Abstract
Background. This article describes 2 cases of post-coronavirus disease 2019 (COVID-19) transient spontaneous osteonecrosis of the knee (PCT-SONK) observed in patients who had previously recovered from COVID-19 without corticosteroid administration.
Objectives. The possible pathomechanisms by which a recent SARS-CoV-2 infection may contribute as a causative factor for osteonecrosis are reviewed, and the differential diagnosis and treatment are discussed.
Materials and methods. Two patients (males, 45- and 47-year-old) presented with sudden onset knee pain with no trauma history. The pain persisted during rest and at night. On magnetic resonance imaging (MRI), no subchondral bone thickening was observed; bone edema was diffusely distributed in the whole femoral condyle, in contrast to the more focal edema that is typically concentrated mainly around the subchondral region in classic SONK. Both patients were treated nonoperatively with no weight bearing and pharmacological agents, and complete resolution of symptoms was achieved.
Results. A follow-up MRI 10 weeks after presentation revealed a near-complete loss of signal in the medial femoral condyle in both patients.
Conclusions. Orthopedic surgeons should be cautious when sudden knee pain without concurrent trauma or a history of injury occurs shortly after severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) infection, even with mild COVID-19 illness. While some studies report the development of post-COVID-19 osteonecrosis after lower doses of corticosteroids and sooner after their administration than in comparable non-COVID-19 cases, our study is the first to report 2 cases with no corticosteroid administration at all. Therefore, the authors believe it adds to the body of knowledge on the potential connections between COVID-19 and PCT-SONK. The transient nature of symptoms and radiological findings suggest that aggressive surgical treatment of non-injury local bone edema occurring shortly after SARS-CoV-2 infection should be avoided.
Key words: knee pain, SARS-CoV-2, avascular necrosis, SONK, COVID-19 complications
Background
Severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is a positive-sense, single-stranded RNA virus that was first identified in late December 2019 in Wuhan, China.1 The disease caused by SARS-CoV-2 infection is referred to as coronavirus disease 2019 (COVID-19). The World Health Organization (WHO) officially declared COVID-19 a pandemic in March 2020.2 By August 2022, the estimated death toll from COVID-19 had risen to 6,446,714.1 Its typical clinical manifestations include fever, cough and shortness of breath, which can progress to pneumonia. This can further deteriorate into a dysregulated immune state characterized by a hyperinflammatory response and a hypercoagulable state, leading to pulmonary and systemic micro- and macro-immunothrombosis, which ultimately may cause multiple organ failure and death.3
Involvement of nearly every organ system has been observed not only during the acute stages of COVID-19 but also after recovery. Recent reports describe post-COVID-19 clinical problems that can extend to affect the knee joint and its function.4 There are also studies reporting secondary osteonecrosis of the knee after COVID-19 treatment with corticosteroids, but with less than the cumulative dose of corticosteroids reported in the literature for osteonecrosis of the knee.5, 6 Similar studies have been published concerning avascular necrosis of the femoral head occurring with lower doses of corticosteroids and sooner after their administration than in comparable non-COVID-19 cases.7 Karaarslan et al. recently performed a prospective study on a group of patients discharged after COVID-19 infection and concluded that “92.3%, 72.7% and 56.3% of patients reported any musculoskeletal symptom at hospitalization, 2-week and 1-month, respectively”.8
Spontaneous osteonecrosis of the knee (SONK) is the most common form of osteonecrosis of the knee joint, but its etiology remains unclear. However, it is believed that SONK might be caused by local blood flow alteration, which leads to subsequent bone ischemia, ultimately resulting in local fluid storage and bone edema that can be visualized on magnetic resonance imaging (MRI).9 Typically, the subchondral lesion due to SONK is observed unilaterally at the medial femoral condyle (MFC) of the knee (94%). The prevalence of SONK is higher in females, especially those over the age of 55, and typically presents as sudden knee pain without causative trauma or injury.10
The COVID-19 has been clearly associated with a hypercoagulable state, referred to as COVID-19-associated coagulopathy (CAC),11 with evidence strongly suggestive of a secondary microangiopathy phenomenon propagating multiple organ failure.12 Laboratory hallmarks of CAC, such as decreased platelets and elevated D-dimer, have been shown to persist for several months after recovery, even in mild cases of COVID-19.13 Therefore, it is possible that SARS-CoV-2 contributes to the development of local blood flow changes and ultimately to post-COVID-19 transient spontaneous osteonecrosis of the knee (PCT-SONK).
This report aims to describe 2 cases of sudden knee pain with radiological features similar to those of SONK that were observed in patients shortly after recovering from COVID-19, and to discuss the possible pathogenesis of this emerging problem for orthopedic surgeons.
Case 1 (K.D.)
A 45-year-old male patient (software engineer, sedentary work) presented with sudden onset right knee pain with no history of trauma. The pain persisted during rest and at night.
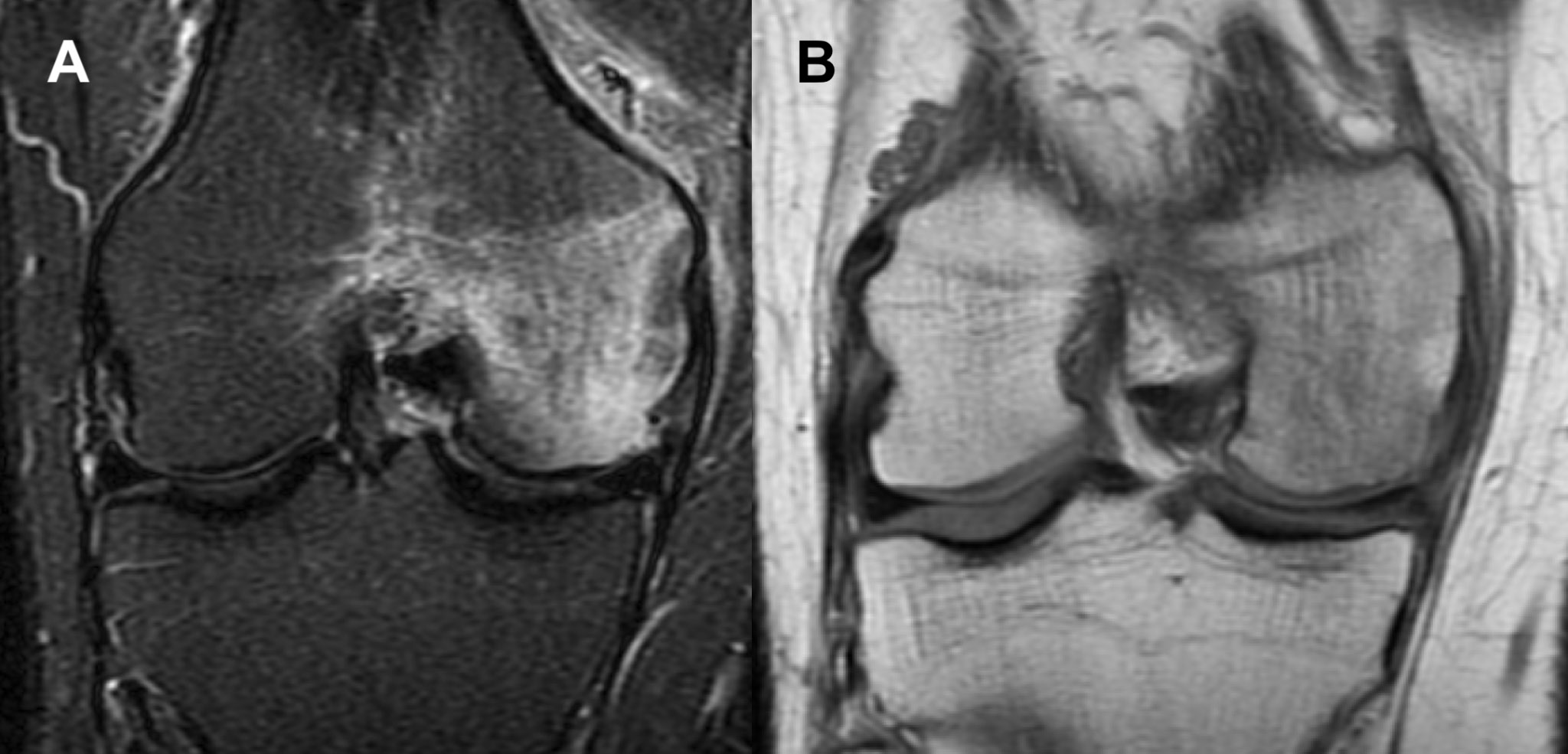
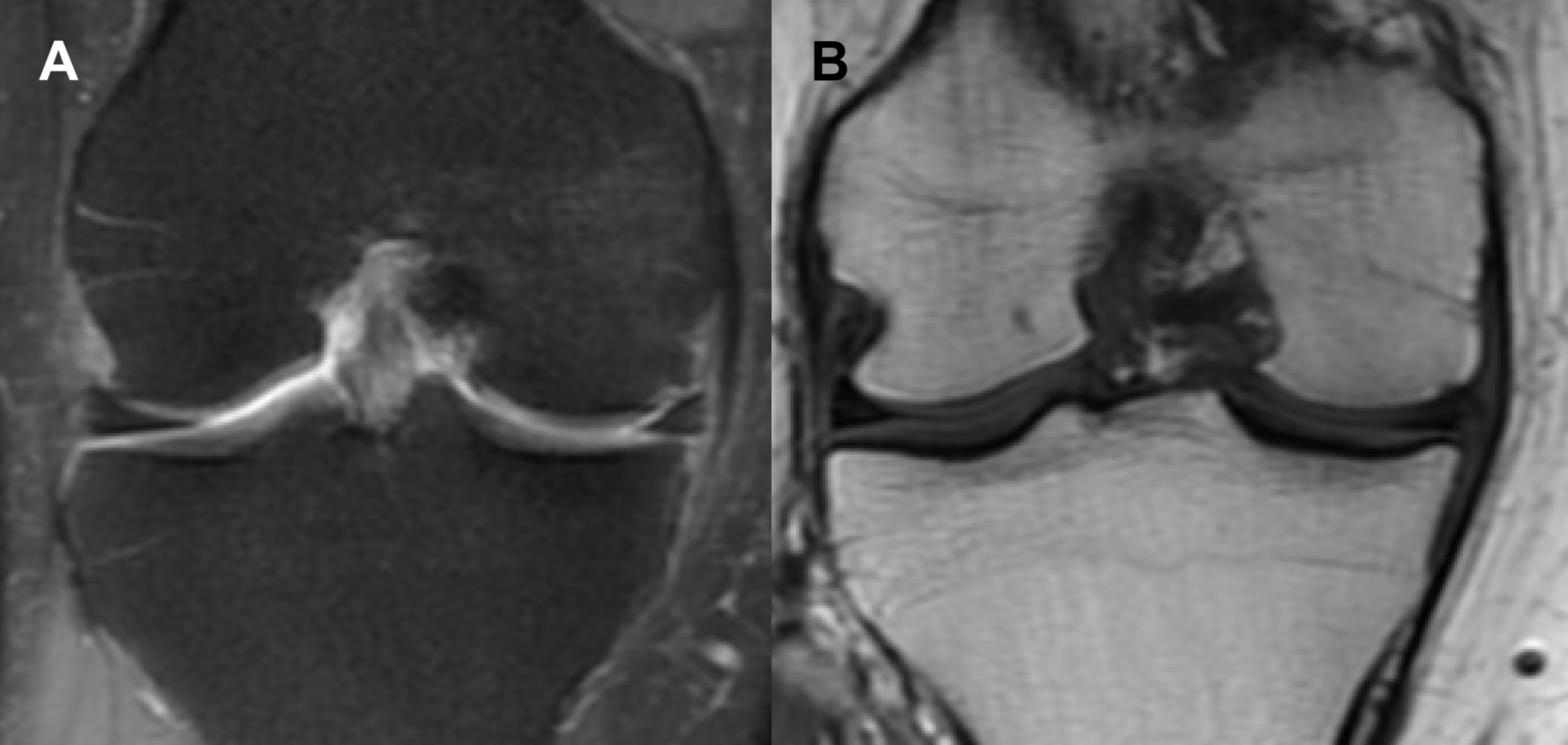
On clinical examination, the patient had a full range of motion and a stable knee joint. The patient experienced pain during extension and near-full extension. Palpation of the MFC and medial joint space revealed local tenderness. Additionally, the patient experienced pain when varus stress was applied to the knee in 10° knee flexion. An MRI of the patient’s right knee was performed immediately following the orthopedic appointment (7 weeks after the onset of knee symptoms). Magnetic resonance imaging in short tau inversion recovery (STIR) sequence revealed a large area of diffuse increased signal in the MFC and the medial aspect of the intercondylar region (Figure 1).
Approximately 3 months prior to the onset of knee symptoms, the patient had been diagnosed with COVID-19, which was confirmed by a positive result of a SARS-CoV-2 RT-PCR test. The patient had begun to experience symptoms of acute respiratory infection 5 days prior to diagnosis, which had progressed to severe flu-like symptoms with dyspnea for approximately next 2 weeks. The patient was not hospitalized despite the severity of the respiratory symptoms, and no corticosteroids were administered.
The patient was put on crutches and instructed to bear no weight on the affected leg. Additionally, the patient was prescribed ossein-hydroxyapatite complex (2490 mg/day), calcium (1000 mg/day), vitamin D₃ (150 µg/day), vitamin K₂ (50 µg/day), pentoxifylline (400 mg/day), vitamin C (2,000 mg/day), and acetylsalicylic acid (75 mg/day).
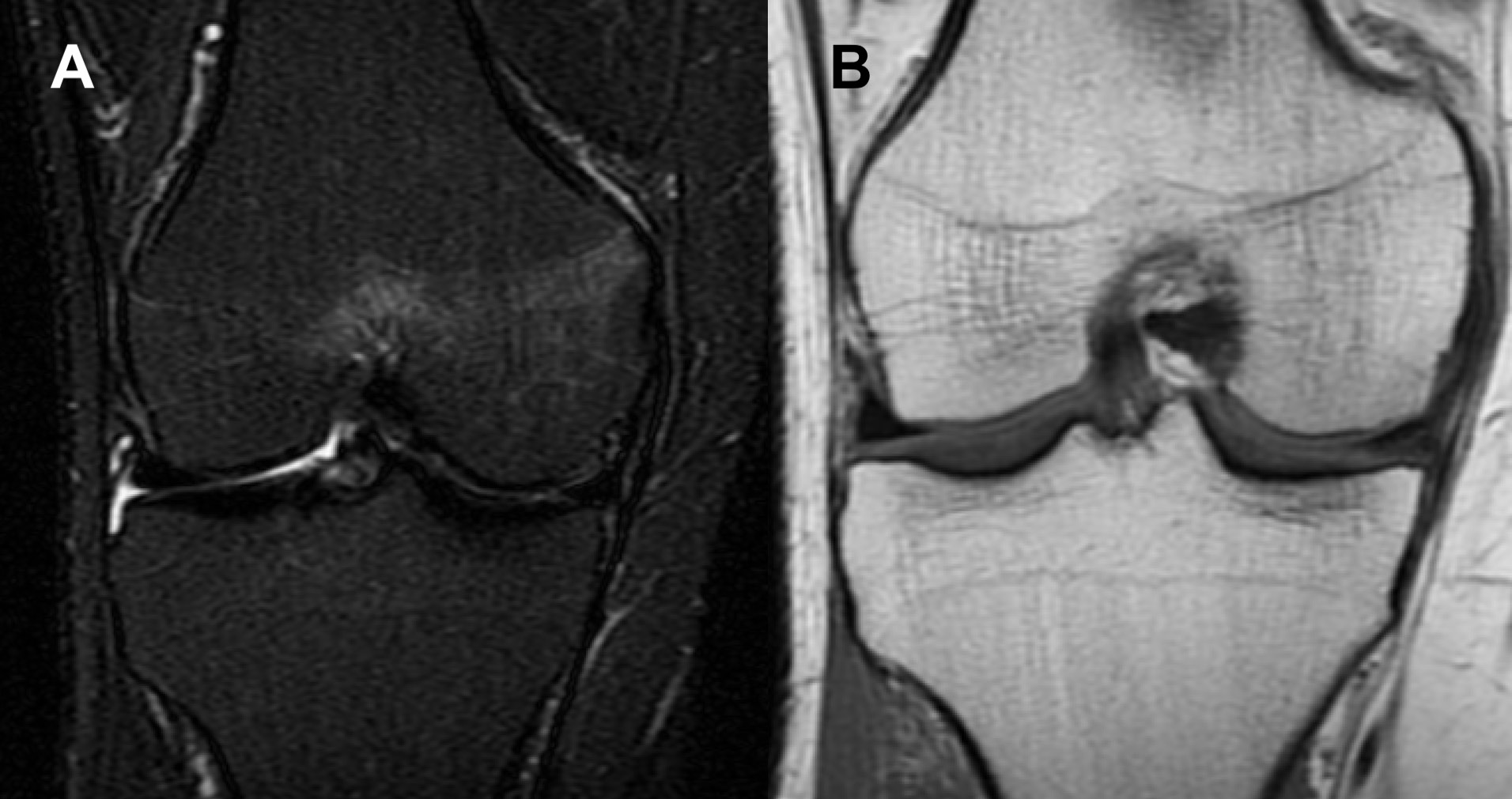
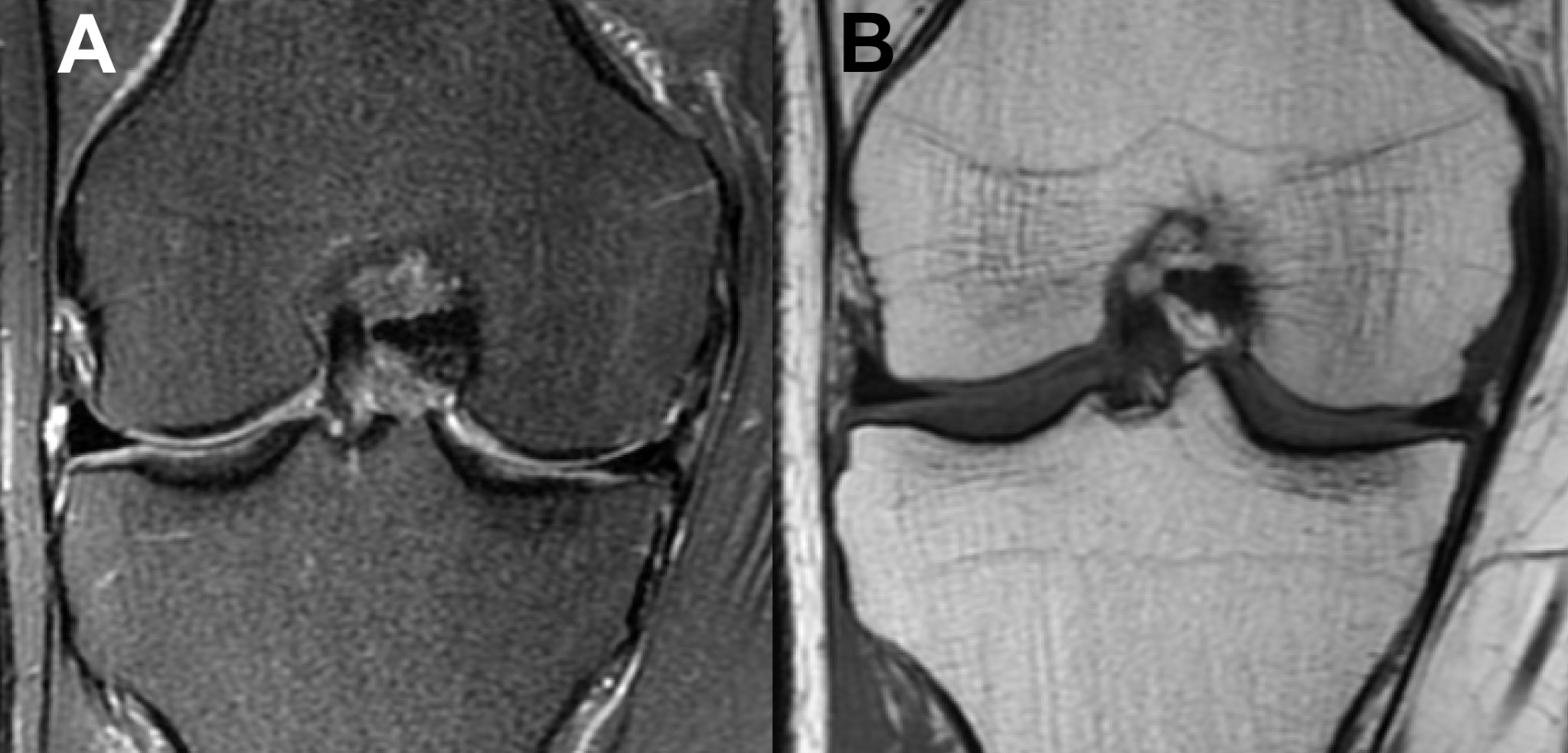
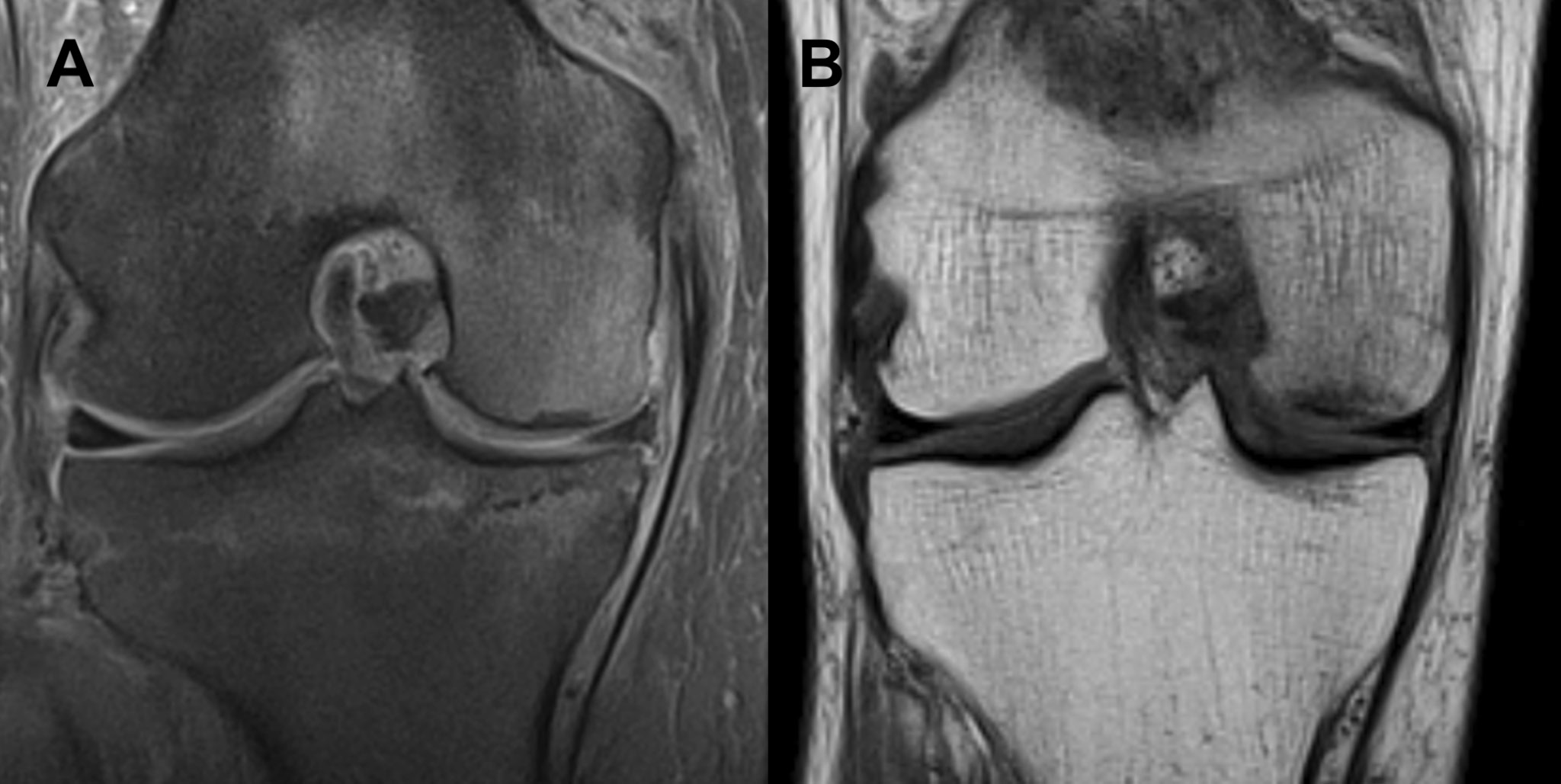
The symptoms subsided completely after approx. 8 weeks of nonoperative treatment. A follow-up MRI scan of the same knee was performed 10 weeks after the first appointment, and showed remission of the radiological findings in the MFC and medial aspect of the intercondylar region (Figure 2). The next follow-up MRI, which was performed 5 months after the first appointment, showed complete resolution of the bony and soft tissue edema (Figure 3). There were no adverse or unanticipated events.
Case 2 (K.M.)
A 47-year-old male hotel owner presented with sudden onset of right knee pain without trauma. The pain persisted during rest and at night.
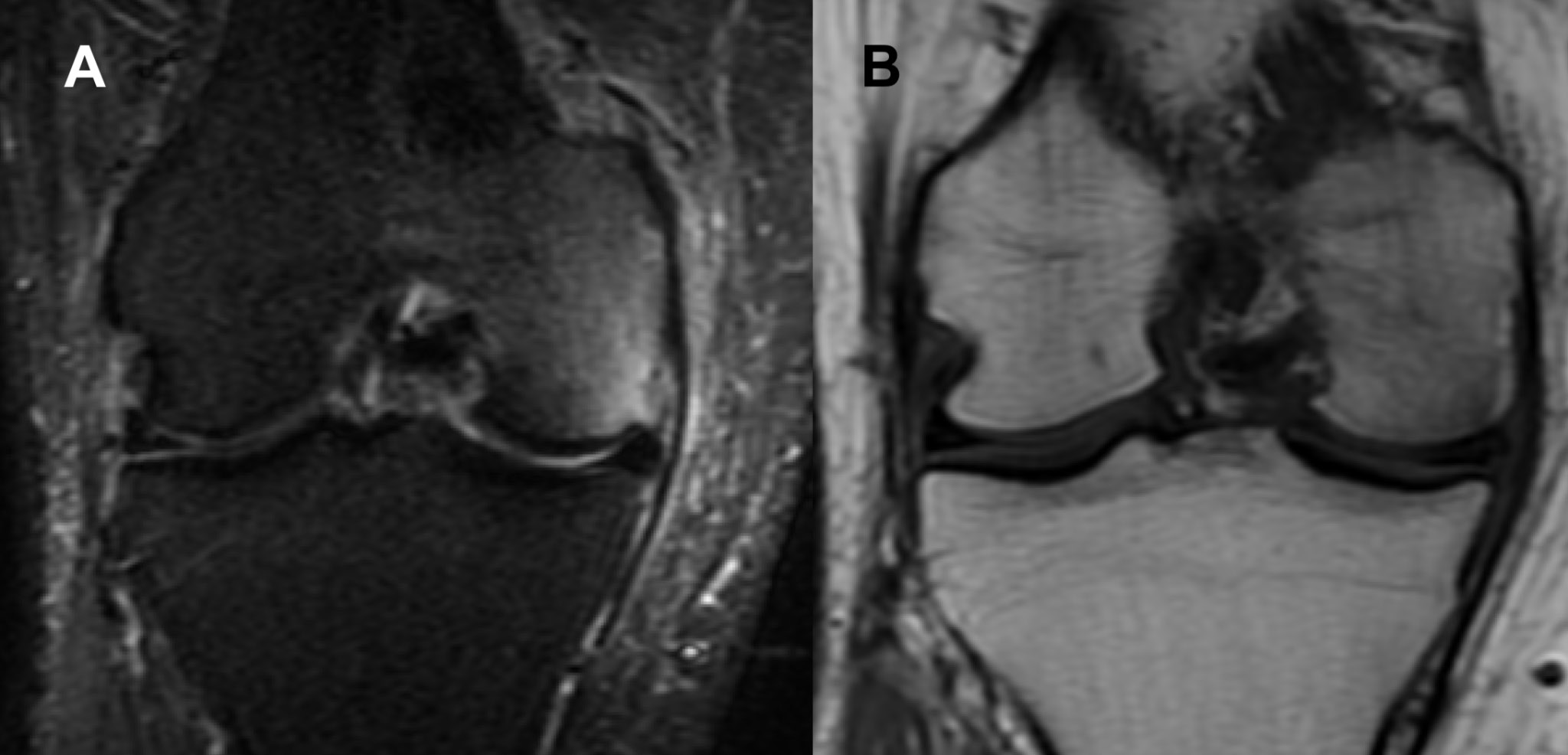
On clinical examination, the patient had a full range of motion, with a stable joint, with pain during extension and near full extension. In addition, the patient experienced pain upon palpation of the MFC and joint spaces of the affected knee. Varus stress test applied to the knee with the leg in 10° flexion also elicited pain. The knee axis was recorded as less than 5° of varus. Magnetic resonance imaging of the knee 4 weeks after the onset of symptoms revealed diffuse high signal in the entire MFC, suggesting an accumulation of edema in the bone marrow (Figure 4).
Approximately 10 weeks before presentation, the patient had tested positive for SARS-CoV-2 with real-time polymerase chain reaction (RT-PCR) test. He had experienced severe flu-like symptoms, with fever as high as 39.5°C, and pneumonia that lasted 10 days. He was not hospitalized, and corticosteroids were not administered.
The patient was put on crutches and instructed to bear no weight on the affected leg for 6 weeks. Additionally, he was prescribed the same pharmacotherapy as the patient in Case 1.
Full remission of symptoms was achieved after approx. 3 weeks of such conservative treatment. Follow-up MRI 10 weeks after presentation revealed near-complete loss of signal in the MFC (Figure 5). There were no adverse or unanticipated events.
Discussion
The 2 cases of PCT-SONK presented in this study add to the differential diagnosis of sudden knee pain in uninjured patients during the ongoing pandemic. It is particularly noteworthy that the radiological findings that constitute PCT-SONK, although similar in both cases presented in this study, are radiologically distinct in appearance from classic SONK (Figure 6). No subchondral bone thickening was observed, and the bone edema was more diffuse in the whole femoral condyle, in contrast to focal edema, which is concentrated mainly around the subchondral region and typically observed in classic SONK (Table 1).
It has been reported that SONK classically occurs in middle-aged females and is caused by excessive stress in the subchondral bone.14 In previously documented cases of SONK unrelated to COVID-19, high rates of surgical intervention, including total knee replacement, resulted in poor prognoses.9 However, in the 2 cases presented in this study, nonoperative treatment yielded good results. The transient nature of PCT-SONK suggests that surgical management should be considered only for progressive cases in which conservative management fails to achieve improvement. Thus, it is of utmost importance, particularly during the pandemic, to raise awareness among sports medicine and orthopedic societies to consider microvascular impairment in subchondral bone as a possible source of sudden knee pain in patients without concurrent trauma or a history of recent injury.
While SONK after COVID-19 in the presented cases may indeed be a coincidence, SARS-CoV-2 infection has been demonstrated to affect nearly every facet of hemostasis. This is likely driven by a combination of a dysfunctional, hyperinflammatory state and direct and indirect endothelial injury, ultimately leading to micro-immunothrombosis both in the lungs and systemic tissues.11, 15 Significant evidence of endotheliopathy and a secondary thrombotic microangiopathy (TMA) or TMA-like phenomenon has emerged. It is often characterized by elevated von Willebrand factor antigen levels and low ADAMTS13 activity,12, 16 overactivation of complement pathways,17 elevated circulating markers of neutrophil extracellular traps,18 high serum lactate dehydrogenase levels,19 and decreased platelet counts,20, 21 as well as the typical clinical manifestations of TMA, such as acute kidney failure.22 Moreover, evidence of SARS-CoV-2 directly infecting endothelial cells has been presented,23 and circulating biomarkers of endothelial injury are frequently reported to be elevated in COVID-19 patients.24 Finally, a TMA-like phenomenon is likely further exacerbated by a state of hypofibrinolysis, impairing the ability to break down newly formed clots in the microcirculation.25, 26
Given the significant potential for multisystem microvascular injury in COVID-19, it can be hypothesized that there is an increased risk of SONK related to COVID-19 – secondary to damage to blood vessels supplying the MFC regions, leading to impaired microcirculation blood flow and tissue perfusion, and ultimately causing ischemic necrosis of subchondral bone.27, 28 Such phenomenon may occur more often in patients with pre-existing, but clinically silent, excessive subchondral bone stress, with SARS-CoV-2 microvascular flow disturbances exacerbating the underlying knee pathology.
Corticosteroids are widely used, sometimes indiscriminately, for the treatment of COVID-19. They could lead to a reduction in bone mineral density (BMD) in patients and subsequently predispose them to SONK, as low BMD has been shown to be associated with the onset of SONK,29, 30 as well as other orthopedic pathologies, such as steroid-induced avascular necrosis of the femoral head.31 However, unlike in many other studies reporting post-COVID-19 osteonecrosis,5, 6, 7 corticosteroids were not administered in either of the cases in this study. As stated in a study by Angulo-Ardoy et al., “it is still unclear whether post-COVID SONK is related to the use of corticosteroids or to the virus itself”.6 While some studies report the development of osteonecrosis after lower doses of corticosteroids, and sooner after their administration than in comparable non-COVID-19 cases,5, 6, 7 this is the first study to report 2 cases with no corticosteroid administration at all. Therefore, the authors believe it adds to the body of knowledge on the potential connections between COVID-19 and PCT-SONK.
A conservative treatment plan should address all etiologic aspects of PCT-SONK. First, reduction of pressure to the affected subchondral bone is achieved by non-weight bearing; second, supplementation of vitamin D and calcium is implemented to enhance BMD restoration; third, pharmacological agents to improve microvascular circulation (i.e., pentoxyfilline) are administered; and finally, anti-aggregative agents, such as acetylsalicylic acid, to inhibit thrombus formation, are given to the patient.32 It is important to emphasize that this approach is supported only by limited observations and based on regular SONK treatment algorithms; further studies utilizing evidence-based methods are required. We emphasize a personalized approach in every case, considering a patient’s general health status and all possible individual contraindications and risks of given therapies.
This study is limited by a relatively short follow-up, although complete radiological and clinical recovery was obtained during the observation period. While this study has no power to identify any causative relationship with SARS-CoV-2, we hope that it will raise awareness and pave the way for broader studies investigating the PCT-SONK phenomenon.
Conclusions
Orthopedic surgeons should be cautious when sudden knee pain without concurrent trauma or a history of injury occurs shortly after SARS-CoV-2 infection, even when the course of COVID-19 was mild. While there are studies reporting post-COVID-19 osteonecrosis after lower doses of corticosteroids and sooner after their administration than in comparable non-COVID-19 cases, this is the first to report 2 cases with no corticosteroid administration at all. The transient nature of symptoms and radiological findings suggest that aggressive surgical treatment of non-injury local bone edema occurring shortly after SARS-CoV-2 infection should be avoided.