Abstract
Background. Inguinal hernia repair ranks among the most prevalent surgical interventions worldwide. Effective postoperative pain management is paramount for enhancing patient comfort, facilitating early mobilization and optimizing recovery outcomes.
Objectives. This study aimed to rigorously evaluate the efficacy of a modified ultrasound-guided (USG) lower abdominal nerve block in conjunction with local anesthetic (LA) injection during inguinal hernia surgery in elderly patients.
Materials and methods. This prospective cohort study included 50 elderly male patients (mean age 71.84 ±5.56 years) who underwent ultrasound-guided transinguinal hernia surgery from May 2022 to June 2023. Patients were divided into the intervention group (n = 28) and the control group (n = 22). Pain scores were assessed using the visual analogue scale (VAS) multiple times as initial, during and within the first 24 h post-surgery. Secondary outcomes, encompassing vital signs, analgesic consumption and overall patient satisfaction, were also monitored.
Results. The mean age of the cohort was 71.84 ±5.56 years, with a mean body mass index (BMI) of 24.96 ±1.59 kg/m2. A substantial proportion of patients (82%) presented with comorbidities, including hypertension and cerebral infarction. The post-surgery VAS score was significantly decreased in both groups in comparison to the pre-group (in the control group: pre vs post Z = −3.494 p = 0.005, in the intervention group: pre vs post Z = −4.373 p ≤ 0.001). The primary VAS scores were at the highest level at admission time in both groups; however, during- and post-VAS scores were significantly lower in the intervention group than in the control group at time points during and after surgery (p < 0.001, U = 610; and p < 0.001, U = 471, respectively).
Conclusions. The implementation of a modified ultrasound-guided lower abdominal nerve block markedly enhances postoperative analgesia, reduces opioid requirements and improves patient satisfaction in elderly patients undergoing transinguinal hernia repair. These findings underscore the importance of effective pain management strategies in this vulnerable population.
Key words: ultrasonography, inguinal hernia, iliohypogastric nerve, ilioinguinal nerve, peripheral nerve block
Background
Hernias occur when a segment of the bowel or peritoneum protrudes through a weakened musculoaponeurotic layer of the abdominal wall.1 Common types include inguinal, umbilical, incisional, femoral, Spigelian, and epigastric hernias.2 In general surgery, inguinal hernia repair is the most frequently performed procedure, with more than 20 million patients treated worldwide each year.3 This reflects the high lifetime risk of developing an inguinal hernia, estimated at approx. 27% in men and 3% in women.4 In particular, a high prevalence of inguinal hernia is observed in the elderly population, likely due to factors common in individuals over 65 years, including weakening of the abdominal wall, benign prostatic hyperplasia, chronic cough, and chronic constipation.5
Several techniques are used to repair inguinal hernias, ranging from open surgery to laparoscopic approaches. Hernia repair procedures are categorized into 3 main types: herniotomy (removal of the hernial sac only), herniorrhaphy (herniotomy with repair of the posterior wall of the inguinal canal) and hernioplasty (herniotomy with reinforcement of the posterior wall using a synthetic mesh).
Transinguinal hernia surgery is a common procedure that is often associated with significant postoperative pain, which may hinder recovery and reduce patient satisfaction, thereby necessitating effective analgesic strategies. In conventional surgery, adequate postoperative analgesia is crucial for improving recovery outcomes by minimizing postoperative pain, facilitating early recovery and potentially reducing the risk of recurrence.6 The type of anesthesia also influences surgical outcomes, as physiological responses vary depending on whether general, regional or local anesthesia (LA) is used.7 While both laparoscopic and open surgical approaches are recommended for inguinal hernia repair, laparoscopic surgery is associated with lower rates of postoperative infection, shorter hospital stays and less postoperative pain.3 Although spinal anesthesia is commonly used, it carries a significant risk of postoperative hypotension due to its effects on peripheral vascular resistance.8
Ultrasound-guided (USG) hernioplasty involves the use of USG imaging to enhance the precision of hernia repair. The use of USG-guided nerve blocks is becoming increasingly common,9 providing effective analgesia during and after surgery while avoiding the risks associated with general or neuraxial anesthesia.10 In lower abdominal surgery, the transversus abdominis plane (TAP) block is commonly employed to manage both acute and chronic postoperative pain. Its combination with USG guidance has gained popularity due to improved effectiveness and reliability.11 In inguinal surgery, the ilioinguinal and iliohypogastric nerve (IIN/IHN) block is a mainstay of postoperative analgesia. The USG-guided approach significantly enhances the success rate of effective nerve blocks.12 Moreover, combining USG-guided IIN/IHN and TAP blocks has been suggested to improve the efficacy and duration of both intraoperative and postoperative analgesia.13, 14
Objectives
As outlined above, the modified USG lower abdominal nerve block represents a promising technique for targeted analgesia. Therefore, this study aims to investigate the efficacy of a modified USG lower abdominal nerve block combined with LA injection in reducing postoperative visual analogue scale (VAS) pain scores and analgesic consumption in patients undergoing transinguinal hernia surgery.
Materials and methods
Study design and participants
This study included a cohort of 50 elderly male patients who underwent USG-guided transinguinal hernia surgery at our institution between January 2023 and January 2024. The study was conducted in accordance with the Declaration of Helsinki, and all research procedures were approved by the Ethics Committee of Shandong Provincial Third Hospital (Jinan, China; approval No. 201901). Written informed consent was obtained from all participants.
Throughout the study, patients’ vital signs – including blood pressure, heart rate (HR), respiratory rate (RR), and oxygen saturation – were continuously monitored and maintained within normal ranges for 24 h postoperatively. All surgical procedures were standardized and performed by experienced surgeons using a consistent technique.
Inclusion and exclusion criteria
The inclusion criteria were as follows: 1) age ≥60 years; 2) surgery performed between January 2023 and January 2024; 3) ability to communicate normally; 4) undergoing USG-guided IIN/IHN block-assisted hernia surgery; and 5) normal vital signs prior to the procedure.
The exclusion criteria were as follows: 1) preoperative hemodynamic instability; 2) incomplete medical records; 3) hernioplasty performed without USG guidance; and 4) transfer to another hospital during treatment.
Anesthetic technique
Upon arrival in the operating room, peripheral intravenous access was established, and patients received premedication with midazolam (1 mg) and sufentanil (5 μg) administered intravenously. Supplemental oxygen at 2 L/min was delivered via nasal cannula, and continuous electrocardiographic (ECG) monitoring, invasive blood pressure measurement, pulse oximetry, and standard vital sign monitoring were implemented. Arterial blood gas analysis was performed prior to the administration of anesthesia. The USG system used was a Mindray M7 color Doppler USG device (Shenzhen Mindray Bio-Medical Electronics, Shenzhen, China), and a UniPlex NanoLine® needle (80 mm, 22 G; Pajunk, Geisingen, Germany) was used for the administration of LA.
Patients were positioned in the supine position, and strict aseptic techniques were observed. A high-frequency (6–12 MHz) linear USG transducer was placed along a line extending from the anterior superior iliac spine (ASIS) to the umbilicus. A sterile transducer sheath (Vygon, Écouen, France) and sterile USG gel were used. The ilioinguinal nerve was identified between the internal oblique and transversus abdominis (or external oblique) muscles, approx. 1–3 cm medial to the ASIS, with the iliohypogastric nerve located in close proximity. A total volume of 35 mL of a solution containing 1% lidocaine and 0.375% ropivacaine was administered.
For the umbilical plane nerve block, a total of 20 mL of the same anesthetic combination was injected: 10 mL at 1/3 of the distance from the ASIS to the umbilicus, 5 mL near the lateral edge of the USG transducer, and 5 mL between the extraperitoneal space and the transversus abdominis muscle at the level of the umbilicus. Correct needle placement was confirmed by visualization of LA spread creating a hypoechoic plane between the internal oblique and transversus abdominis muscles. The IIN/IHN appeared as distinct hypoechoic structures surrounded by adipose tissue.
A continuous intravenous infusion of dexmedetomidine was initiated at the end of the anesthetic procedure and maintained for 15–20 min at a rate of 1 mL/kg/h, after which it was reduced to 0.1 mL/kg/h. The infusion was discontinued 15–20 min before the end of the procedure. Postoperative analgesia was administered as needed and typically consisted of morphine (2–4 mg intravenously every 2–4 h), tramadol (50–100 mg orally every 4–6 h) or oxycodone (5–10 mg orally every 4–6 h).
Visual analogue scale pain scores (0 = no pain; 10 = worst possible pain) were recorded at 4 time points: immediately after skin incision, during spermatic cord or round ligament traction, during sac ligation, and 24 h postoperatively.
Statistical analyses
The study included 50 elderly male participants, all aged ≥60 years, who met the inclusion criteria. Participants were allocated to 2 groups: the intervention group (n = 28) and the control group (n = 22). Data analysis was performed using IBM SPSS Statistics v. 24.0 (IBM Corp., Armonk, USA) and Microsoft Excel 2013 (Microsoft Corp., Redmond, USA).
The normality of data distributions was assessed using the Shapiro–Wilk test. Normally distributed continuous variables were reported as the mean ± standard deviation (SD),15 whereas categorical variables were presented as frequencies and percentages. Visual analogue scale scores, as ordinal data, were expressed as the minimum, maximum and median (Q1–Q3). Student’s t-test was used to compare demographic and baseline characteristics, including age, weight, height, body mass index (BMI), systolic blood pressure (SBP), diastolic blood pressure (DBP), and HR, between groups. No correction for multiple comparisons was applied due to the exploratory nature of the study.
Pain scores at different surgical stages were analyzed using repeated-measures analysis. Friedman’s test was applied to assess within-group differences before, during and after surgery. Post hoc pairwise comparisons were performed using the Wilcoxon signed-rank test with Bonferroni correction (preoperative vs intraoperative, preoperative vs postoperative, and intraoperative vs postoperative). Results were reported using χ2 statistics, degrees of freedom, z values, and p-values. Between-group VAS scores at each time point were compared using the Mann–Whitney U test. A p < 0.05 was considered statistically significant.
Results
Clinical baseline data
A total of 50 elderly male patients underwent transinguinal hernia surgery during the study period. Participants were allocated to 2 groups: the intervention group (n = 28) and the control group (n = 22). The mean age was 71.84 ±5.56 years, with a mean body weight of 71.80 ±5.07 kg and a mean BMI of 24.96 ±1.59 kg/m2. The majority of patients (82%) had comorbidities, including cerebral hemorrhage (6%), hypertension (34%) and cerebral infarction (16%); only 18% (n = 9) had no underlying conditions.
Baseline clinical characteristics, including age, sex, BMI, and comorbidities, were comparable between the intervention and control groups, as shown in Table 1. Independent t-tests were used for between-group comparisons, and no statistically significant differences were observed (all p > 0.05). The comparability of baseline characteristics reduces the risk of bias and strengthens the validity of subsequent between-group comparisons.
Vital signs
As shown in Table 2, vital signs – including SBP, DBP, HR, RR, and O2 saturation – remained stable throughout the study. Prior to anesthesia, the mean SBP, DBP and HR in the intervention group were 136.09 ±17.74 mm Hg, 78.95 ±6.17 mm Hg and 74.72 ±6.89 bpm, respectively. These values did not differ significantly from those in the control group, which showed mean SBP, DBP and HR values of 136.14 ±19.99 mm Hg, 77.10 ±8.44 mm Hg, and 76.14 ±6.80 bpm, respectively. No significant between-group differences were observed for other vital parameters (p > 0.05). All variables were continuously monitored during and after surgery.
Evaluation of pain score
All patients received LA (ropivacaine and lidocaine) during surgery. Sensory block assessment performed 30 min after block administration confirmed that all patients achieved a block grade of 2 or higher, indicating effective anesthesia.
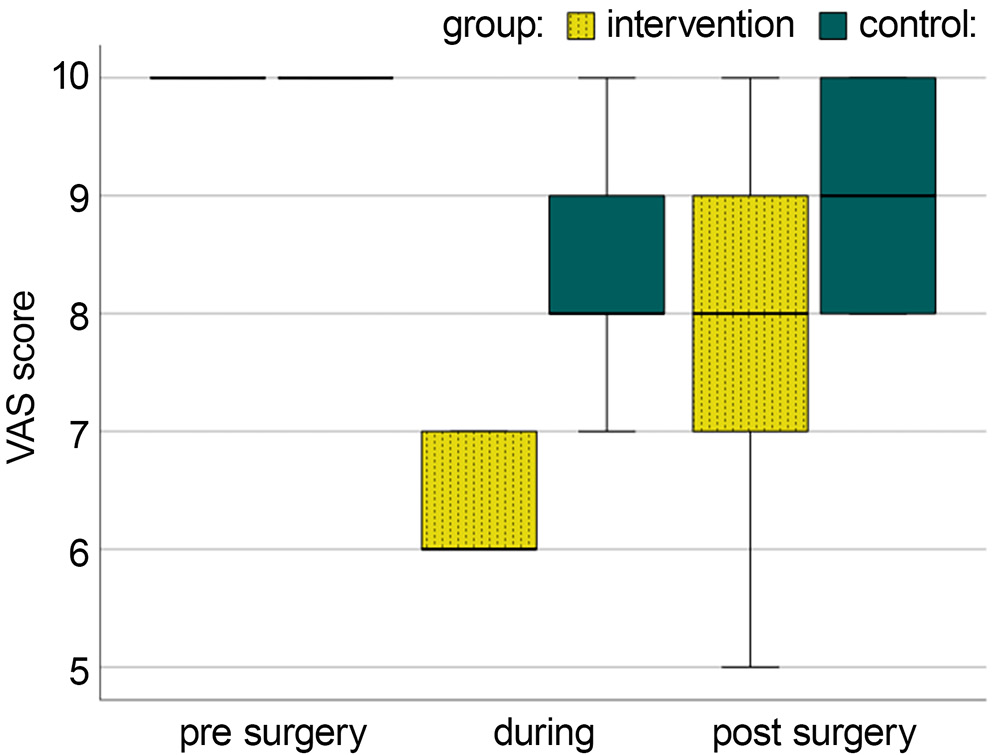
Visual analogue scale scores were recorded at multiple time points: immediately after skin incision, during spermatic cord or round ligament traction, during sac ligation, and 24 h postoperatively. Figure 1 illustrates the mean VAS scores.
Pain score comparisons were performed using repeated-measures analysis with Friedman’s test, followed by post hoc Wilcoxon signed-rank tests with Bonferroni correction (significance threshold: p < 0.017). Friedman’s test demonstrated a statistically significant reduction in pain scores across time points in both groups (control group: χ2 = 12.861, p = 0.002; intervention group: χ2 = 49.078, p < 0.001).
Post hoc pairwise comparisons demonstrated a significant reduction in VAS scores from the preoperative to intraoperative stages in both groups (control group: z = −3.255, p = 0.001; intervention group: z = −4.774, p < 0.001). A significant decrease from the preoperative to postoperative stages was also observed in both groups (control group: z = −3.494, p = 0.005; intervention group: z = −4.373, p < 0.001). A significant difference between intraoperative and postoperative VAS scores was observed only in the intervention group (z = −4.159, p < 0.001), whereas no significant difference was found in the control group (z = −1.768, p = 0.077).
Between-group comparisons using the Mann–Whitney U test demonstrated that VAS scores were significantly lower in the intervention group during both the intraoperative and postoperative periods (p < 0.001; U = 601 and U = 471, respectively), as shown in Table 3 and Figure 1.
With regard to postoperative analgesic consumption, 21 patients (42%) required no additional analgesia, 14 patients (28%) required 1 dose, 14 patients (28%) required 2 doses, and 1 patient (2%) required 3 doses.
Discussion
Inguinal hernia repair is one of the most commonly performed surgical procedures, with many operations carried out on a day-care basis.16 Despite being considered relatively minor, these procedures are frequently associated with significant postoperative pain, which, if inadequately managed, may progress to chronic pain.16, 17 Depending on the anatomical distribution of the IIN/IHN, hernia repair may result in parietal pain.18 Both IIN/IHN blocks and TAP blocks have previously been used, with varying degrees of success, to alleviate postoperative pain in these patients.16 Although the IIN/IHN block is often considered a relatively straightforward technique based on surface anatomy and skin landmarks, anatomical variations may render accurate nerve localization challenging.19 In contrast, USG-guided IIN/IHN or TAP blocks enable precise administration of LA, with real-time confirmation of anesthetic spread under USG visualization.20 However, compared with the IIN/IHN block, the TAP block appears to be associated with a higher incidence of urinary retention,21 primarily due to the larger volumes of LA required.
This study evaluated the efficacy of a modified USG-guided lower abdominal nerve block in 50 elderly patients undergoing transinguinal hernia surgery. Within 30 min, all patients achieved a nerve block grade of 2 or higher, as confirmed by cutaneous sensory testing in the inguinal region. These findings indicate that achieving effective analgesia and anesthesia in patients undergoing inguinal hernia repair using an USG-guided IIN/IHN block is feasible and reliable. Similarly, previous studies have demonstrated that the use of USG guidance increases the success rate of IIN/IHN blocks while requiring lower volumes of LA and resulting in shorter onset times.19 Ultrasound guidance enhances the accuracy of nerve localization, resulting in higher success rates of nerve blockade. Traditional landmark-based (blind) techniques typically achieve success rates of approx. 70–80%, whereas USG-guided techniques have been reported to reach success rates of up to 95%.22, 23 Compared with TAP block, Faiz et al. reported that USG-guided IIN/IHN block provides superior analgesia while requiring a smaller volume of LA, as the anesthetic is deposited directly around the target nerves.24
Conversely, another study reported that TAP blocks administered with lower concentrations of LA were associated with an earlier requirement for morphine.25 This finding suggests that lower LA concentrations in TAP blocks are linked to increased postoperative opioid requirements.25, 26 Similarly, the present study demonstrated a low requirement for postoperative analgesics during the first 24 h after surgery. Furthermore, a recent systematic review and meta-analysis showed that USG-guided IIN/IHN blocks are superior to landmark-based techniques in providing perioperative analgesia.27
Multiple studies have demonstrated that the use of USG-guided IIN/IHN blocks results in a significant and clinically meaningful reduction in postoperative pain.12, 13, 24, 28 In a study by Elwany et al., USG guidance was shown to improve the accuracy of nerve localization, thereby increasing the success rate of effective analgesia.29 Additional studies have reported that patients receiving USG-guided IIN/IHN blocks experience lower postoperative pain scores compared with those receiving other forms of analgesia, such as TAP block. For example, one study found that the mean time to rescue analgesia was significantly longer in patients receiving IIN/IHN blocks, indicating superior pain control with this technique.13 Pain scores recorded in the present study also demonstrated significant reductions at multiple time points. Comparisons between intraoperative and postoperative pain scores revealed significant differences, with both being lower than preoperative scores. Similarly, one study demonstrated that patients who received an USG-guided IIN/IHN block experienced significantly less postoperative pain at rest, with pain scores progressively declining over 12 h and becoming negligible 24 h after block administration.24 The use of USG-guided IIN/IHN blocks has been shown to reduce pain in day-surgery settings and decrease the need for rescue analgesics, as reported in a meta-analysis by Wang et al.27 This benefit is most likely attributable to improved needle positioning and precise deposition of LA around the IIN/IHN nerves.27 Furthermore, both Bærentzen et al. and Frassanito et al. recommend incorporating USG-guided IIN/IHN blocks into a multimodal analgesic regimen to achieve more effective acute postoperative pain management in adult patients.12, 30 Although no complications were observed in the present study, potential risks associated with these techniques remain, including vascular injury,31 inadvertent puncture of intraperitoneal viscera32 and unintended extension of the block to the femoral nerve.33, 34 The use of USG guidance substantially mitigates these risks by enabling real-time visualization of anatomical structures and needle trajectory.
Limitations
The present study has several limitations that may affect the generalizability of the findings and should be considered when interpreting the results. One of the primary limitations is the relatively small sample size, which may have reduced the robustness and statistical power of the analyses. Future studies with larger cohorts are therefore warranted to enhance the reliability and validity of the findings.
The assessment of postoperative pain in this study was limited to a single day. Although this short observation period provides insight into the immediate postoperative phase, it does not capture longer-term pain trajectories that many patients may experience following surgery. Postoperative pain is a multifactorial phenomenon that can evolve over time and is influenced by factors such as tissue healing, psychological state and individual pain sensitivity. By focusing exclusively on the first 24 h after surgery, this study may have overlooked important information regarding the progression or resolution of pain in the days and weeks thereafter.
The findings demonstrated a significant reduction in analgesic requirements, with effective analgesia achieved using lower doses of LA. However, the limited study duration was insufficient to provide definitive evidence regarding the long-term impact of this technique on postoperative pain management. Studies incorporating longer follow-up periods would provide a more comprehensive understanding of postoperative pain trajectories. In addition, a larger sample size combined with extended follow-up would further strengthen the robustness and generalizability of the results.
The study did not include between-group comparisons for several key procedural outcomes, such as the onset time of anesthesia, overall analgesic consumption and detailed pain management parameters. In addition, because pain rating scales are inherently subjective, interindividual variability in pain perception and reporting may have influenced the results.
Another potential limitation of this study is the exclusive inclusion of male patients with inguinal hernia. Owing to biological differences, outcomes in female patients may differ with respect to pain perception, response to analgesic interventions and postoperative recovery. However, this limitation reflects the epidemiological reality rather than a design flaw, as inguinal hernia occurs far more frequently in men than in women. Nevertheless, future studies should aim for a more balanced sex distribution to enhance the generalizability of the findings to the broader patient population.
Another important limitation of this study is its single-center design. This characteristic may limit the generalizability of the findings, as results obtained in a single institution may not be representative of outcomes across different healthcare settings or patient populations. Variations in surgical techniques, patient demographics and institutional protocols could contribute to differences in postoperative pain management outcomes.
A further critical limitation of this study is the absence of a comparison group. Without a control group or an alternative treatment group, it is difficult to draw definitive conclusions regarding the effectiveness of the modified nerve block technique. Future studies should therefore incorporate appropriate control groups to enable a more rigorous evaluation and validation of the intervention’s efficacy.
Finally, this study assessed only pain at rest, measured at the bedside. Future studies with larger sample sizes and appropriate control groups should also evaluate pain during movement and daily activities to provide a more comprehensive assessment of postoperative recovery.
Conclusions
The present study demonstrates that USG-guided IIN/IHN blocks in elderly patients undergoing inguinal hernia repair provide a clinically meaningful and statistically significant reduction in intraoperative and early postoperative pain. This technique was associated with an earlier onset of sensory block and reduced LA requirements. However, owing to the short observation period of 24 h, no conclusions can be drawn regarding longer-term outcomes or patient satisfaction. Further large-scale, multicenter studies with extended follow-up are warranted to confirm the efficacy and safety of this technique.
Data Availability Statement
The datasets supporting the findings of the current study are openly available in Zenodo at https://zenodo.org/records/13950171.
Consent for publication
Not applicable.
Use of AI and AI-assisted technologies
Not applicable.