Abstract
Background. The introduction of intravitreal injections of melphalan (IVIM) has significantly improved the efficacy of retinoblastoma treatment and the prognosis for eye preservation.
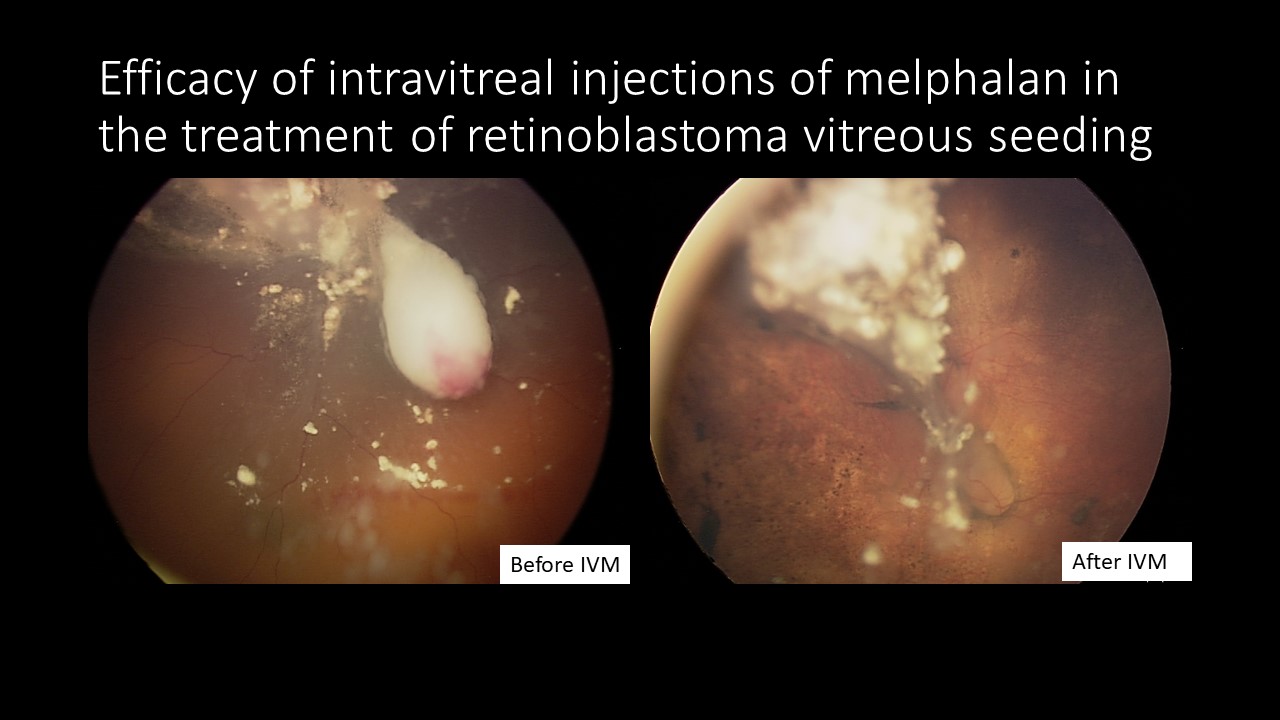
Objectives. To evaluate the results of using IVIM to treat retinoblastoma vitreous seeding.
Materials and methods. This was a clinical, retrospective, single-center study. Twenty-six children (27 eyes) who met all of the following inclusion criteria qualified for the study: 1) active vitreous seeding at the time of retinoblastoma diagnosis; 2) IVIM performed between 1 January 2017 and 30 September 2020; and 3) a minimum follow-up period of 12 months since the last IVIM. Doses of 20–40 μg melphalan per injection were used.
Results. The eye observation period from the last IVIM to the last ophthalmic examination averaged 32.41 months (median 30.00; range 13.00–56.00). Success (no active tumors in the vitreous body) was achieved in 24 eyes (88.9%), and a doubtful result (recurrence in the retina with a difficult-to-determine etiology) in 2 eyes (7.4%). In 1 eye (3.7%), despite treatment, active tumors were still present in the vitreous body. Out of all 27 eyes, 4 eyeballs were removed, but the direct cause of enucleation was not vitreous seeding. There were no complications in the form of intraocular inflammation, extraocular retinoblastoma or distant metastases. There was 1 case of anterior uveitis and 1 case of cataract.
Conclusions. The IVIM is a highly effective and safe form of treatment for retinoblastoma vitreous seeding.
Key words: therapy, retinoblastoma, injection, melphalan, intravitreal
Background
The introduction of intravitreal injections of melphalan (IVIM or, more generally, intravitreal chemotherapy (IVitC)) has significantly improved the efficacy of retinoblastoma treatment and the prognosis for eye preservation.1, 2
For decades, in many cases of retinoblastoma vitreous seeding (Figure 1, Figure 2), even in patients with good visual acuity, the treatment of choice was enucleation or external beam radiotherapy, which had serious side effects. When active retinoblastoma seeding is present in the vitreous body, IVIM allows therapeutic concentrations of the chemotherapeutic agent to be achieved in the vitreous chamber.3 No other treatment is as effective in eradicating retinoblastoma seeds from the vitreous body as IVitC. The use of systemic chemotherapy, intra-arterial chemotherapy (IAC) or periocular chemotherapy does not make it possible to achieve adequate concentrations of chemotherapeutics in this compartment because of the blood–retina barrier and the lack of blood vessels in the normal vitreous body.1
For decades, when treating retinoblastoma, there was a concern that intravitreal injections might carry the risk of extraocular spreading of the tumor due to needle penetration of the sclera. It was not until the pioneering work of researchers in Japan that melphalan was identified as a highly effective chemotherapeutic agent against retinoblastoma cells. Effective and safe concentrations of this drug for ocular tissues were determined, and it was introduced into intra-arterial and intravitreal therapy.1, 4, 5, 6 Further research on the safety of IVIM and dissemination of the treatment is largely due the research by Munier.2, 3
Objectives
The aim of this study was to determine the efficacy and safety of IVIM in the treatment of retinoblastoma vitreous seeding in patients treated at our center.
Materials and methods
This study was approved by the Ethics Committee of the Children’s Memorial Health Institute (Warsaw, Poland; approval No. 1/KBE/2022). The study was a retrospective analysis of clinical data.
Setting and participants
The study group included all patients from the Department of Ophthalmology at our center who fulfilled all 3 of the following inclusion criteria: 1) active vitreous seeding at the time of retinoblastoma diagnosis; 2) treatment with IVIM between January 1, 2017 and September 30, 2020; and 3) a follow-up period of at least 12 months since the last IVIM.
Indications and dosages of IVIM therapy
In each patient, the indication for the 1st course of IVIM was the presence of retinoblastoma in the vitreous body. The IVIM was started before the completion of primary retinoblastoma treatment (systemic or IAC). If the 1st course of IVIM failed, the eye was eligible for a 2nd course of IVIM and simultaneously for other forms of therapy, if needed (IAC or local treatment). None of the eyes qualified for a 3rd course of IVIM. A prerequisite for completion of a given course of IVIM was the absence of an active tumor in the vitreous body at the 1st follow-up examination, which was at the latest 6 weeks after the last IVIM.
The number of IVIMs and the injection dose varied between patients and differed between the 1st and eventual 2nd courses of IVIM. It depended on the severity of retinoblastoma seeding in the vitreous body at the time of qualification for IVIM and the response to IVIM therapy. Dust-like seeds localized in 1 quadrant of the vitreous body were an indication to schedule fewer injections (1 or 2 IVIMs) and use a lower single dose (20 μg or 25 μg). Seeding consisting of spheres or larger tumor fragments or those occupying 2 or more quadrants were indications to schedule more injections (3 or more) and use a higher single dose (30 μg). Some patients received different doses in successive injections (IVIM was started with higher doses that were then reduced to minimize toxicity). Individual injections were planned at intervals of 1–2 weeks.
Criteria for assessing the success
of IVIM therapy
The efficacy of IVIM was assessed in follow-up examinations under general anesthesia. Patients underwent follow-up sessions every 1–2 months in the 1st year after treatment, then every 2–6 months.
We considered the IVIM therapy to be successful if there were no active tumors in the vitreous body and no active tumors on the retina originating from the vitreous body. We considered the IVIM therapy to be a failure in the case of the presence of active retinoblastoma in the vitreous body (recurrence or tumor not responding to treatment) or the appearance of new active tumors on the retina originating from the vitreous. We did not consider the therapy to be a failure in the case of the presence of an active retinal tumor originating from the retina or in the case of enucleation if it was performed for reasons other than an active tumor in the vitreous body or an active tumor on the retina originating from the vitreous body. We considered the presence of an active retinal tumor of difficult-to-determine etiology to be a doubtful result (i.e., it was not possible to exclude its origin from seeding in the vitreous body).
IVIM surgical technique
At the time of qualification for IVIM, a seed-free quadrant was chosen as the injection site. The most frequently chosen location was the 11 o’clock meridian. The skin of the eyelids was cleaned with a solution of 10% povidone-iodine (PI) and then the conjunctival sac was cleaned with with 5% PI eye drops for 1 min. The needle insertion site was planned 2.0–3.5 mm from the corneal limbus (depending on the child’s age). An injection of 20–40 μg of melphalan in a volume of 0.02–0.04 mL was performed with a 30G × ½” needle (0.3 × 13 mm). The injection was followed by applying cryotherapy at the injection site during needle withdrawal or a subconjunctival injection of 0.005 μg of melphalan in a volume of 0.005 mL. Finally, the conjunctival sac was rinsed with AquaPro injection solution.
During the study period, we used the following melphalan preparations, subject to availability: Melphalan Tillomed 50 mg, Aspen Melphalan 50 mg, or Mederan Melphalan 50 mg.
Systemic or intra-arterial chemotherapy
All patients who qualified for the study were administered systemic chemotherapy (VEC – vincristine, etoposide and carboplatin), intra-arterial chemotherapy or a combination of both, as primary treatment for retinoblastoma. Systemic chemotherapy, as standard, consisted of 6 cycles of VEC administered at 3-week intervals. The IAC, as standard, consisted of 3 courses of melphalan, topotecan and carboplatin at intervals of 3–4 weeks. The VEC+IAC combination consisted of 2 courses of VEC followed by 3 courses of IAC.
Participants
Twenty-seven eyes of 26 children (15 boys and 11 girls) were eligible for the study (1 child had IVIM administered to both eyes). The mean age at retinoblastoma diagnosis was 28.04 months (median 26.00; range 2.00–83.00). Bilateral retinoblastoma was diagnosed in 9 patients, and unilateral retinoblastoma was diagnosed in 17 children. All patients were Caucasian (relatively low amounts of pigment in the eye tissues).
At the time of diagnosis, the eyes were classified according to the International Classification for Retinoblastoma, as modified by Shields, into the following groups: A – 0 eyes, B – 0 eyes, C – 7 eyes, D – 15 eyes, and E – 5 eyes.7 Retinoblastoma seeding in the vitreous body found at the time of diagnosis was retrospectively classified according to Munier’s classification into the following groups: dust – 12 eyes, spheres – 8 eyes, clouds – 3 eyes, and no data – 4 eyes.3 The eye observation period from the last IVIM to the last ophthalmic examination averaged 32.41 months (median 30.00; range 13.00–56.00).
First course of IVIM
All patients were administered a 1st course of IVIM to treat retinoblastoma seeding in the vitreous body. The mean number of injections during the 1st course of IVIM was 3.41 (median: 3.00, range: 1.00–8.00). The single doses of melphalan used in the 1st course of IVIM were 20 μg – 42 injections (46.2%), 25 μg – 19 injections (20.9%), 30 μg – 29 injections (31.9%), and 40 μg – 1 injection (1.1%). The mean cumulative dose of melphalan per eye in the 1st course of IVIM was 83.33 μg (median: 80.00, range: 30.00–200.00).
Second course of IVIM
If the 1st course of IVIM failed, a 2nd course of IVIM was administered. The mean number of injections during the 2nd course of IVIM was 2.83 (median: 3.00, range: 2.00–4.00). The single doses of melphalan used in the 2nd course of IVIM were 20 μg – 4 injections (23.5%), 25 μg – 5 injections (29.4%) and 30 μg – 8 injections (47.1%). The mean cumulative dose of melphalan per eye in the 2nd course of IVIM was 74.17 μg (median: 67.50, range: 55.0–120.00).
Systemic or intra-arterial chemotherapy
During the 1st IVIM cycle, the following were used as primary treatments for retinoblastoma: systemic chemotherapy only – 3 eyes, IAC only – 9 eyes and VEC+IAC combination – 15 eyes. During the 2nd IVIM cycle, the following were administered: IAC only – 4 eyes, and neither IAC nor systemic chemotherapy – 2 eyes.
Results
The results of IVIM treatment (after the 1st and 2nd courses of IVIM combined) are shown in Table 1. The eye classified in the treatment failure group had active seeding in the vitreous body at the time of enucleation, despite 6 IVIMs, and a subretinal recurrence. Seeding in the vitreous body was not the direct cause of any enucleations.
Results of the 1st course of IVIM
The 1st course of IVIM was successful in 20 eyes (74.1% of 27 eyes). No form of retinoblastoma recurrence was found in 16 eyes (59.3% of 27 eyes). In 4 eyes (14.8% of 27 eyes), a recurrence was found on or under the retina, probably not originating from the vitreous body (one of those eyes was later removed due to a recurrence anterior to the ora serrata).
Failure of the 1st course of IVIM was observed in 7 eyes (25.9% of 27 eyes). No adequate response was found in 1 eye (3.7% of 27 eyes); the tumor remained active in the vitreous body, and there was a recurrence under the retina, which was the immediate cause of eye removal. Recurrence in the vitreous body was found in 4 eyes (14.8% of 27 eyes). A retinal recurrence, probably originating from the vitreous body, was found in 2 eyes (7.4% of 27 eyes).
The time to recurrence in the vitreous body from the last IVIM injection (4 eyes after the 1st course of IVIM) averaged 11.50 months (median: 5.00, range: 2.00–34.00).
The time to retinal recurrence from the last IVIM (2 eyes after the 1st course of IVIM), probably originating from persistent retinal cells in the vitreous body, was, on average, 5 months (4 months and 6 months).
Results of the 2nd course of IVIM
Six eyes were eligible for a 2nd course of IVIM. After it, no recurrence was observed in 4 eyes. A difficult-to-interpret result was reported in 2 eyes. One of those eyes was removed 1 month after completion of the 2nd course of IVIM due to a lack of retinal tumor response to IAC. The short follow-up period for this eye makes it impossible to determine the efficacy of the 2nd course of IVIM. The 2nd of those eyes, 1 month after completion of the 2nd course of IVIM, had a recurrence on the retina with a difficult-to-determine etiology, and was removed after a further 7 months due to a recurrence near the main tumor mass (this recurrence did not originate from the vitreous body).
Complications after IVIM
One patient developed anterior uveitis the day after IVIM, which was probably due to the administration of melphalan under the conjunctiva as a form of prevention of local spreading of retinoblastoma through the injection site. In 2 patients, sharply demarcated, semicircular in shape, retinal and choroidal atrophy was found on the fundus around the IVIM insertion site on the periphery of the retina near the ora serrata. One patient was diagnosed with a cataract 21 months after the last IVIM injection. No patient was diagnosed with intraocular inflammation, extraocular spreading of retinoblastoma, or distant metastases. None of the patients died.
Discussion
Numerous factors can significantly influence the effectiveness of IVIM treatment of retinoblastoma seeding in the vitreous body: IVIM procedure technique, number of IVIM injections and individual injection doses, diameter of the seeds in the vitreous body, concomitant treatment (IAC and systemic chemotherapy), diameter of the eyeball, presence of posterior vitreous detachment, time interval between IVIM injections, sensitivity of cells with a given genetic mutation to melphalan, and type of melphalan preparation used.1, 3, 8, 9, 10, 11
Based on the results of the therapy of our previously treated patients and the group of patients discussed here, it seems that a single IVIM dose of 20 μg is too low. The initial response of the seeding in the vitreous body is very good, and the tumors become invisible, but within a few months, recurrences in the vitreous body appear. As recommended by Munier, the 20 μg dose should be reserved for the smallest eyeballs because it allows the therapeutic concentration of melphalan to be achieved in only a small volume of the vitreous chamber.12 Our study was conducted at a time when we tried to decrease the single dose of 30 μg melphalan to lower doses of 25 μg or 20 μg to reduce retinal toxicity, but we did see an increase in the number of recurrences of retinoblastoma in the vitreous body, which we had not observed before in patients with the 30 μg dose. Liao et al. published a comparison of 2 groups of patients receiving doses of 25 μg or 30 μg, finding no statistically significant difference in retinal toxicity between these groups based on the results of electroretinography (ERG) tests.13 Currently, the dose we use most frequently is 30 μg. In the case of massive seeding in the vitreous body and a high risk of perioperative extraocular spreading (seeding occupying a very large part of the vitreous body), we start with an initial dose of up to 40 μg and reduce subsequent doses to 30 μg.
Another issue is the number of IVIM injections. From 2012, in the early days of IVIM at our center, we administered only 1 dose of melphalan – 30 μg. After a very good initial response to treatment, we observed recurrences in the vitreous body at a later time. Currently, based on the results of our study and the literature, we use a dose of 30 μg for retinoblastoma seeding in the vitreous body, repeated a minimum of 3 times if “dust” (according to Munier’s classification) is present, 4–5 times if “spheres” are present, and up to 8 times if large retinoblastoma fragments detached from the main tumor mass are found.3 The interval between injections is 1–2 weeks.
Late recurrences originating from persistent retinal cells in the vitreous body are a major problem.1, 14, 15 In our study, the longest follow-up period after which recurrence was observed was almost 3 years (34 months). After such a long period, ophthalmological screenings become less frequent, and tumors can grow significantly in the interval between examinations, necessitating not only the administration of IVIM but often renewed IAC as well. It is, therefore, extremely important to dose the drugs appropriately (number of injections and single dosages) to avoid late recurrence of retinoblastoma in the vitreous body or on the retinal surface. Berry et al. described a relationship between the occurrence of more severe IVIM toxicity of the retina and a lower rate of recurrence in the vitreous body, as well as a lower rate of secondary enucleation.14
Despite the use of relatively low doses of melphalan in IVIM during the analyzed period, active seeding in the vitreous body alone was not the direct cause of any enucleation in our patient group. In the event of recurrence of a retinoblastoma in the vitreous body, we performed a 2nd course of IVIM. The only eye classified in the treatment failure group was enucleated because of a subretinal recurrence and not because of seeding in the vitreous body.
Causes of retinoblastoma seeding in the vitreous body may include perforation of the internal limiting membrane (ILM) in the form of a characteristic hypovascularized “cap” on the retinal surface (Figure 1), large size of the endophytic tumor, and the use of certain therapeutic methods, such as laser photocoagulation and transpupillary thermotherapy.3, 12
Prophylactic administration of IVIM should be considered if the above risk factors for vitreous seeding are present. Based on experience, it appears that a dose of 20 μg is insufficient, even as a preventative measure. In the case of the above risk factors, we currently administer 2 injections of 30 μg IVIM.
Munier’s “dust, spheres and clouds” classification could possibly be enhanced with a “high risk of seeding” point referring to a condition in which ILM puncture occurs or a large endophytic tumor is found without obvious seeding in the vitreous body.
Any active retinoblastoma seeds in the vitreous cavity at the time of diagnosis, therapy or follow-up should be an indication to plan more IVIM. At our center, the decision to plan IVIM is most often made at the time of diagnosis because later, after IAC or systemic chemotherapy, retinoblastoma seeds usually disappear from the vitreous body; however, if not treated with IVIM, they will most often reoccur.
A space with an increased concentration of melphalan is created around the needle in the vitreous body when melphalan is administered.3, 8 For this reason, retinal atrophy is observed at the puncture site, but at the same time, the risk of retinoblastoma spreading beyond the eyeball through this site is low. We have noted that the area of pigment regrouping at the puncture site has a semicircular shape and a very clear boundary. It appears that this may be due to reflux of melphalan along the needle remaining in the vitreous body (in children with thick gel consistency) and partial displacement of the injected drug through the hole created by the needle in the base of the vitreous body into the space between the vitreous body and the retina. If this actually occurs, as described above, the risk of extraocular spreading of the retinoblastoma through the injection orifice should be relatively low. It is likely that the focus of choroidal atrophy around the insertion site is a result of the higher concentration of the chemotherapeutic agent at this site.
We perform cryotherapy at the injection site during the 1st course of IVIM when viable tumor cells in the vitreous are most abundant, as well as during subsequent rounds of treatment if we believe there is still an increased risk of extraocular spreading. There are no studies that support the effectiveness of IVIM injection site cryotherapy in preventing extraocular spreading. Cryotherapy induces tissue necrosis around the needle track, which may impair postoperative wound healing and paradoxically facilitate spreading.
In the past, instead of cryotherapy at the injection site, we used a subconjunctival injection of melphalan. We stopped performing this procedure because of the single complication of anterior uveitis described above. This complication appears to have occurred due to an unintentional overdosing of melphalan administered under the conjunctiva. To perform subconjunctival injections safely, a separate puncture with a 2nd syringe and needle is needed. The subconjunctival dose we used was 0.005 μg in a volume of 0.005 mL.
During IVIM, we administer melphalan into the vitreous chamber in a very small volume of fluid (30 μg of melphalan in 0.03 mL) in order to minimize the possibility of reflux outside the eyeball and an increase in intraocular pressure, with all its consequences.
The IVIM is planned before the completion of systemic or intra-arterial chemotherapy so that IVIM is administered during the period of the lowest tumor activity. The administration of a course of systemic chemotherapy or IAC after the 1st IVIM injection reduces the possibility of extraocular spreading. Starting IVIM treatment before systemic chemotherapy or IAC would pose a higher risk of extraocular spreading due to the large number of viable tumor cells.
There are various techniques for administering IVIM. One technique, promoted by Munier, includes paracentesis of the anterior chamber and aspiration of the same volume of fluid that is then injected into the vitreous chamber. Melphalan is then administered in a relatively large volume of fluid into the anterior vitreous body to the area immediately behind the lens. When the needle is withdrawn, cryotherapy is applied at the injection site, and the eye is shaken to distribute the drug evenly.3 Yu et al. proposed a more precise method of administering melphalan into the area of the retinoblastoma seeding in the vitreous body using an indirect ophthalmoscope.15 The technique used in our center is similar to standard intravitreal injections of anti-VEGF preparations, taking into account the specificity of retinoblastomas. There are no studies proving superior efficacy or safety of any of the IVIM techniques. Our IVIM technique is easier, cheaper and faster, and significantly reduces the number of manipulations performed on the eye and the risk of mechanical or toxic damage to the lens. Compared to the techniques described by Suzuki et al., Munier, Francis et al., and Yu et al., we use the smallest volume of injected fluids.1, 3, 8, 15, 16 In the course of any IVIM technique, care should be taken when performing ultrasound before the procedure to ensure that the needle does not enter the retrohyaloid space created after the rapid regression of a large tumor because then the administration of a standard dose of melphalan may cause retinal atrophy in the entire area under the detached vitreous body. A similar complication can arise from unintentional insertion of the needle tip into Cloquet’s canal.
At our center, we do not perform eyeball shaking with tweezers immediately after IVIM to distribute melphalan throughout the vitreous body (some authors recommend such shaking).1, 3 It is not recommended after injections of other agents, such as anti-VEGF for example. Our observations of the fluorescence of intravitreal topotecan indicate a relatively rapid (within minutes) diffusion of this drug into the vitreous chamber. There are no published studies indicating the superior efficacy of the treatment of retinoblastoma seeding with IVIM combined with ocular shaking compared to IVIM without shaking.
We do not use IVIM to treat solid tumors of the retina, as proposed by other authors.9, 14 This is because we have observed a lack of noticeable changes in solid retinal tumors after treatment with IVIM during ophthalmoscopic examination.
Limitations of the study
There are 2 main limitations to this study. First, the nature of qualifying eyes into appropriate groups when assessing treatment outcomes is partially subjective. Second, the follow-up period for the regression of tumors in the vitreous body was relatively short.
Conclusions
The IVIM is a highly effective and safe form of treatment for retinoblastoma vitreous seeding. It enables the preservation of eyes in patients for whom, in the past, the only effective treatment was external radiotherapy or enucleation.