiratory, cardiovascular, neurological, and gastrointestinal (GI) side effects.
Objectives. To synthesize data on adverse events and procedural efficacy from randomized controlled trials (RCTs) involving the use of different drugs/drug combinations used for procedural sedation.
Materials and methods. A systematic literature review was conducted using MEDLINE and CENTRAL databases for RCTs that involved adult patients who were administered drugs for procedural sedation in the ED. Outcomes such as hypoxia, hypotension, apnea, agitation, GI disturbances (nausea, vomiting), and bradycardia/dysrhythmia were extracted from the trials. A meta-analysis was performed using OpenMeta (Analyst) software to determine the pooled incidence of each adverse outcome and procedural success for the different drugs and drug combinations.
Results. A total of 50 RCTs were included in the meta-analysis with information from 5,398 patients. Meta-analysis of the data showed a pooled incidence of 136 events/1,000 sedations (95% confidence interval (95% CI): 112–160) for hypoxia, 23 events/1,000 sedations (95% CI: 15–31) for hypotension, 143 events/1,000 sedations (95% CI: 110–177) for agitation, 51 events/1,000 sedations (95% CI: 38–64) for apnea, 45 events/1,000 sedations (95% CI: 30–60) for GI disturbances, and 30 events/1,000 sedations (95% CI: 19–41) for bradycardia/dysrhythmia. The procedural success rate was 921 events/1,000 sedations (95% CI: 896–946).
Conclusions. The results of this meta-analysis shed light on the adverse effects of various procedural sedation agents used for different procedures in the ED. Ketamine–propofol combinations were associated with less hemodynamic instability, whereas propofol caused hypotension and respiratory events and GI disturbances were associated with the administration of fentanyl and ketamine. Thus, individual patient factors such as age and comorbidities and the risk profile of different agents should be considered during procedural sedation protocols. This meta-analysis summarizes the side effects of several drugs to help physicians administer them safely.
Key words: ketamine, emergency department, propofol, midazolam, procedural sedation
Introduction
Procedural sedation, also referred to as procedural sedation and analgesia (PSA), is a cornerstone practice in the emergency department (ED) to relieve anxiety, discomfort, and pain during painful procedures, thus ensuring patient comfort and safety. The American College of Emergency Physicians (ACEP) defines procedural sedation as “a technique of administering sedatives or dissociative agents with or without analgesics to induce a state that allows patients to tolerate unpleasant procedures while maintaining cardiorespiratory function. PSA is intended to result in a depressed level of consciousness that allows the patient to maintain oxygenation and airway control independently.”1, 2 Therefore, it is important that the medications that are administered alleviate the pain of performing procedures as well as preserve vital physiological functions.3 Common procedures for which PSA is used in the ED include orthopedic manipulations (closed reduction of fractures or dislocations), laceration repairs, burn debridement and wound care, chest tube insertions, and cardioversions for arrhythmias.
Single drugs, or more often multiple drug combinations, are used to achieve suitable levels of sedation, analgesia, and anxiolysis, and their application is guided based on patient characteristics (age, comorbidities), type of procedure, and safety considerations. Sedatives such as short-acting benzodiazepines (midazolam), diazepam, and propofol are often used along with opioid analgesics (fentanyl, morphine and remifentanil), and dissociative agents (ketamine) are commonly administered.4, 5 Despite careful consideration of patient characteristics prior to the administration of medications for procedural sedation, trained emergency medicine personnel, and policies in place to monitor patients and report adverse events, individual responses to procedural sedation drugs are variable. Jones et al. have reported the occurrence of several respiratory adverse events such as hypoxemia, over-sedation, and apnea, which are related to age and comorbidities.6 Therefore, despite advances in monitoring and vigilance protocols, and titration of drug/drug combinations, adverse events are common and should be analyzed to determine the safety of different drugs and drug combinations.
Studies conducted to determine the safety and efficacy of procedural sedation agents have been single-center randomized controlled trials (RCTs), observational studies, and most often studies with smaller sample sizes (<100 patients), making it difficult to draw conclusions on the safety of these agents. Meta-analysis is necessary to synthesize the data from various trials, thereby providing a more robust knowledge base regarding the safety of drugs and drug combinations used for procedural sedation.
Objectives
The objective of this work is to synthesize and analyze the available literature and evaluate the efficacy and safety of procedural sedation in adult patients in the ED, with a focus on adverse effects of various drugs and drug combinations.
Materials and methods
Search strategy
A systematic literature search was conducted of MEDLINE (PubMed) and the Cochrane Register of Controlled Trials (CENTRAL) in December 2024, covering the period from 1990 to 2024. The following search terms were used in various combinations: “procedural sedation”, “conscious sedation”, “emergency department”, “hypoxia”, “adverse events”, “midazolam”, “fentanyl”, “propofol”, “ketamine”, “etomidate”, and “lidocaine”. Additionally, a comprehensive list of search terms, including Medical Subject Headings (MeSH) terms, was applied. The titles and abstracts of potentially relevant studies were scanned, and the full-text versions of the appropriate articles were read. Additional studies were identified by cross-checking the reference lists of the relevant studies.
Study selection
We included RCTs that included patients undergoing different types of procedures and undergoing procedural sedation and analgesia. Adult patients (≥18 years) in the ED given all types of medication for procedural sedation were included in our analysis. Exclusion criteria included non-randomized studies and prospective and retrospective study designs.
Data extraction and quality assessment
Following identification of articles that met the inclusion criteria, data were extracted using a predefined data extraction form that included the following items: authors, year, study design, intervention (drug and drug combinations), and type of procedure. Outcomes such as procedural success and adverse events (hypoxia, hypotension, agitation, apnea, gastrointestinal (GI) disturbances, and bradycardia or dysrhythmia) were analyzed. One reviewer checked the articles and extracted the required information. The Cochrane Collaboration’s risk of bias tool (RoB 2)7 was used to assess the methodological quality of the included studies. This tool includes the following criteria: randomization, allocation concealment, blinding, and completeness of follow-up. The risk of bias for each item was graded as high, low, or unclear risk.
Quantitative data synthesis
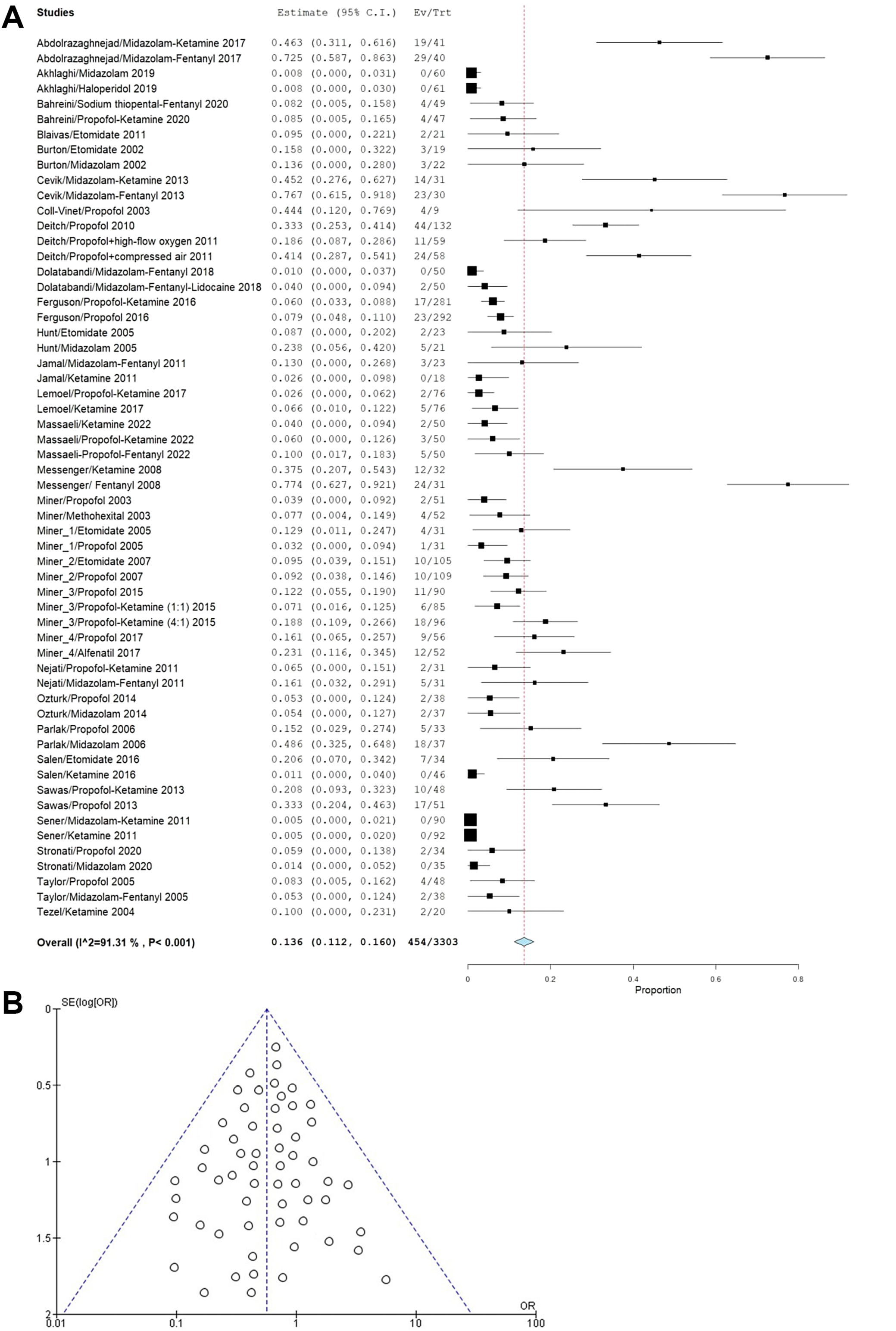
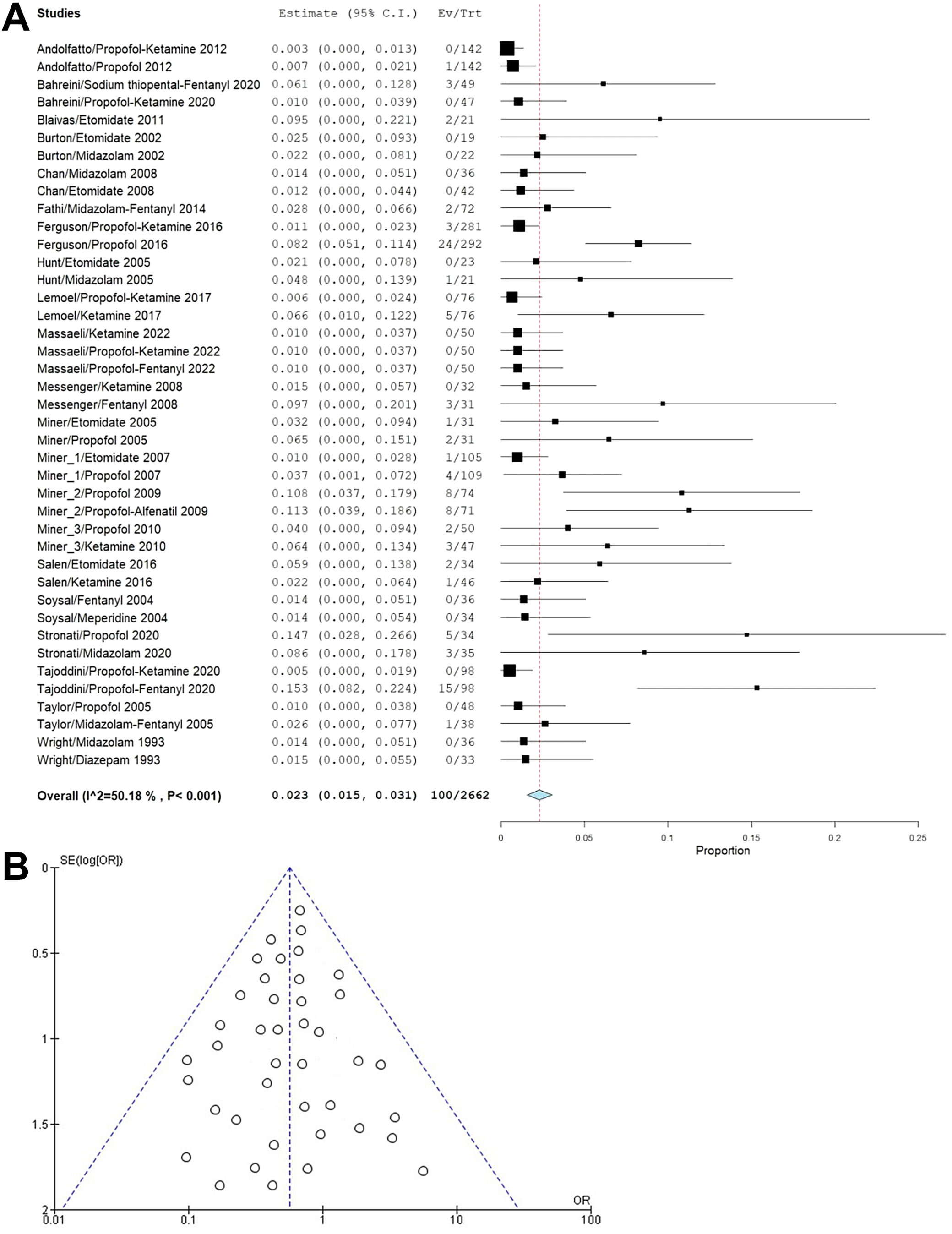
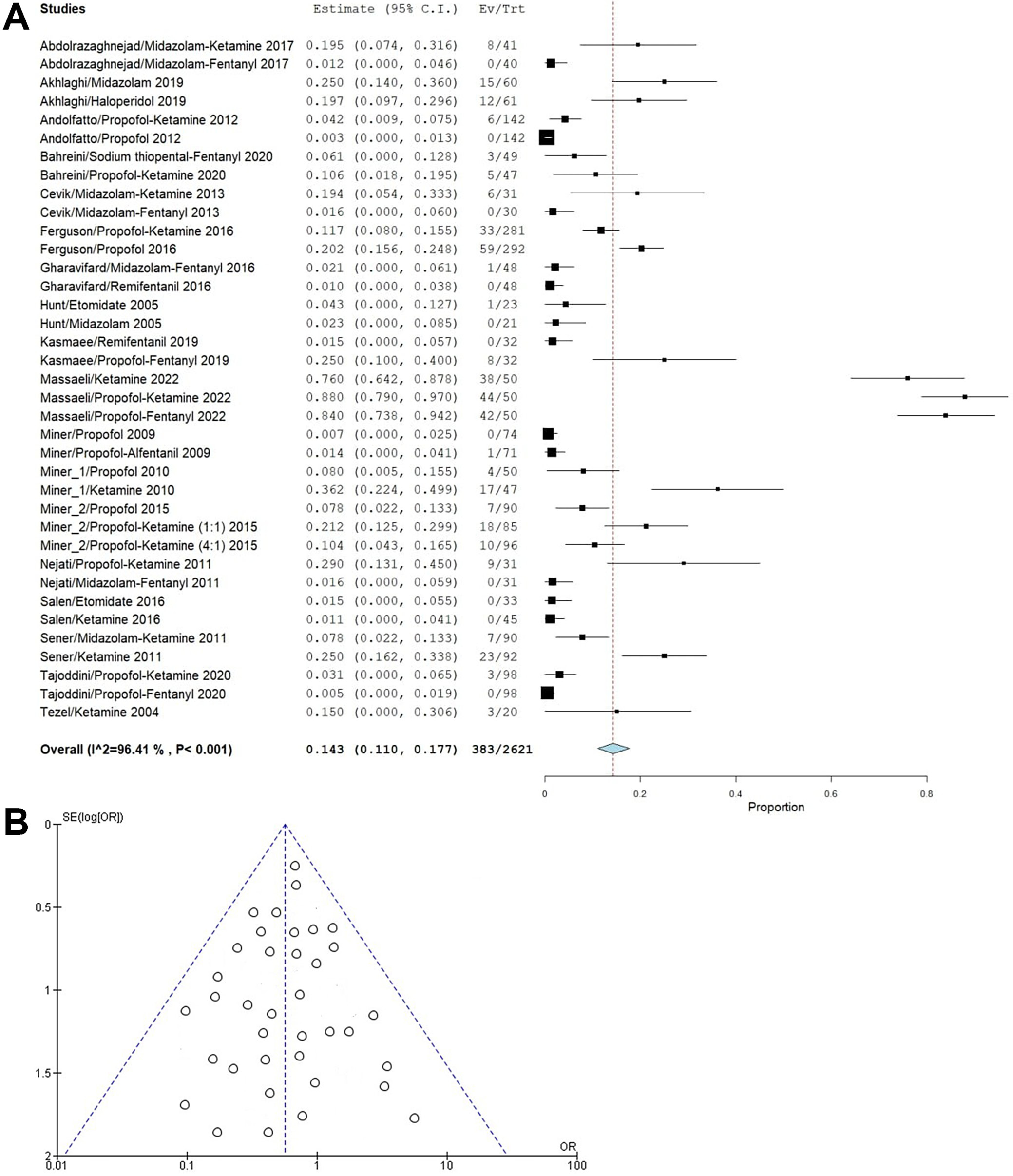
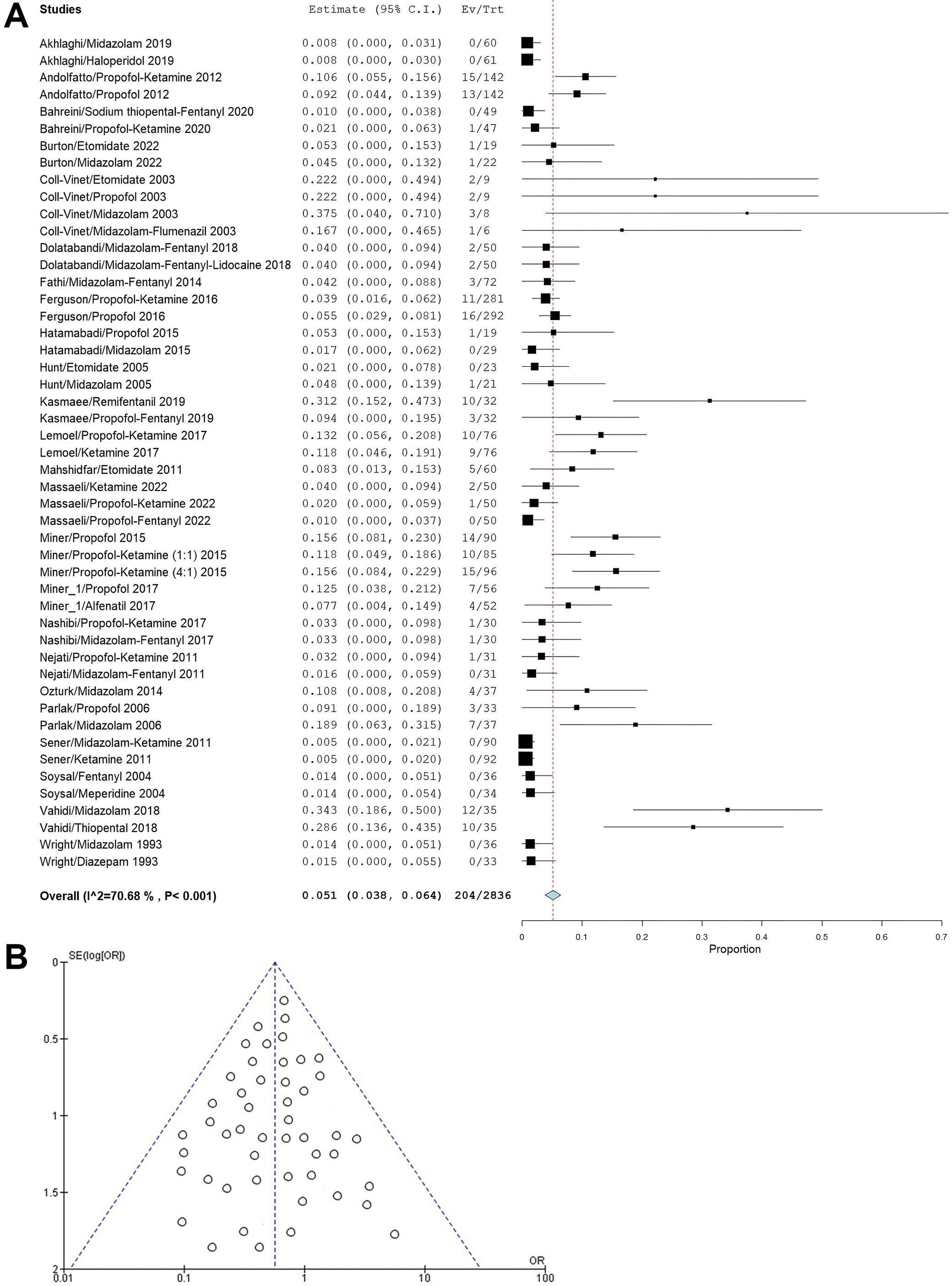
Meta-analysis and statistical calculations were performed using OpenMeta (Analyst) software (https://www.cebm.brown.edu/openmeta) and a random-effects model (DerSimonian–Laird) to calculate the pooled proportion of patients experiencing various adverse effects and procedural success related to the use of procedural sedation medications. The studies included in this single-arm meta-analysis are comparable, as they all evaluate the safety profile of [intervention name] in [patient population], sharing similar study designs, population demographics, and outcome measures. The data from different studies were pooled together to provide pooled estimates for each adverse event based on similarities such as adult patients, ED procedures, and definition of outcomes. Heterogeneity in the included studies was evaluated using the I2 statistic, with small heterogeneity for I2 values <25%, moderate heterogeneity for I2 values of 25–50%, and high heterogeneity for I2 values >50%. Forest plots were constructed and p < 0.05 was statistically significant. Publication bias was assessed with Egger’s test and a funnel plot, in which the log odds ratio (OR) for each study was plotted against its standard error (SE) for the hypoxia outcome.
Results
Identification of studies
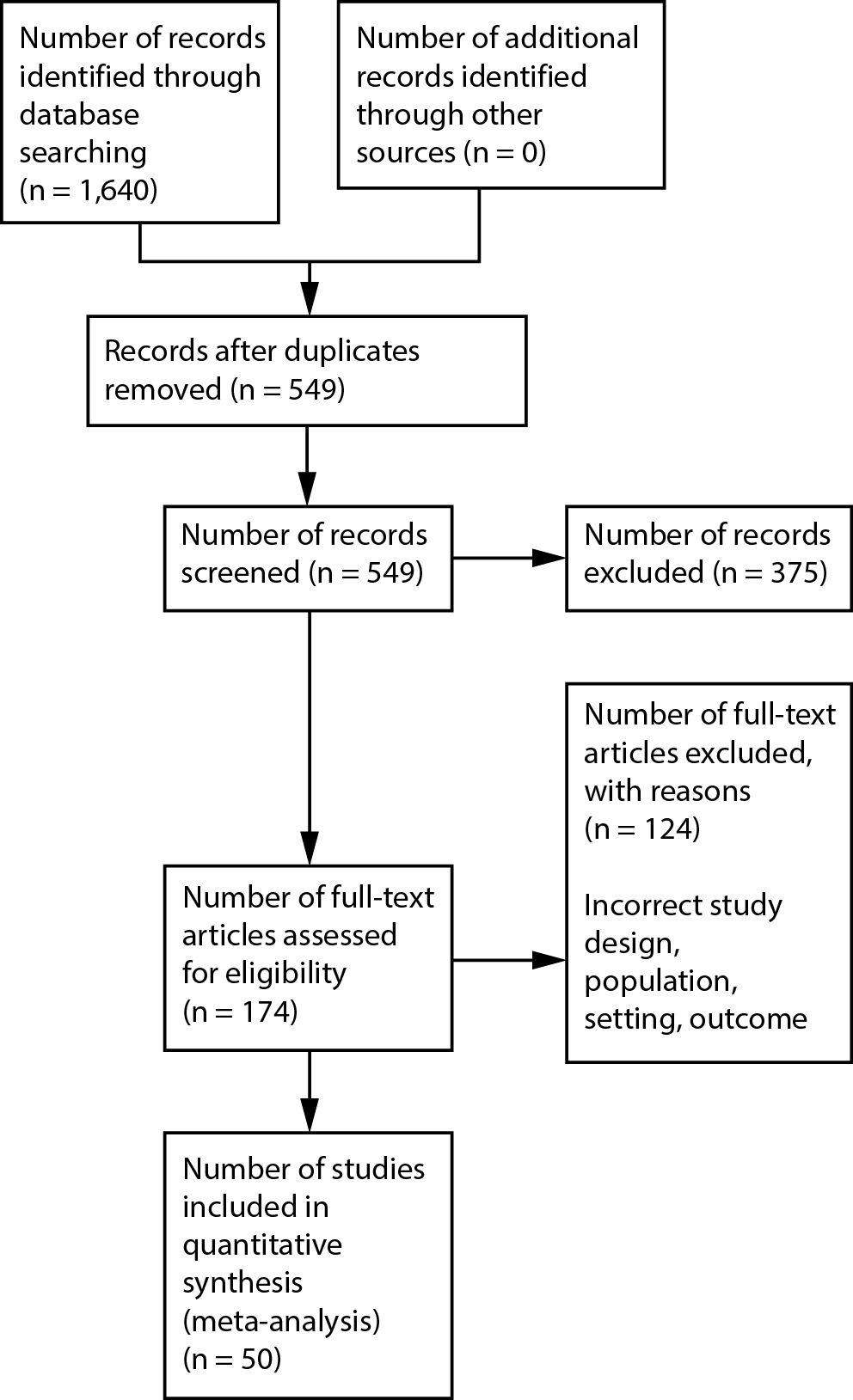
A total of 1,640 records were identified by database searching, of which 549 were screened by title and abstract. Duplicates and irrelevant records were removed (n = 375), and 174 RCTs were assessed for eligibility. However, 124 RCTs were excluded due to reasons such as incorrect study design, inappropriate outcome, setting, and population. The process of selection is shown in Figure 1.
Study characteristics
In total, 50 RCTs totaling 5,398 participants met the inclusion criteria for the meta-analysis. Ketamine, midazolam, propofol, and fentanyl and their combinations were the most frequently used medications for sedation, and some studies also included the use of etomidate, lidocaine, and diazepam. Most procedures for which procedural sedation and analgesia were used were fracture and dislocation reductions, and in some cases cardioversion and abscess incision and drainage. Table 1 shows the details of the various drugs/drug combinations included in each study, along with the procedures that were conducted in the ED for which procedural sedation was used.8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58
Characteristics of participants
All participants in the studies were ≥18 years of age and were trauma patients admitted to the ED for various reasons, of which orthopedic manipulations and abscess incision and drainage procedures were the most common. To be included in the analysis, all procedures were required to be performed by emergency physicians and personnel in the ED and drugs or drug combinations for sedation were administered parenterally.
Bias assessment
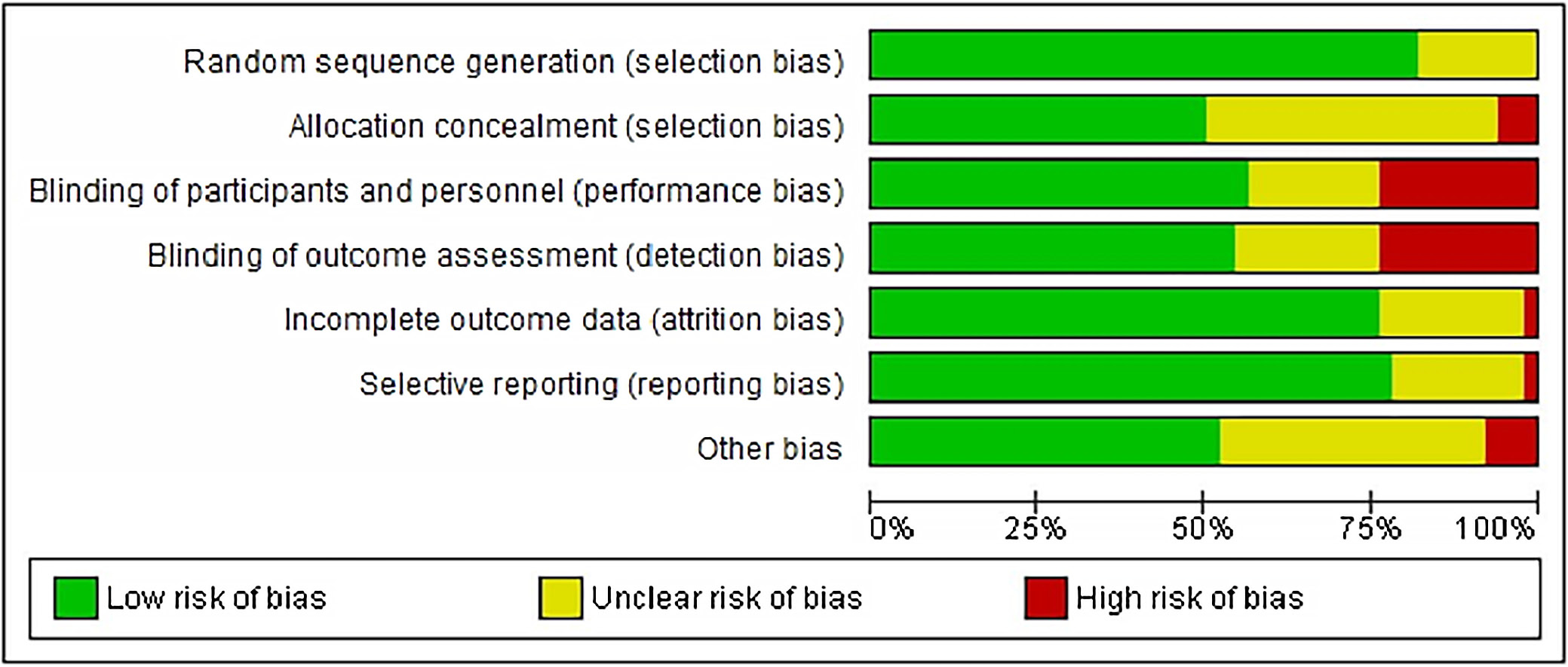
The results of the risk of bias evaluation are shown in Figure 2. It was seen that the risk was low to unclear across most fields but a relatively high risk (>20%) was observed in the categories of blinding of personnel, participants, and outcome assessment. Difficulties in completely masking the medication administered, and side effects associated with the use of certain drugs that physicians and other healthcare personnel involved in the ED are well-versed in, make it difficult to perform blinding.
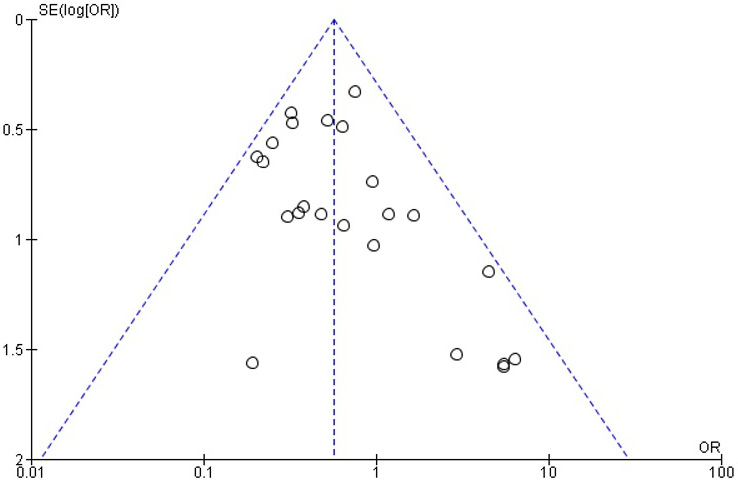
The funnel plot was not symmetrical (Figure 3) for the hypoxia outcome for the use of different drugs and drug combinations, indicating the risk of publication bias.
Meta-analysis results
A total of 50 RCTs comprising 5,398 patients were included in the meta-analysis. The studies spanned from 1993 to 2022, with sample sizes ranging from 20 to 591. The studies evaluated the effects of procedural sedation in adult populations undergoing orthopedic manipulations, cardioversions, abscess incision, drainage procedures, and chest tube placements. Various drugs were used as a single entity such as midazolam (n = 10), propofol (n = 18), ketamine (n = 5), etomidate (n = 9), fentanyl (n = 2), alfentanil (n = 10), methohexital (n = 1), remifentanil (n = 3), meperidine (n = 1), thiopental (n = 2), and diazepam (n = 1). Drug combinations that were used included propofol–fentanyl (n = 4), midazolam–ketamine (n = 5), midazolam–fentanyl (n = 12), propofol–ketamine (n = 11), propofol–alfentanil (n = 2), and remifentanil–propofol (n = 1).
The most reported adverse events that were included in the studies and that were analyzed to determine the pooled proportion (Table 2) are discussed below.
Hypoxia: Of the included studies, 30 reported hypoxia as an adverse outcome. The incidence of hypoxia was 136 per 100 events (95% confidence interval (95% CI): 112–160). Patients receiving midazolam and fentanyl and their combinations showed the highest incidence of hypoxia. The high I2 value of 91.31% was caused by pooling of studies with different drugs/drug combinations and procedures for which sedation was used, along with heterogeneous patient populations in terms of age and previous medications received (Figure 4A). The funnel plot (Figure 4B) exhibited pronounced symmetry across various control treatments, indicating a minimal risk of publication bias. This observation was further validated using Egger’s test, which yielded statistically insignificant p-values (p = 0.217 > 0.05).
Hypotension: This adverse effect was reported in 21 studies, and its incidence was 23 per 1,000 sedations (95% CI: 15–31). Propofol and its combinations were associated with the highest incidence of hypotension events, and moderate heterogeneity of 50.18% was associated with heterogeneous patient characteristics, drug/drug combinations, and procedures (Figure 5A). The funnel plot (Figure 5B) displayed notable symmetry across different control treatments, suggesting a low likelihood of publication bias. This finding was corroborated with Egger’s test, which produced a nonsignificant p-value (p = 0.178), exceeding the conventional significance threshold of 0.05.
Agitation: This adverse outcome was seen in 18 studies and had an incidence of 143 events per 1,000 sedations (95% CI: 110–177). Patients receiving propofol and propofol–ketamine were seen to report high incidences of agitation, and the heterogeneity was high at 96.41% due to pooling of different studies (Figure 6A). The funnel plot (Figure 6B) showed symmetry, indicating minimal publication bias risk, confirmed using Egger’s test, which yielded a nonsignificant p-value (p = 0.135 > 0.05).
Apnea: 24 studies reported apnea as an adverse outcome with an incidence of 51 per 1,000 sedations (95% CI: 38–64). Midazolam administration was related to a relatively high incidence of apnea, and a high heterogeneity of 70.68% was also seen as studies with different drugs were pooled together (Figure 7A). The funnel plot (Figure 7B) revealed a symmetrical pattern across various control treatments, implying a low risk of publication bias. Egger’s test reinforced this conclusion, yielding p = 0.165, which falls above the standard significance level of 0.05.
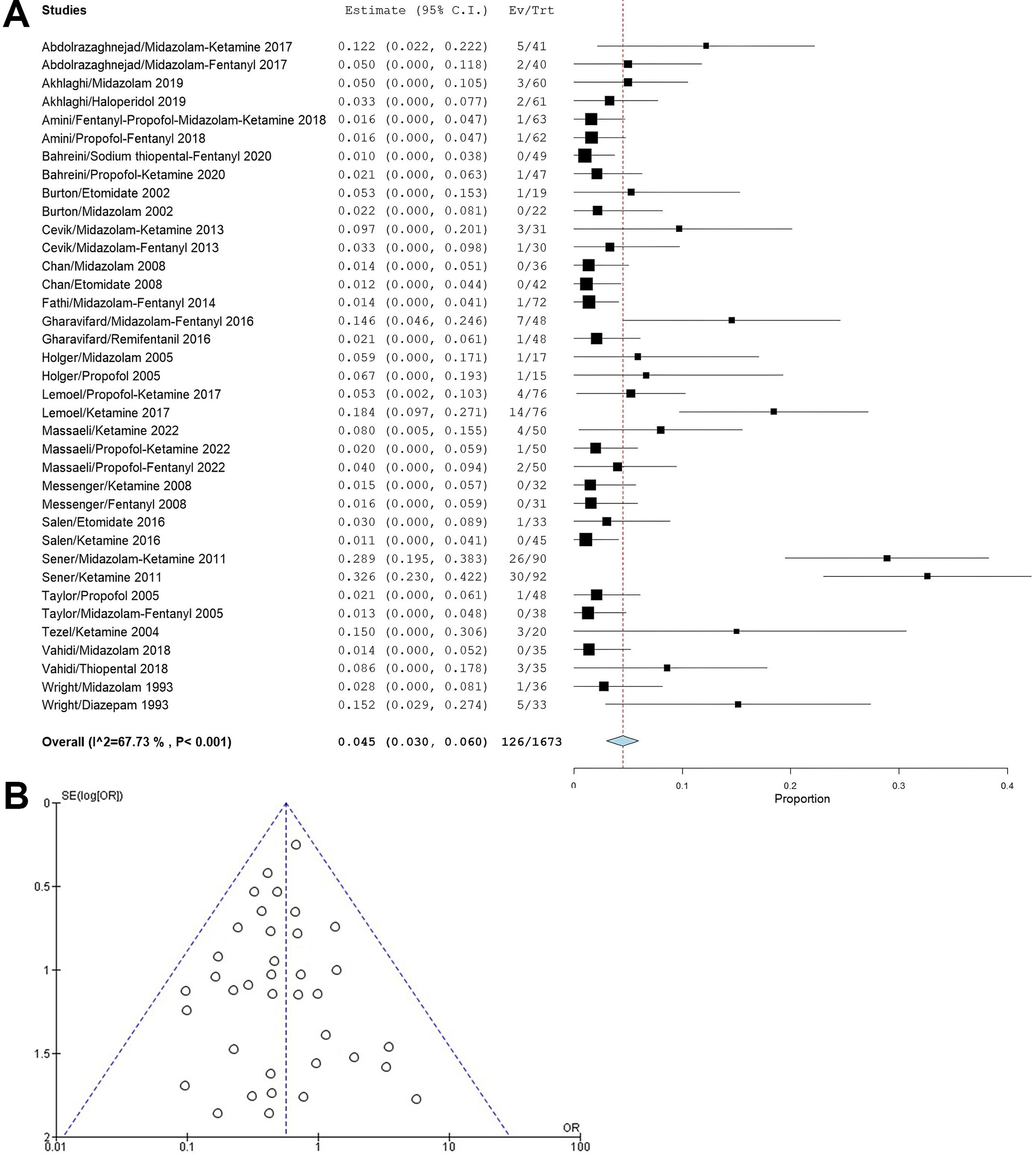
Gastrointestinal disturbances: Nausea and vomiting were the most reported GI disturbances in 19 studies. The pooled incidence was 45 events per 1,000 sedations (95% CI: 30–60). The highest incidence of GI disturbances was seen in patients receiving ketamine, and a high heterogeneity of 67.73% was attributed to combining differing studies (Figure 8A). The symmetry observed in the funnel plot (Figure 8B) suggests that publication bias is unlikely to be a significant concern. This assessment was supported by Egger’s test, which returned a p = 0.298, indicating no statistically significant evidence of bias.
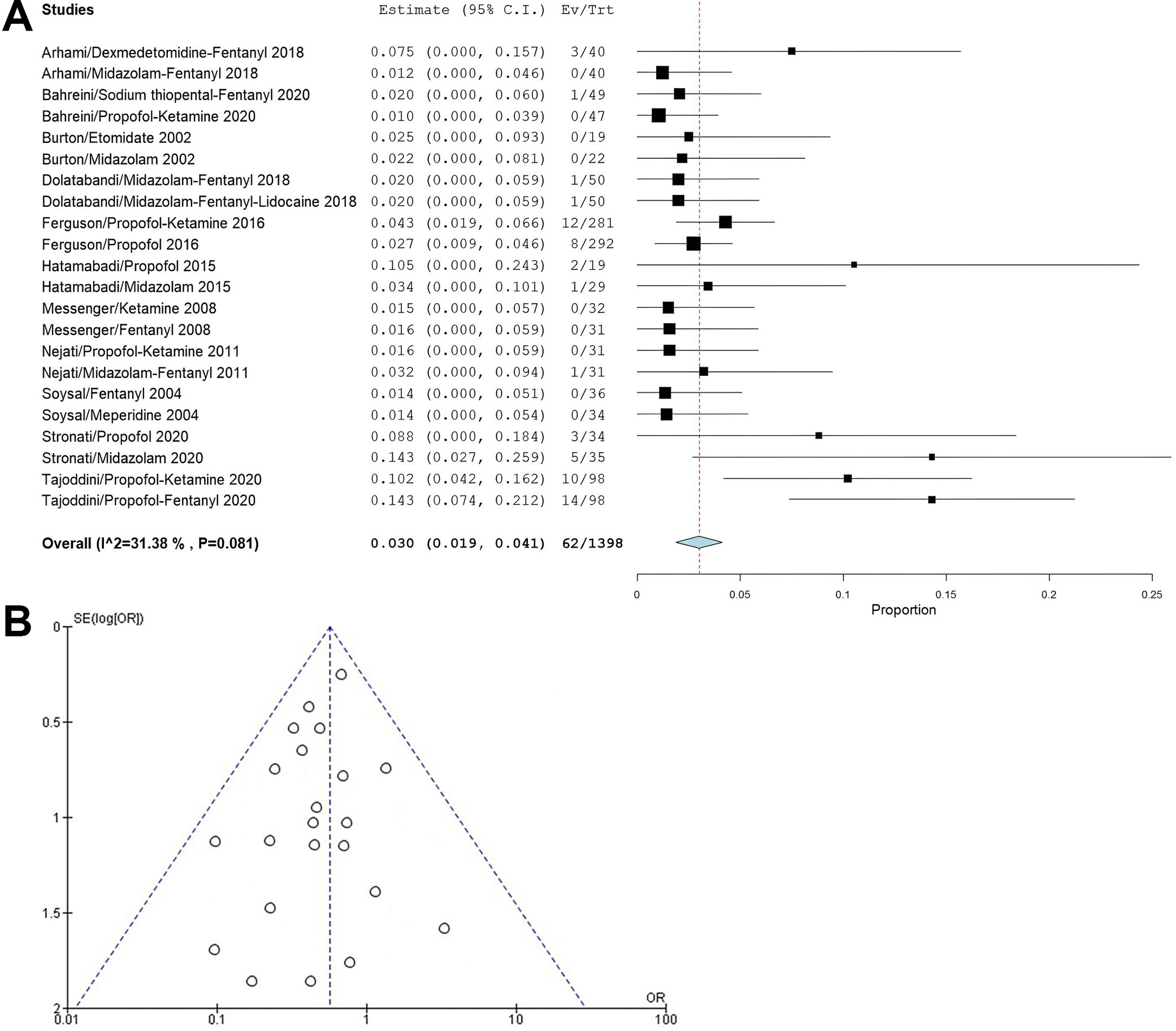
Cardiovascular disturbances: Bradycardia and dysrhythmias were reported in 11 studies in the meta-analysis, with an incidence of 30 per 1,000 sedations (95% CI: 19–41). Propofol and propofol combinations and midazolam caused cardiovascular symptoms. Moderate heterogeneity of 31.38% indicated some homogeneity between the studies chosen (Figure 9A). The funnel plot (Figure 9B) demonstrated symmetry, indicating a low risk of publication bias. The results of Egger’s test (p = 0.251) further substantiated this finding, suggesting that the observed effects are unlikely to be influenced by publication bias.
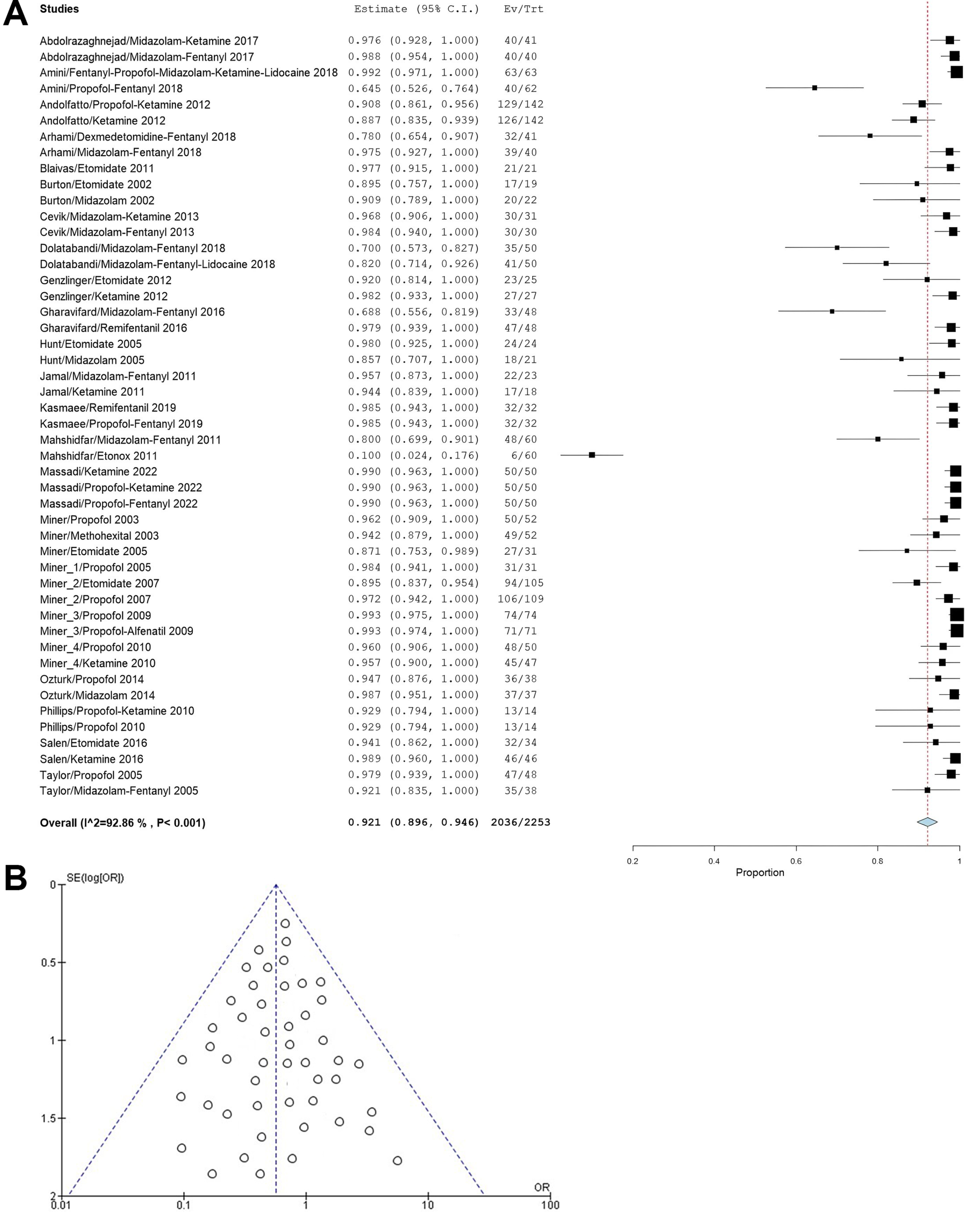
Procedural success: The outcome of procedural success, which can be related to the efficacy of the medications, had a pooled incidence of 921 events per 1,000 sedations (95% CI: 896–946), with only midazolam, fentanyl, and nitrous oxide (Entonox) showing a lower success rate than the pooled incidence estimate (Figure 10A). The symmetrical shape of the funnel plot (Figure 10B) provides evidence against publication bias. This conclusion is reinforced by the nonsignificant result of Egger’s test (p = 0.178), which suggests that the results are robust and unbiased. The p-value for the pooled incidences of all adverse events and procedural success was <0.001, indicating statistically significant pooled incidence estimates for all outcomes (except cardiac disturbances; p = 0.081). Table 3 summarizes the results of the meta-analysis across all outcomes. Supplementary Table 1 demonstrates that the studies incorporated in the meta-analysis included the effect size (incidence rate) of the outcomes.
Discussion
This meta-analysis provides a comprehensive and up-to-date review of the adverse outcomes associated with the use of several drugs and drug combinations administered for procedural sedation in the ED. Most of the procedures included in the analysis were orthopedic manipulations, mainly fracture and joint dislocation reductions. We included 40 randomized controlled trials comparing different drugs, all of which were administered by trained emergency medicine physicians in ED. Since this meta-analysis aimed to determine the pooled incidence of adverse events in patients receiving different drugs, a single-arm meta-analysis was used with the application of a random-effects model to account for differences in studies regarding patient characteristics, settings, and interventions.
The administration of procedural sedation involves a delicate balance of the benefit–risk profile, as analgesics and sedatives are meant to ease procedural comfort but can cause respiratory side effects such as hypoxia and hypoxemia, apnea, hypoventilation, and more serious side effects such as airway obstructions and laryngospasm. Thus, individual patient characteristics need to be considered heavily prior to deciding on a choice of any drug or drug combination to induce sedation. In line with previous meta-analyses, the incidence of respiratory adverse outcomes such as apnea and hypoxia was relatively frequent, with propofol, midazolam and their combinations with fentanyl showing higher incidences of these side effects. Respiratory depression is due to their action on gamma-aminobutyric acid (GABA) receptors, leading to inhibitory neurotransmission.59 Although ketamine is expected to be a relatively safe option for minimizing the incidence of respiratory events, our study showed mixed results, with ketamine and its combinations with other drugs causing a low incidence of hypoxia and apnea in some cases, whereas propofol–ketamine combinations caused a higher incidence than the pooled effect size.
Hypotension and bradycardia are the most common cardiovascular disturbances observed during procedural sedation, and their occurrence is dependent on patient factors such as pre-existing cardiovascular diseases, elderly patients, diabetic neuropathy, and dehydration. Etomidate, midazolam and midazolam–opioid combinations are considered, as they have a neutral cardiovascular profile with minimal hemodynamic effects. These agents and their combinations were associated with a low incidence of hypotension and bradycardia/dysrhythmia in our study. In contrast, propofol was associated with a high risk of cardiovascular adverse effects caused by its cardiac depressant activity that leads to a decrease in cardiac output and arterial pressure.60, 61 To overcome the negative effects of propofol on cardiovascular outcomes, combinations of ketamine–propofol, known as ‘ketofol’, are commonly used. This combination includes the analgesic effect of ketamine and the sedative effect of propofol, and the drugs balance each other out in terms of each one’s adverse effects, thereby resulting in a better safety profile. A RCT has shown that ketofol has better hemodynamic stability than propofol during the first 30 min of general anesthesia.62 Similarly, our meta-analysis also showed that the incidences of hypotension were higher than the pooled incidence of this adverse outcome in patients receiving propofol alone compared to the ketofol combination.
Gastrointestinal side effects such as nausea and vomiting are commonly associated with procedural sedation agents such as ketamine and opioids such as fentanyl. However, ketofol and combinations of opioids with propofol or midazolam resulted in fewer GI disturbances. Neurological adverse events caused by procedural sedation include dizziness, confusion, seizures, and agitation. Ketamine and its combinations were seen to cause high incidences of agitation in our meta-analysis, which is in line with previous studies.63
Procedural success rates were high in our study with all sedation agents irrespective of the adverse outcomes in each trial, highlighting the importance of procedural sedation in emergency procedures. Other meta-analyses on adverse outcomes associated with procedural sedation have included similar events as our study, such as agitation, bradycardia, hypotension, hypoxia, intubation, and aspiration, and concluded that these events were relatively rare and PSA could be used safely.64
Through this meta-analysis, it is clear that the choice of drug that is used for procedural sedation should be dictated by specific clinical scenarios and patient needs. It is important to consider comorbidities, use continuous monitoring and vigilance strategies to prevent serious adverse outcomes, and have emergency equipment, medications and resuscitation agents available in case of respiratory distress. Furthermore, recovery times are also a crucial factor that should be taken into account when using a particular agent for specific patient populations and procedure types.
Limitations
Although our meta-analysis includes the most recent trials and information on procedural sedation, it is not without certain limitations. We included all studies that had the adverse event outcomes that we had defined a priori in our study, regardless of the sample size and bias. Additionally, we did not perform subgroup analyses as per the procedure and drug type or combination due to the limited amount of data for each, which may not have been statistically robust. However, pooling data across all studies resulted in high statistical heterogeneity, which was seen in our study. Adverse outcomes such as agitation, hypotension, and hypoxia were not consistently defined across all the studies, which also could contribute to heterogeneity. Since some studies included only a few patients (<50) in most cases, the confidence intervals were wide, decreasing the confidence in the results. Small single-center studies and publication bias could have also influenced the quality and reliability of the data.
To allow for the use of data on procedural sedation agents in emergency medicine scenarios, it is necessary to have results available from larger, multicenter RCTs with a standardized definition of adverse events.
Conclusions
This paper provides a current meta-analysis on trials using procedural sedation agents in the ED and the adverse outcomes and efficacy associated with the use of these agents. Meta-analysis results for each adverse event show that certain agents are safer than others in specific patients and procedures. Whilst midazolam and ketofol administration cause fewer cardiovascular events, ketamine was associated with a high incidence of agitation, and propofol caused respiratory depression and a high incidence of hypotension. Thus, tailoring the choice of the sedative and analgesic agent to patient needs is of prime importance and must be guided by data from clinical trials, and should not be a one-size-fits-all approach.
Supplementary data
The supplementary materials are available at https://doi.org/10.5281/zenodo.14884141. The package contains the following files:
Supplementary Table 1. Effect size (incidence rate) of outcomes for studies included in this meta-analysis.
Data Availability Statement
The datasets supporting the findings of the current study are openly available in Zenodo at https://doi.org/10.5281/zenodo.15757978.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.