Abstract
Background. Oral squamous cell carcinoma (OSCC) is one of the most common cancers in humans. The role of neoadjuvant chemotherapy (NAC) in OSCC remains controversial.
Objectives. The study aimed to investigate the effect of NAC on locally advanced OSCC and identify prognostic factors varying is different therapies to ultimately guide the optimal selection of future treatment.
Materials and methods. A total of 156 patients with locally advanced OSCC were enrolled. The clinical characteristics and survival outcomes of patients with and without NAC were compared. The primary endpoint was overall survival (OS), and the secondary endpoint was disease-free survival (DFS).
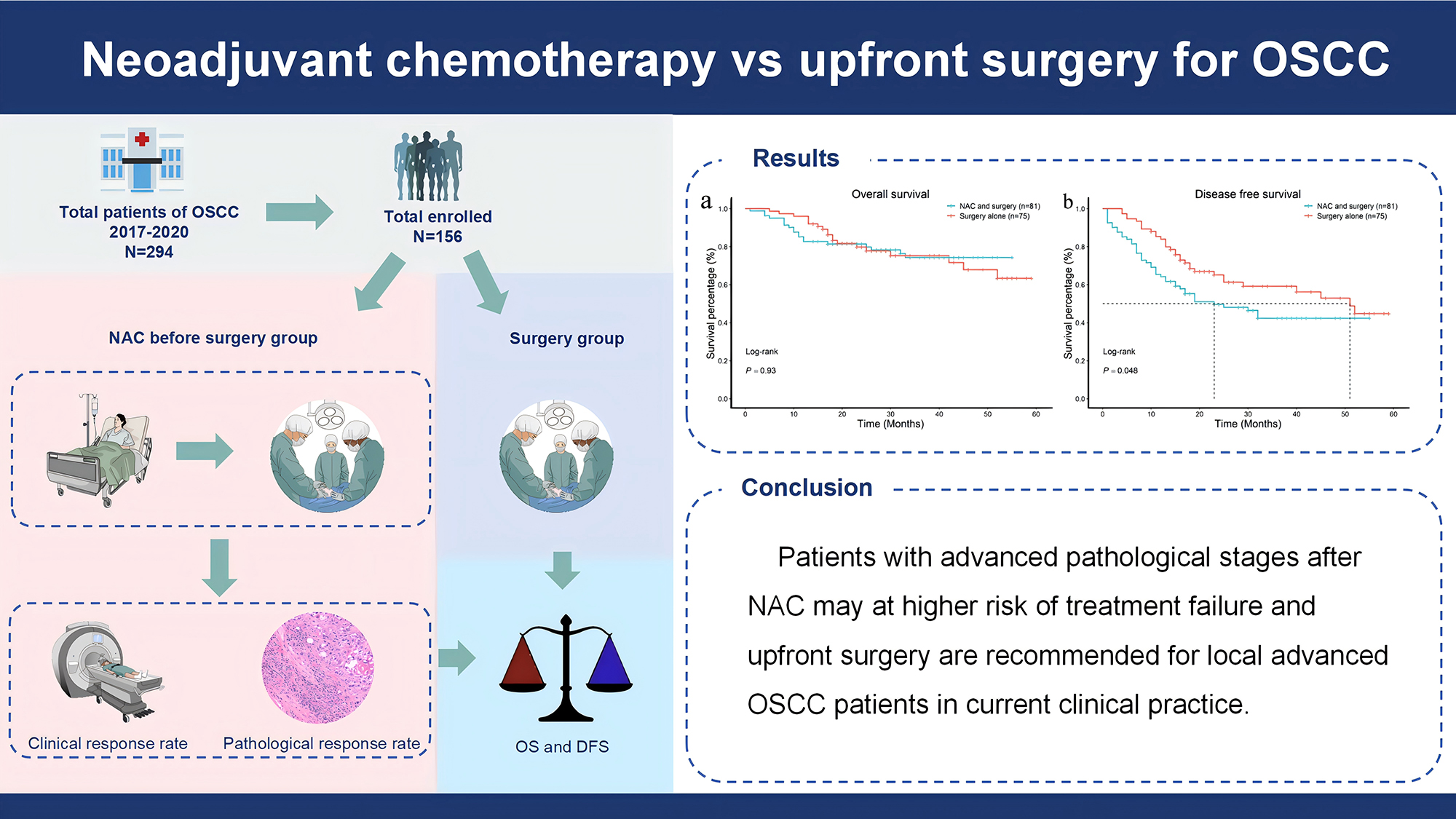
Results. Among the 156 patients enrolled in this study, 81 patients received NAC followed by surgery and 75 patients received surgery alone. No significant difference in OS at 3 years was detected (78.3% vs 79.8%, p = 0.76). However, a significantly worse DFS was observed in the NAC group (42.4% vs 59.2%, p = 0.048). Within the NAC group, 50 patients (61.7%) had a favorable clinical response, and 12 patients (14.8%) had a complete pathological response. Better survival outcomes were observed in patients with favorable clinical responses. In stratified analysis, patients of pT3/4 OSCC after NAC showed worse DFS than those of the same stage who underwent surgery alone (40.2% vs 58%, p = 0.033). In Cox regression, clinical response and pathological stage were predictors of survival in the NAC group, while pathological stage was the only predictor of OS in the surgery group.
Conclusions. Patients with advanced pathological stages after NAC may be at a higher risk of treatment failure, and upfront surgery is recommended for locally advanced OSCC patients in current clinical practice.
Key words: prognosis, neoadjuvant chemotherapy, oral squamous cell carcinoma
Background
Oral cancer represents one of the most common malignancies. There were approx. 373,097 (95% uncertainty interval (95% UI): 340,884–403,865) incident cases and 199,379 (95% UI: 181,651–218,058) deaths worldwide in 2019, according to the national epidemiological profiles of cancer burden in the Global Burden of Disease Study (https://ghdx.healthdata.org/ihme_data). The mainstays of oral squamous cell carcinoma (OSCC) therapeutics are surgery, radiotherapy, chemotherapy, or a combination of these modalities, depending on the extent of the disease. Oral squamous cell carcinoma has long been regarded as a favorable indication for primary surgical intervention.1 Advances in surgical techniques over the past 4 decades have resulted in significantly enhanced outcomes. Minimally invasive surgery is widely acknowledged as striking a favorable balance between cancer resection and functional restoration for early-stage OSCC.2 Numerous clinical trials have demonstrated the efficacy of surgical interventions for early-stage OSCC, with ongoing research focusing on the impact of various surgical techniques and extent of resection on the local recurrence rates and preservation of tissue function in early-stage human papilloma virus (HPV)-negative OSCC.3 Compared to non-surgical interventions, oral surgery resection in OSCC has been shown to increase the survival rate in oral cancer.4, 5 A large-scale, 10-year clinical study on borderline oral cavity cancers demonstrated a significant increase in the 10-year survival rate for patients who underwent surgery compared to those who did not. This finding strongly underscores the importance of surgical treatment.6 However, the prognosis for locally advanced OSCC remains grim. The overall and long-term survival rates for patients with OSCC continue to be unsatisfactory, with a 5-year survival rate of approx. 60% across all stages.7 A significant challenge lies in enhancing the survival outcomes of patients with advanced OSCC through the utilization of various chemotherapy regimens in conjunction with surgical interventions, a matter that remains unresolved.8
Neoadjuvant chemotherapy (NAC) has been routinely utilized for several cancers, with the goal of down-staging the primary tumor and controlling potential micrometastasis.9, 10 Neoadjuvant chemotherapy has been introduced for head and neck squamous cell carcinoma (HNSCC), with the goal of function preservation and better survival outcomes. However, several clinical trials have failed to prove the benefits in survival with the addition of chemotherapy prior to chemoradiotherapy or surgery in HNSCC patients.11, 12, 13 Although the superiority of NAC remains controversial, there are some promising results. First, a reduction in the distant metastasis rate was reported in OSCC patients treated with NAC and surgery compared to those who received surgery directly.14 Second, NAC was reported to be a strategy for selecting patients for conservative surgery. A retrospective study of small samples revealed that limited surgery without flap reconstruction was feasible in advanced OSCC patients who had a favorable clinical response to chemotherapy.15 Similarly, another study identified that for patients needing mandibular resection for paramandibular disease without obvious bone erosion, NAC was also a feasible option for mandibular preservation with no compromise of survival.16 Third, specific subsets of patients are likely to benefit from neoadjuvant treatment, especially those who respond well to preoperative chemotherapy.13 A phase II clinical trial of 36 patients demonstrated the safety of pembrolizumab and a decrease in 1-year recurrence rate in patients with locally advanced, HPV-negative HNSCC.17 However, there are still many patients who cannot benefit from NAC combined with immunotherapy, and its effect in oral cancer still needs to be supported by more clinical research data.
Objectives
This study aimed to investigate the effects of NAC and identify the potential clinicopathological risk factors in patients with different treatment regimens and thus guide reasonable therapeutic options in locally advanced OSCC patients for future practice.
Methods
Patients and study design
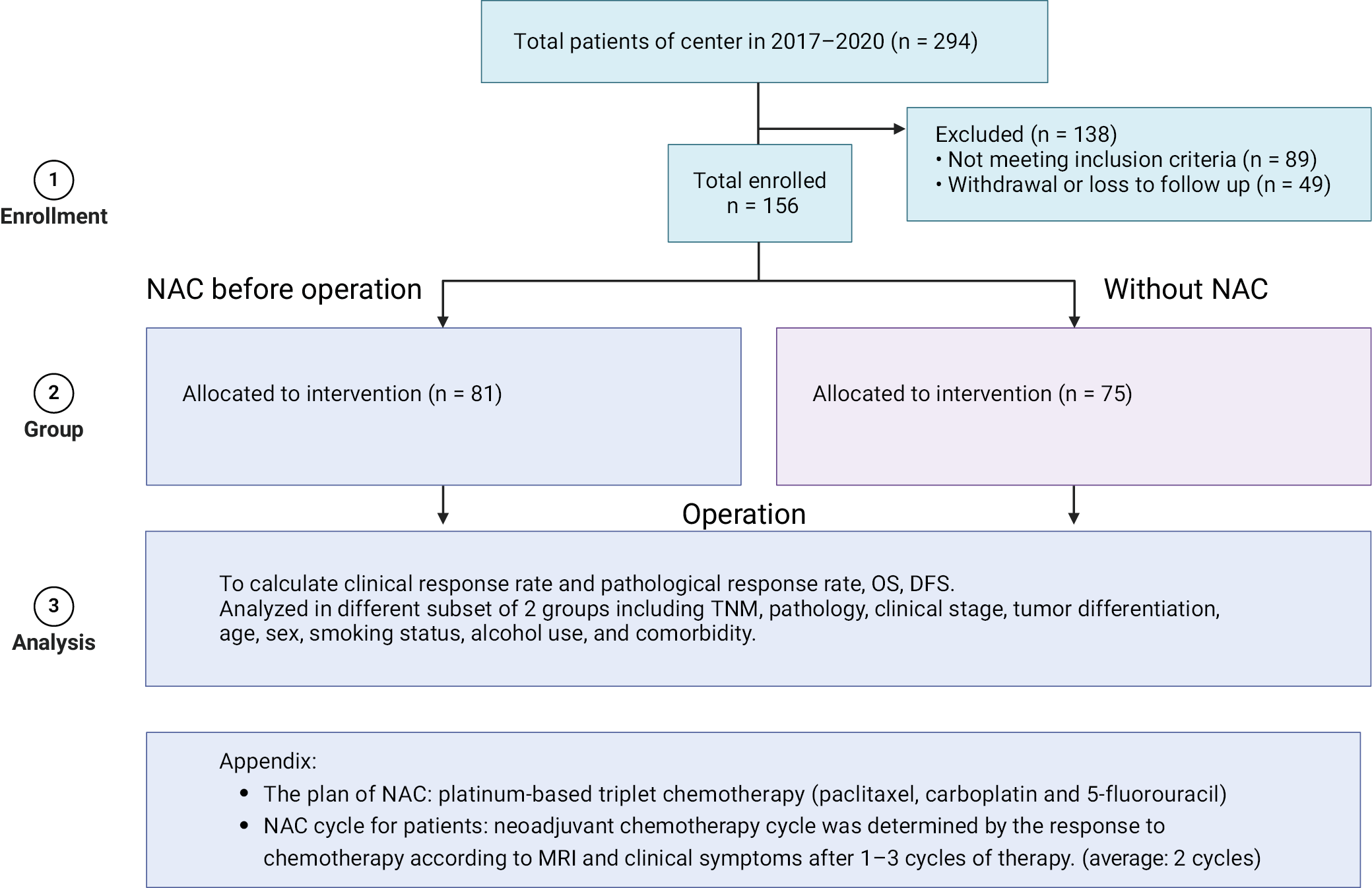
The clinical data for this retrospective study were collected at the First Affiliated Hospital of Zhejiang University School of Medicine (Hangzhou, China) from 2017 to 2020. The study was approved by the institutional review board and was conducted in accordance with the principles of the Declaration of Helsinki (approval No. IIT20220674A). Informed consent was obtained from all individual participants included in the study. The inclusion criteria were as follows: 1) pathological examination confirmed squamous cell carcinoma (SCC); 2) resectable clinical stage III–IV OSCC without metastasis; and 3) received surgery in our hospital. Patients with previous surgery involving a primary tumor or clinical evidence of metastasis were excluded. Patients who received preoperative chemotherapy were enrolled in the NAC group, while patients who received surgery directly were enrolled in the surgery alone group (Figure 1). For the present study, a total of 294 patients with locally advanced resectable SCC from 2017 to 2020 were considered. After excluding 89 patients who did not meet the inclusion criteria and 49 patients who were lost to follow-up, 156 patients were ultimately included in the analysis. These patients were divided into 2 groups: the NAC plus surgery group (n = 81), who received neoadjuvant therapy followed by surgery, and the surgery group (n = 75), who underwent surgery alone.
Patient characteristics and treatment information, including age, sex, smoking status, alcohol use, comorbidity (collected using the ACE-27 index), NAC regimen, type of reconstruction, and postoperative adjuvant therapy, were recorded. Tumor characteristics, including the site of the primary lesion, pathological grade and clinical and pathological tumor-node-metastasis (TNM) stage, were also retrieved for each patient. The clinical and pathological TNM staging were determined according to the 8th edition of the American Joint Committee on Cancer (AJCC) classification.
Clinical management
For the patients in the NAC group, we mainly used platinum-based triplet chemotherapy (paclitaxel, carboplatin and 5-fluorouracil) for preoperative treatment. There is currently no unified standard for the number of cycles of NAC. In this study, the number of NAC cycles was determined by the response to chemotherapy according to magnetic resonance imaging (MRI) and clinical symptoms after 1–3 cycles of therapy. Patients who do not respond to NAC should be discontinued early for surgery, and for those who respond to NAC, the number of chemotherapy cycles should be determined according to the adverse effects and the degree of remission. The duration of neoadjuvant therapy may be appropriately prolonged if the patient’s physical condition permits. Magnetic resonance imaging of patients with mandibular gingival disease in the NAC group revealed the absence of medullary infiltration. The average number of preoperative chemotherapy cycles was 2 (1–3). Before chemotherapy, the palpable edge of the primary tumor was marked by at least 4 points in patients who were assigned to the NAC group to determine the extent of radical surgery. During the 3–4 weeks after NAC completion, they proceeded to radical resection, with 1 cm resection extension to the markers and the proper type of neck dissection. In the surgery alone group, the patients received the same surgery procedure directly, and the excision extension was also defined 1 cm outside the primary lesion. Postoperative therapy was jointly determined by the surgeons, radiation oncologists and medical oncologists, as well as the patient’s choice.
Follow-up and outcomes
The primary outcome of interest was overall survival (OS), and the secondary outcome of interest was disease-free survival (DFS). Overall survival was defined as the time from the start of treatment to the time of death from OSCC. Disease-free survival referred to the time from treatment to the time of tumor relapse or metastasis, as confirmed by the oral and maxillofacial surgeons.
Assessment of chemotherapy response
Clinical response was determined by clinical examination or imaging studies according to the response evaluation criteria in solid tumors v. 1.1 (RECIST 1.1): complete response – all target lesions including the primary lesions and any lymph nodes disappeared; partial response – the sum of the diameters of the target lesions decreased at least 30% compared to the baseline; progressive disease – the sum of the diameters of the target lesions increased at least 20% compared to the baseline; and stable disease – no significant tumor regression or increase.18 Patients with complete or partial response were defined as clinical responders, while patients who had stable or progressive disease were classified as clinical non-responders. All patients underwent radical resection and pathological examination in our study, and patients with no remaining viable tumor cells were enrolled in the pathological complete response (pCR) group. Those with the presence of viable tumor cells were enrolled in the non-pCR group.
Statistical analyses
Pearson’s χ2 test or Fisher’s exact test (if n < 5 in ≤20% of cells) were conducted for categorical data, and Student’s t-test was used for continuous variables (Table 1). The normality of continuous variables was assessed using the Shapiro–Wilk test (p > 0.05) (Supplementary Fig. 1), and the homogeneity of variances was evaluated using Levene’s test (p > 0.05). Survival analysis and survival curves were performed using the Kaplan–Meier (KM) method, with differences analyzed using the log-rank test. Multicollinearity was assessed using the variance inflation factor (VIF). All VIF values were below the widely accepted threshold of 10, indicating a lack of multicollinearity among the predictors (Supplementary Tables 1 and 2).
The proportional hazards assumption was tested using Schoenfeld residuals (Supplementary Fig. 2), and residual plots were generated to examine and confirm the absence of discernible trends between the log-hazard function and predictor variables (Supplementary Fig. 3). A Cox proportional hazard model was performed to estimate the hazard ratio (HR) with 95% confidence intervals (95% CIs) and to identify prognostic factors associated with survival. All hypothesis-generating tests with a p < 0.05 were considered statistically significant. All analyses were performed using the R statistical software v. 4.1.2 (R Foundation for Statistical Computing, Vienna, Austria).
Results
Baseline characteristics
Between 2017 and 2020, 156 eligible patients with untreated locally advanced OSCC received surgical treatment at our center. Eighty-one patients received preoperative chemotherapy followed by radical surgery, and 75 patients received surgery alone. The median follow-up duration was 36 months (range: 3.7–58.9 months). Table 1 presents the baseline characteristics of the patients included in our study for both the surgery alone and NAC groups. Statistical analysis showed no significant differences in sex, age, clinical TNM stage, or pathological grade between the 2 groups despite differences in tumor location and postoperative treatment.
Survival outcomes
Patients with or without preoperative chemotherapy had comparable OS at 3 years (78.3% vs 79.8%, log-rank test: p = 0.76) (Figure 2A). However, the surgery alone group had better 3-year DFS when compared with the NAC group (59.2% vs 42.4%, log-rank test: p = 0.048) (Figure 2B).
Kaplan–Meier survival analyses in the prespecified substrata of pathological T stage and N stage are presented in Figure 3. Patients with pT0–2 OSCC after NAC and surgery exhibited similar survival patterns to patients with pT0–2 OSCC after surgery alone (Figure 3A,B). Inferior 3-year DFS was detected in patients with pT3–4 stage after NAC and surgery when compared to patients with the same stage after surgery alone (40.2% vs 58%, log-rank test: p = 0.033), but OS rates were comparable in the 2 groups (Figure 3C,D). There were no significant differences in OS or DFS rates when patients were stratified by pathologic nodal status (Figure 2E–H).
Response to NAC
According to RECIST 1.1, 50 (61.7%) of 81 patients showed a favorable clinical response, consisting of complete response in 6 patients (7.40%) and partial response in 44 patients (54.3%). Twenty-nine patients (35.8%) had stable disease and 2 patients (2.50%) had progressive disease after preoperative chemotherapy. Pathological complete response was observed in 12 patients (14.8%), while viable tumor cell residue of varying degrees was observed in most patients. Clinical responders had a significantly higher 3-year OS compared to clinical non-responders (82.4% vs 60.7%, log-rank test: p = 0.023; Figure 4A). The 3-year DFS also presented similar results (54.3% vs 18.7%, log-rank test: p = 0.005; Figure 4B). There was a slight survival advantage for patients with pCR compared to patients without pCR, but the difference was not significant (Figure 3C,D).
Cox regression
Table 2, Table 3 show the results of univariate Cox regression models for patients in both the NAC and surgery alone groups. For patients who received NAC and surgery, pathological T stage (pT) and clinical response to NAC were important predictors of OS in univariate analysis. Pathological tumor stage and clinical response, as well as pathological grade, were significant predictors of DFS. However, no clinical factors were found to be significant in the multivariate analysis for either OS or DFS. For patients who underwent surgery alone, pT stage was associated with lower OS, but no predictor of DFS was detected.
Discussion
Neoadjuvant chemotherapy treatment strategies for locally advanced OSCC patients
Preserving tissue function and maintaining the patient’s quality of life are fundamental criteria in the management of treatment for patients with locally advanced OSCC.1 Advancements in flap tissue repair and reconstruction have expanded the indications for surgery.19 The question of whether tumor volume reduction prior to surgery is necessary in OSCC remains a topic of debate.
Neoadjuvant chemotherapy has been discussed for decades; whether OSCC patients benefit from it is controversial. Supporters argue that NAC can reduce tumor volume, make margins clearer, increase the success rate of complete resection, reduce the difficulty of tissue reconstruction, and improve survival rates and quality of life. However, opponents believe that for patients with locally advanced OSCC, the tumor progresses rapidly, and non-response to NAC may delay the timing of treatment and increase the surgical risks. A study on the timing of NAC has shown that when the delayed time to surgery is over 34 days, the 3-year DFS rate is significantly worse.20
To evaluate the influence of NAC on survival in patients with locally advanced OSCC and compare the risk factors among patients with different treatment regimens, we retrospectively analyzed 2 groups of stage III or stage IVA OSCC patients with comparable baseline characteristics. Our data showed that NAC had no positive impact on the survival outcomes of OSCC patients, which suggests that upfront surgery could be more appropriate for patients with locally advanced OSCC in current practice. Theoretically, NAC can cause a reduction in tumor volume and improve long-term clinical outcomes by pretreating possible micrometastasis.21 Despite a few studies demonstrating the benefits of NAC on survival,22, 23 many clinical trials have proved that neoadjuvant administration does not prolong the survival of patients with locally advanced OSCC.13 A clinical trial in 2013 involving 256 patients with locally advanced OSCC demonstrated that patients who received 2 cycles of paclitaxel, cisplatin and 5-fluorouracil (TPF) regimen as NAC before surgery did not achieve better survival outcomes compared to those who underwent radical surgery directly.13 Another randomized clinical trial involving 198 patients with OSCC showed that NAC with cisplatin and fluorouracil (PF) regimen before surgery did not significantly improve survival outcomes after 11.5 years of follow-up compared to direct surgery.24 A meta-analysis on locally advanced HNSCC concluded that the addition of adjuvant chemotherapy or NAC on top of platinum-based chemoradiotherapy did not provide survival benefits for patients.25 A recent meta-analysis including 1,373 patients indicated that there was no statistically significant difference between treatment groups in terms of OS, DSF, locoregional recurrence, and distant metastasis.26 In this study, we also found no improvement in OS for patients with OSCC that were treated with NAC and radical surgery. However, worse DFS was observed in the NAC group, which indicated that more local recurrence and distant metastasis occurred in patients who received chemotherapy before radical surgery. Another retrospective study showed similar results.15
A possible reason we assumed for the inferior DFS in the NAC group was that worse locoregional control resulted from the challenge of the surgical margin after chemotherapy. The tumor can be less palpable after chemotherapy, which makes it more difficult to determine the extent of surgical resection. Furthermore, tumor regression was proven to be irregular and non-centripetal, leading to residual tumor cells being located away from the main tumor bed, which may result in false negative margins in frozen section examination and routine pathological examination.27, 28 On the other hand, the factors contributing to our research findings may also encompass a diminished chemotherapy response rate. The chemotherapy regimen, margin status, extranodal extension (ENE) status, and tumor differentiation level may be important factors influencing NAC. Within our study, the favorable prognostic implications observed in patients attaining a pCR did not result in an OS advantage for the entire cohort undergoing induction chemotherapy. This discrepancy could be ascribed to the relatively restricted number of patients achieving pCR, which may potentially dilute the cumulative survival benefit. The improved survival outcomes observed in pCR patients may be attributed to the beneficial effects of chemotherapy or could indicate an inherent positive prognosis in responsive individuals. Subsequent investigations should prioritize the identification of predictive biomarkers for response to induction chemotherapy to facilitate the optimized use of preoperative medications in chemosensitive patients while mitigating toxicities in nonresponsive cases.
Possible risk factors for failure of NAC
To identify patients with a higher risk of treatment failure and guide the choice of appropriate treatment modality, we conducted a KM analysis in subgroups stratified by pathological features. The results showed that the survival pattern was similar in patients with early-stage tumors (pT0/2) in the NAC group. However, patients with pT3/4 stage after upfront surgery had better DFS than patients who remained in stage pT3/4 after NAC. No significant difference in survival was found when we stratified the patients according to lymph node status. Local–regional recurrence has been proven to be the main failure pattern for OSCC patients both with and without preoperative treatment,13, 29 suggesting that patients with pT3/T4 stage after chemotherapy may have a higher risk of locoregional relapse than patients with the same stage after surgery alone. More to the point, the optimal postoperative administration for patients receiving NAC remains unclear.30 Referring to postoperative therapy for patients without NAC, pT3/4 is only a relative indication for radiotherapy recommended by National Comprehensive Cancer Network (NCCN) guidelines. Given our results that patients who remained in pT3/4 stage after NAC faced a higher risk of locoregional failure, we believe that aggressive postoperative treatment is necessary to realize better local control for these patients, including radiotherapy or chemoradiotherapy. The benefits of enhanced locoregional control in the survival of patients pretreated with NAC further substantiate this view. In a clinical trial by Zhong et al.,13 equivalent survival outcomes were achieved between the TPF induction arm and the upfront surgery arm, as all patients in both groups underwent postoperative radiotherapy. Nevertheless, more research is required to confirm this speculation, since our present study did not involve the decision-making of adjuvant administration directly.
Our analysis tried to identify the risk factors for survival in patients with different treatment regimens using Cox proportional hazards regression models. Pathologic tumor stage and clinical response to NAC were important factors in predicting survival outcomes after pretreated chemotherapy and surgery, but neither was an independent predictor, as the 2 factors interact strongly. For patients treated with upfront surgery, pathologic tumor stage exhibited a consistent effect on predicting OS, as a previous study reported.31 The histologically confirmed tumor stage after NAC is usually considered invalid for prognosis.32 However, our findings suggest that pathologic stage plays a similar prognostic role for patients both with and without NAC in whom advanced pathological stage was a predictor of worse survival. The degree of pathological differentiation is an independent prognostic factor for DFS, with patients of lower pathological differentiation being at a higher risk of regional recurrence and distant metastasis compared to those of moderate-to-high differentiation. Due to the higher microvessel density in histologically poorly differentiated SCC, it is believed that poorly differentiated SCC may be more sensitive to chemotherapy.33 A retrospective analysis by Kina et al.34 suggested that compared to well-differentiated OSCC, moderately-to-poorly differentiated OSCC exhibits higher chemotherapy sensitivity and benefits from sequential neoadjuvant therapy (bleomycin regimen). However, our data did not reveal chemotherapy sensitivity in poorly differentiated patients. Similar to studies in the non-NAC population,35, 36 our results indicated that patients with histologically poorly differentiated oral cancer in the NAC + radical surgery group had worse DFS compared to those with moderate-to-high differentiation, possibly due to the higher invasiveness of poorly differentiated OSCC.
In this study, patients in the NAC group had a clinical response rate of 61.7% and a pCR rate of 14.8%, which are comparable to previous reports.20, 37 It is noteworthy that in patients with a favorable clinical response, survival was significantly improved compared to those with an unfavorable clinical response. Pathologic response was considered to be a more important predictor of survival than clinical response, which was determined imprecisely by image examination.38 However, in this study, we failed to demonstrate a significant improvement in survival for patients with pCR due to a relatively small sample number. The identification of chemotherapy response as a prognostic factor for prolonged survival underscores the importance of identifying the potential chemosensitive subgroup of OSCC before determining the treatment regimen. Neoadjuvant chemotherapy before tumor excision should be avoided in patients who may have a poor response to chemotherapy to prevent chemotherapy complications and unnecessary surgical delays.20 Recently, a few attempts have been made to solve this problem. For example, the expression levels of GDF1539 and Annexin A140 have been reported to be potential predictive biomarkers for benefitting from TPF NAC for OS in patients with OSCC who were treated with NAC and radical surgery. Tumor blood flow assessed using perfusion computed tomography,22 body mass index (BMI)41 and preoperative mean platelet volume42 have also been reported to be helpful in predicting NAC outcomes. Unfortunately, no universally available biomarker is currently in clinical use. For future studies, further investigations are necessary to identify the subgroups of patients who are sensitive to preoperative chemotherapy and thereby contribute to the advancement of personalized medicine practice.
Simultaneously, our results underscore the importance of postoperative adjuvant therapy. There are no established postoperative treatment guidelines for patients who have previously undergone NAC. Some patients who have received NAC may overlook postoperative adjuvant therapy due to issues such as tumor stage downgrading after surgery, poor economic conditions and decreased quality of life. However, several studies43, 44 have demonstrated the significance of postoperative radiotherapy/chemotherapy in improving DFS, and emphasis should be placed on postoperative adjuvant therapy for patients who have undergone NAC. Of course, this conclusion requires further confirmation through large-scale clinical studies.
The challenges and future of NAC for OSCC patients
Although studies on different chemotherapy regimens have shown some variations in their impact on DFS and OS rates in different research reports, there is still no traditional chemotherapy drug that has consistently demonstrated excellent efficacy. In current clinical practice, the TPF regimen is considered the standard induction chemotherapy regimen for advanced oral cancer patients due to its higher response rates.45 However, fluorouracil has been associated with stronger toxic reactions, such as mucositis, gastrointestinal toxicity, hematologic toxicity, and cardiac toxicity,46 prompting researchers to explore alternative neoadjuvant regimens in hopes of achieving higher response rates and lower toxicity. A 2014 retrospective study by Herman et al. 47 included 143 patients who received induction TPF or cisplatin plus paclitaxel (CT) prior to definitive chemoradiotherapy. The study results indicated that the CT regimen showed similar or even better progression-free survival and local control rates compared to the TPF regimen, with lower renal toxicity. Response rates for the CT regimen reported in other studies ranged from 50% to 85%.46, 48, 49, 50 A large-scale clinical study on borderline resectable oral cancer has demonstrated that using more than 2 drugs in combination increases the survival rate of patients compared to using only 2 drugs.6
Several meta-analyses have shown consistent results, indicating that NAC does not confer a survival or response rate benefit.26 However, the efficacy of NAC in the context of surgical margin clearance remains inadequately investigated. At this juncture, surgery should be upheld as the primary treatment modality for locally advanced OSCC. Furthermore, for patients requiring neoadjuvant therapy, it should adhere closely to the principles of comprehensive cancer care, including postoperative adjuvant radiotherapy or chemotherapy. Although various studies have reported differences in DFS and OS rates with different chemotherapy regimens, no traditional chemotherapy agent has consistently demonstrated superior efficacy. In recent years, novel neoadjuvant regimens incorporating immune checkpoint inhibitors, such as anti-PD-1/PD-L1, anti-KIR, anti-Tim3, and anti-CTLA-4, collectively known as neoadjuvant immunotherapy, have shown promising results in terms of survival outcomes.37, 51 When conditions permit, immunotherapy may be combined to enhance the rate of response to neoadjuvant therapy.
Further research is warranted to validate the efficacy of neoadjuvant immunotherapy. For example, the expression of PDL1 in tumors and tumor-infiltrating immune cells can be utilized to predict the response rate to PD1/PDL1 immune checkpoint inhibitor therapy. However, the unknown expression patterns of immune checkpoints in tumor cells during NAC treatment pose challenges in predicting treatment efficacy based on protein expression and gene mutations. In the context of OSCC, there remains a significant lack of broadly effective immune checkpoint inhibitors for the majority of patients with OSCC. Therefore, neoadjuvant immunotherapy may entail dual risks, including the potential side effects of immune checkpoint inhibitors and a high financial burden.
It is challenging to definitively classify NAC as entirely inconsequential solely based on the outcomes of a single clinical study. With stringent control over indications, our objective is to augment the response rate to chemotherapy and lengthen patients’ DFS through a combination of diverse medications and immunotherapy. We maintain the belief that NAC remains advantageous for responsive patients. Furthermore, NAC plays a critical role in the management of patients with locally advanced unresectable oral cancer. We believe that, alongside exploring evolving chemotherapy combinations, investigating the patient’s clinical characteristics and the genetic molecular markers that influence treatment response represents a promising avenue for future research in the field of NAC.
Limitations
As a retrospective study, this research faced challenges in controlling for patients’ baseline data and was limited to a cohort of 156 patients with advanced OSCC from a single center. Furthermore, there were some missing clinical baseline data, complicating efforts to mitigate the bias introduced by clinical decision-making in the NAC group through methods such as matched grouping. Moreover, patients requiring NAC, even within the same advanced stage, may present with more severe conditions and higher disease grades. The absence of randomization introduced multiple factors that contribute to the uncertainty of the study results. While local recurrence and distant metastasis represent 2 patterns of treatment failure in OSCC patients, this study lacked the statistical power to differentiate between the two.
Conclusions
Patients with advanced pathological stages after NAC may be at a higher risk of treatment failure, and upfront surgery is recommended for locally advanced OSCC patients in current clinical practice.
Supplementary data
The Supplementary materials are available at https://doi.org/10.5281/zenodo.13373456. The package includes the following files:
Supplementary Fig. 1. Normality test of age.
Supplementary Fig. 2. Schoenfeld residuals plots for each covariate.
Supplementary Fig. 3. Residual plot of age.
Supplementary Table 1. Assessment of multicollinearity using the VIF.
Supplementary Table 2. Assessment of multicollinearity using the VIF.
Data availability
The datasets generated and/or analyzed during the current study are available from the corresponding author on reasonable request.
Consent for publication
Not applicable.

















.jpg)
.jpg)
.jpg)